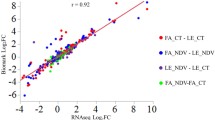
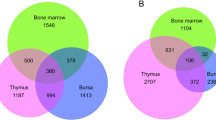
Abstract
Campylobacter jejuni (C. jejuni) is a leading cause of human bacterial enteritis worldwide with poultry products being a major source of C. jejuni contamination. The chicken is the natural reservoir of C. jejuni where bacteria colonize the digestive tract of poultry, but rarely cause symptoms of disease. To understand the systemic molecular response mechanisms to C. jejuni infection in chickens, total splenic RNA was isolated and applied to a whole genome chicken microarray for comparison between infected (I) and non-infected (N) chickens within and between genetic lines A and B. There were more total splenic host genes responding to the infection in resistant line A than in susceptible line B. Specifically, genes for lymphocyte activation, differentiation and humoral response, and Ig light and heavy chain were upregulated in the resistant line. In the susceptible line, genes for regulation of erythrocyte differentiation, hemopoiesis, and RNA biosynthetic process were all downregulated. An interaction analysis between genetic lines and treatment demonstrated distinct defense mechanisms between lines: the resistant line promoted apoptosis and cytochrome c release from mitochondria, whereas the susceptible line responded with a downregulation of both functions. This was the first time that such systemic defensive mechanisms against C. jejuni infection have been reported. The results of this study revealed novel molecular mechanisms of the systemic host responses to C. jejuni infection in chickens that warrant further investigation.




Similar content being viewed by others
References
Acar JF, Goldstein FW (1997) Trends in bacterial resistance to fluoroquinolones. Clin Infect Dis 24(Suppl 1):S67–S73
Al-Shahrour F, Diaz-Uriarte R, Dopazo J (2004) FatiGO: a web tool for finding significant associations of gene ontology terms with groups of genes. Bioinformatics 20(4):578–580
Altekruse SF, Stern NJ, Fields PI, Swerdlow DL (1999) Campylobacter jejuni—an emerging foodborne pathogen. Emerg Infect Dis 5(1):28–35
Barrett T, Troup DB, Wilhite SE, Ledoux P, Rudnev D, Evangelista C, Kim IF, Soboleva A, Tomashevsky M, Edgar R (2007) NCBI GEO: mining tens of millions of expression profiles—database and tools update. Nucleic Acids Res 35(Database issue):D760–D765
Beery JT, Hugdahl MB, Doyle MP (1988) Colonization of gastrointestinal tracts of chicks by Campylobacter jejuni. Appl Environ Microbiol 54(10):2365–2370
Bingham-Ramos LK, Ribardo DA, Hiendrixson DR (2008) Colonization characteristics of Campylobacter jejuni in a natural avian host 108th general meeting of American Society for Microbiology. Boston, MA, pp. D-047.
Boyd Y, Herbert EG, Marston KL, Jones MA, Barrow PA (2005) Host genes affect intestinal colonisation of newly hatched chickens by Campylobacter jejuni. Immunogenetics 57(3–4):248–253
Cawthraw S, Ayling R, Nuijten P, Wassenar T, Newell D (1994) Isotype, specificity, and kinetics of systemic an mucosal antibodies to Campylobacter jejuni antigens, including flagellin, during experimental oral infections of chickens. Avian Dis 38:341–349
Cleveland WS (1974) Robust locally weighted regression and smoothing scatterplots. J Am Stat Assoc 74:829–836
Cox NA, Richardson LJ, Buhr RJ, Bailey JS, Wilson JL, Hiett KL (2006) Detection of Campylobacter jejuni in various lymphoid organs of broiler breeder hens after oral or intravaginal inoculation. Poult Sci 85(8):1378–1382
de Vries FWTP (1975) The cost of maintenance processes in plant cells. Ann Bot 39(1):77–92
Dennis G Jr, Sherman BT, Hosack DA, Yang J, Gao W, Lane HC, Lempicki RA (2003) DAVID: database for annotation, visualization, and integrated discovery. Genome Biol 4(5):P3
Diaz de Tuesta AM, Chow Q, Geijo Martinez MP, Dimas Nunez J, Diaz de Tuesta FJ, Herranz CR, Val Perez E (2001) Tularemia outbreak in the province of Cuenca associated with crab handling. Rev Clin Esp 201(7):385–389
Faherty CS, Maurelli AT (2008) Staying alive: bacterial inhibition of apoptosis during infection. Trends Microbiol 16(4):173–180
Ferro PJ, Swaggerty CL, Kaiser P, Pevzner IY, Kogut MH (2004) Heterophils isolated from chickens resistant to extra-intestinal Salmonella enteritidis infection express higher levels of pro-inflammatory cytokine mRNA following infection than heterophils from susceptible chickens. Epidemiol Infect 132(6):1029–1037
Gaillard J, Finlay B (1996) Effect of cell polarization and differentiation on entry of Listeria monocytogenes into the enterocyte-like Caco-2 cell line. Infect Immun 64(4):1299–1308
Gruntar I, Ocepek M, Avbersek J, Micunovic J, Pate M (2010) A pulsed-field gel electrophoresis study of the genetic diversity of Campylobacter jejuni and Campylobacter coli in poultry flocks in Slovenia. Acta Vet Hung 58(1):19–28
Hu L, Kopecko DJ (1999) Campylobacter jejuni 81–176 associates with microtubules and dynein during invasion of human intestinal cells. Infect Immun 67(8):4171–4182
Hu Z, Bao J, Reecy JM (2008) CateGOrizer: a web-based program to batch analyze gene ontology classification categories. Online Journal of Bioinformatics 9(2):108–112
Huang DW, Sherman BT, Lempicki RA (2009) Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat Protocols 4(1):44–57
Janicke RU, Ng P, Sprengart ML, Porter AG (1998) Caspase-3 is required for alpha-fodrin cleavage but dispensable for cleavage of other death substrates in apoptosis. J Biol Chem 273(25):15540–15545
Jeurissen SH, Janse EM, van Rooijen N, Claassen E (1998) Inadequate anti-polysaccharide antibody responses in the chicken. Immunobiology 198(4):385–95
Kopecko DJ, Hu L, Zaal KJ (2001) Campylobacter jejuni—microtubule-dependent invasion. Trends Microbiol 9(8):389–396
Lehrer RI, Ganz T (2002) Defensins of vertebrate animals. Curr Opin Immunol 14(1):96–102
Li X, Chiang HI, Zhu J, Dowd SE, Zhou H (2008a) Characterization of a newly developed chicken 44K Agilent microarray. BMC Genomics 9(1):60
Li X, Swaggerty CL, Kogut MH, Chiang H, Wang Y, Genovese KJ, He H, Stern NJ, Pevzner IY, Zhou H (2008b) The paternal effect of Campylobacter jejuni colonization in ceca in broilers. Poult Sci 87(9):1742–1747
Li X, Swaggerty CL, Kogut MH, Chiang HI, Wang Y, Genovese KJ, He H, Zhou H (2010) Gene expression profiling of the local cecal response of genetic chicken lines that differ in their susceptibility to Campylobacter jejuni colonization. PLoS One 5(7):e11827
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(−Delta Delta C(T)) Method. Methods 25:402–408
Lynn DJ, Higgs R, Gaines S, Tierney J, James T, Lloyd AT, Fares MA, Mulcahy G, O’Farrelly C (2004) Bioinformatic discovery and initial characterisation of nine novel antimicrobial peptide genes in the chicken. Immunogenetics 56(3):170–177
Lynn DJ, Higgs R, Lloyd AT, O’Farrelly C, Herve-Grepinet V, Nys Y, Brinkman FS, Yu PL, Soulier A, Kaiser P, Zhang G, Lehrer RI (2007) Avian beta-defensin nomenclature: a community proposed update. Immunol Lett 110(1):86–89
Meade KG, Narciandi F, Cahalane S, Reiman C, Allan B, O’Farrelly C (2008) Comparative in vivo infection models yield insights on early host immune response to Campylobacter in chickens. Immunogenetics 61(2):101–110
Meade KG, Narciandi F, Cahalane S, Reiman C, Allan B, O’Farrelly C (2009) Comparative in vivo infection models yield insights on early host immune response to Campylobacter in chickens. Immunogenetics 61(2):101–110
Mills SD, Finlay BB (1994) Comparison of Salmonella typhi and Salmonella typhimurium invasion, intracellular growth and localization in cultured human epithelial cells. Microb Pathog 17(6):409–423
Murphy KM, Travers JP, Walport M (2007) Janeway’s immunobiology (immunobiology: the immune system (Janeway)). Garland Science, New York
Smith CK, Abuoun M, Cawthraw SA, Humphrey TJ, Rothwell L, Kaiser P, Barrow PA, Jones MA (2008) Campylobacter colonization of the chicken induces a proinflammatory response in mucosal tissues. FEMS Immunol Med Microbiol 54(1):114–21
Stern NJ, Meinersmann RJ, Cox NA, Bailey JS, Blankenship LC (1990) Influence of host lineage on cecal colonization by Campylobacter jejuni in chickens. Avian Dis 34(3):602–606
Storey JD, Tibshirani R (2003) Statistical significance for genomewide studies. Proc Natl Acad Sci U S A 100(16):9440–9445
Swaggerty CL, Ferro PJ, Pevzner IY, Kogut MH (2005a) Heterophils are associated with resistance to systemic Salmonella enteritidis infections in genetically distinct chicken lines. FEMS Immunol Med Microbiol 43(2):149–154
Swaggerty CL, Lowry VK, Ferro PJ, Pevzner IY, Kogut MH (2005b) Disparity in susceptibility to vancomycin-resistant Enterococcus organ invasion in commercial broiler chickens that differ in innate immune responsiveness. Food & Agricultural Immunology 16(1):1–15
Swaggerty CL, Genovese KJ, He H, Duke SE, Pevzner IY, Kogut MH (2011) Broiler breeders with an efficient innate immune response are more resistant to Eimeria tenella. Poult Sci 90(5):1014–1019
van Putten JP, van Alphen LB, Wosten MM, de Zoete MR (2009) Molecular mechanisms of campylobacter infection. Curr Top Microbiol Immunol 337:197–229
Wickstrum JR, Hong KJ, Bokhari S, Reed N, McWilliams N, Horvat RT, Parmely MJ (2007) Coactivating signals for the hepatic lymphocyte gamma interferon response to Francisella tularensis. Infect Immun 75(3):1335–1342
Yang YH, Dudoit S, Luu P, Lin DM, Peng V, Ngai J, Speed TP (2002) Normalization for cDNA microarray data: a robust composite method addressing single and multiple slide systematic variation. Nucleic Acids Res 30(4):e15
Zilbauer M, Dorrell N, Boughan PK, Harris A, Wren BW, Klein NJ, Bajaj-Elliott M (2005) Intestinal innate immunity to Campylobacter jejuni results in induction of bactericidal human beta-defensins 2 and 3. Infect Immun 73(11):7281–7289
Acknowledgments
The authors thank Norman J. Stern from Poultry Microbiological Safety Research Unit, USDA, Athens, GA for kindly providing the bacterial strain. This project was supported by National Research Initiative Grant no. 2007–35604–17903 from the USDA Cooperative State Research, Education, and Extension Service Animal Genome program. The mention of commercial products is for the sole purpose of providing specific information and not a recommendation or endorsement by the USDA.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Xianyao Li and Christina L. Swaggerty contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplemental file 1
Overlapped genes between AI/AN and BI/BN. This table provides a list of overlapped genes between AI/AN and BI/BN (XLS 54 kb)
Supplemental file 2
Fold enrichment for a significant gene in the comparison of AI/AN. This table shows the enriched GO BP terms for the differentially expressed genes in the comparison of AI/AN (XLS 38 kb)
Rights and permissions
About this article
Cite this article
Li, X., Swaggerty, C.L., Kogut, M.H. et al. Systemic response to Campylobacter jejuni infection by profiling gene transcription in the spleens of two genetic lines of chickens. Immunogenetics 64, 59–69 (2012). https://doi.org/10.1007/s00251-011-0557-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00251-011-0557-1