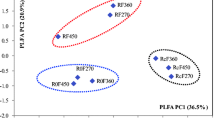
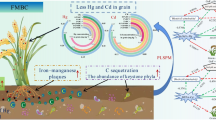
Abstract
Soil bacterial and fungal communities are suitable soil ecosystem health indicators due to their sensitivity to management practices and their role in soil ecosystem processes. Here, information on composition and functions of bacterial and fungal communities were evaluated at two phenological stages of sugarcane (six and twelve months, equivalent to the most intensive vegetative stage and to final maturation, respectively) when organomineral fertilizer, combined with phosphate-solubilizing bacteria (PSB), was added into the soil. Organic compost enriched with apatite (C + A) or phosphorite (C + P) and compost without phosphate enrichment (C) were used in the presence or absence of PSB. In addition, we used a control fertilized with soluble triple superphosphate. The differences were more related to the sampling period than to the type of organomineral fertilizer, being observed higher available phosphorus at six months than at twelve months. Only in the C treatment we observed the presence of Bacillaceae and Planococcaceae, while Pseudomonadaceae were only prevalent in inoculated C + A. As for fungi, the genera Chaetomium and Achroiostachys were only present in inoculated C + P, while the genus Naganishia was most evident in inoculated C + A and in uninoculated C + P. Soliccocozyma represented 75% of the total fungal abundance in uninoculated C while in inoculated C, it represented 45%. The bacterial community was more related to the degradation of easily decomposable organic compounds, while the fungal community was more related to degradation of complex organic compounds. Although the microbial community showed a resilient trait, subtle changes were detected in microbial community composition and function, and this may be related to the increase in yield observed.





Similar content being viewed by others
Data Availability
The authors declare that the data will be available when requested.
Code Availability
Not applicable.
References
Tilman D, Balzer C, Hill J, Befort BL (2011) Global food demand and the sustainable intensification of agriculture. PNAS 108:20260–20264. https://doi.org/10.1073/pnas.1116437108
Jacoby R, Peukert M, Succurro A, Koprivova A, Kopriva S (2017) The role of soil microorganisms in plant mineral nutrition—current knowledge and future directions. Front Plant Sci 8:1617. https://doi.org/10.3389/fpls.2017.01617
Mitter EK, Tosi M, Obregón D, Dunfield KE, Germida JJ (2021) Rethinking crop nutrition in times of modern microbiology: innovative biofertilizer technologies. Front Sustain Food Syst 5:29. https://doi.org/10.3389/fsufs.2021.606815
Muller A, Schader C, El-Hage Scialabba N, Brüggemann J, Isensee A, Erb KH, Smith P, Klocke P, Leiber F, Stolze M, Niggli U (2017) Strategies for feeding the world more sustainably with organic agriculture. Nat Commun 8:1290. https://doi.org/10.1038/s41467-017-01410-w
Mącik M, Gryta A, Frąc M (2020) Biofertilizers in agriculture: an overview on concepts, strategies and effects on soil microorganisms. Adv Agron 162:31–87. https://doi.org/10.1016/bs.agron.2020.02.001
Moraes ER, Mageste JG, Lana RMQ, Da Silva RV, Camargo R (2017) Sugarcane: organo-mineral fertilizers and biostimulants. In: Oliveria AB (ed) Sugarcane-Technology and Research. IntechOpen, London, pp 193–206
Borges BMMN, Abdala DB, Souze MF, Viglio LM, Coelho MJA, Pavinato PS, Frango HCJ (2019) Organomineral phosphate fertilizer from sugarcane byproduct and its effects on soil phosphorus availability and sugarcane yield. Geoderma 339:20–30. https://doi.org/10.1016/j.geoderma.2018.12.036
Crusciol CAC, Campos MD, Martello JM, Alves CJ, Nascimento CAC, Pereira JCR, Cantarella H (2020) Organomineral fertilizer as source of P and K for sugarcane. Sci Rep 10:5398. https://doi.org/10.1038/s41598-020-62315-1
Zhao Y, Sun Y, Pei M, Fu J, Zhao L, Xiao X (2021) Enhanced rice yields are related to pronounced shifts in soil resident bacterial community structures in response to Rhodopseudomonas palustris and Bacillus subtilis inoculation. J Soils Sediments 21:2369–2380. https://doi.org/10.1007/s11368-021-02929-8
Sharma SB, Sayyed RZ, Trivedi MH, Gobi TA (2013) Phosphate solubilizing microbes: sustainable approach for managing phosphorus deficiency in agricultural soils. Springerplus 2:587. https://doi.org/10.1186/2193-1801-2-587
Granada CE, Passaglia LM, de Souza EM, Sperotto RA (2018) Is phosphate solubilization the forgotten child of plant growth-promoting rhizobacteria? Front Microbiol 9:2054. https://doi.org/10.3389/fmicb.2018.02054
Alori ET, Glick BR, Babalola OO (2017) Microbial phosphorus solubilization and its potential for use in sustainable agriculture. Front Microbiol 8:971. https://doi.org/10.3389/fmicb.2017.00971
Dhillon J, Torres G, Driver E, Figueiredo B, Raun WR (2017) World phosphorus use efficiency in cereal crops. Agron J 109:1670–1677. https://doi.org/10.2134/agronj2016.08.0483
Soltangheisi A, Withers PJ, Pavinato PS, Cherubin MR, Rossetto R, Do Carmo JB, Rocha GC, Martinelli LA (2019) Improving phosphorus sustainability of sugarcane production in Brazil. GCB Bionergy 11:1444–1455. https://doi.org/10.1111/gcbb.12650
Pavinato PS, Cherubin MR, Soltangheisi A, Rocha GC, Chadwick DR, Jones DL (2020) Revealing soil legacy phosphorus to promote sustainable agriculture in Brazil. Sci Rep 10:15615. https://doi.org/10.1038/s41598-020-72302-1
Raymond NS, Gómez-Muñoz B, van der Bom FJ et al (2021) Phosphate-solubilising microorganisms for improved crop productivity: a critical assessment. New Phytol 229:1268–1277. https://doi.org/10.1111/nph.16924
Bononi L, Chiaramonte JB, Pansa CC et al (2020) Phosphorus-solubilizing Trichoderma spp. from Amazon soils improve soybean plant growth. Sci Rep 10:2858. https://doi.org/10.1038/s41598-020-59793-8
Ditta A, Imtiaz M, Mehmood S, Rizwan MS et al (2018) Rock phosphate-enriched organic fertilizer with phosphate-solubilizing microorganisms improves nodulation, growth, and yield of legumes. Commun Soil Sci Plant Anal 49:2715–2725. https://doi.org/10.1080/00103624.2018.1538374
Ditta A, Arshad M, Zahir ZA, Jamil A (2015) Comparative efficacy of rock phosphate enriched organic fertilizer vs. mineral phosphatic fertilizer for nodulation, growth and yield of lentil. Int J Agric Biol 17:589–595. https://doi.org/10.17957/IJAB/17.3.14.954
Ditta A, Muhammad J, Imtiaz M et al (2018) Application of rock phosphate enriched composts increases nodulation, growth and yield of chickpea. Int J Recycl Org Waste Agricult 7:33–40. https://doi.org/10.1007/s40093-017-0187-1
Elhaissoufi W, Khourchi S, Ibnyasser A, Ghoulam C et al (2020) Phosphate solubilizing rhizobacteria could have a stronger influence on wheat root traits and aboveground physiology than rhizosphere P solubilization. Front Plant Sci 11:979. https://doi.org/10.3389/fpls.2020.00979
Alori ET, Babalola OO (2018) Microbial inoculants for improving crop quality and human health in Africa. Front Microbiol 9:2213. https://doi.org/10.3389/fmicb.2018.02213
Sundara B, Natarajan V, Hari K (2002) Influence of phosphorus solubilizing bacteria on the changes in soil available phosphorus and sugarcane and sugar yields. Field Crops Res 77:43–49. https://doi.org/10.1016/S0378-4290(02)00048-5
Rosa PAL, Mortinho ES, Jalal A, Galindo FS, Buzetti S, Fernandes GC, Barco Neto M, Pavinato OS, Teixeira Filho MCM (2020) Inoculation with growth-promoting bacteria associated with the reduction of phosphate fertilization in sugarcane. Front environ sci 8:32. https://doi.org/10.3389/fenvs.2020.00032
Mawarda PC, Le Roux X, Dirk van Elsas J, Salles JF (2020) Deliberate introduction of invisible invaders: a critical appraisal of the impact of microbial inoculants on soil microbial communities. Soil Biol Biochem 148:107874. https://doi.org/10.1016/j.soilbio.2020.107874
Jack CN, Petipas RH, Cheeke TE, Rowland JL, Friesen ML (2020) Microbial inoculants: silver bullet or microbial Jurassic Park? Trends Microbiol 4:299–208. https://doi.org/10.1016/j.tim.2020.11.006
Wang Z, Chen Z, Kowalchuk GA, Xu Z, Fu X, Kuramae EE (2021) Succession of the resident soil microbial community in response to periodic inoculations. AEM 87:e00046-e121. https://doi.org/10.1128/AEM.00046-21
Estrada-Bonilla GA, Durrer A, Cardoso EJBN (2021) Use of compost and phosphate-solubilizing bacteria affect sugarcane mineral nutrition, phosphorus availability, and the soil bacterial community. Appl Soil Ecol 157:103760. https://doi.org/10.1016/j.apsoil.2020.103760
Lopes CM, Silva AMM, Estrada-Bonilla GA, Ferraz-Almeida R, Vieira JLV, Otto R, Vitti GC, Cardoso EJBN (2021) Improving the fertilizer value of sugarcane wastes through phosphate rock amendment and phosphate-solubilizing bacteria inoculation. J Clean Prod 298:126821. https://doi.org/10.1016/j.jclepro.2021.126821
Soil Science Division Staff (2017) C. Ditzler, K. Scheffe, and H.C. Monger (ed) Soil survey manual. USDA Handbook 18. Government Printing Office, Washington, D.C. Accessed 13 Nov 2020
Embrapa (2018) Brazilian system of soil classification, 5th edn. Embrapa, Rio Janeiro, p 356
Estrada-Bonilla GA, Lopes CM, Durrer A, Alves PRL, Passaglia N, Cardoso EJBN (2017) Effect of phosphate-solubilizing bacteria on phosphorus dynamics and the bacterial community during composting of sugarcane industry waste. Syst Appl Microbiol 40:308–313. https://doi.org/10.1016/j.syapm.2017.05.003
Scarpare FV, Hernandes TAD, Ruiz-Corrêa ST, Picoli MCA, Scanlon BR, Chagas MF, Duft DG, de Fátima CT (2016) Sugarcane land use and water resources assessment in the expansion area in Brazil. J Clean Prod 133:1318–1327. https://doi.org/10.1016/j.jclepro.2016.06.074
INMET - Instituto Nacional de Meteorologia [National Institute of Meteorology]. https://bdmep.inmet.gov.br/. Accessed 27 Feb 2018
van Raij B, Andrade JC, Cantarella H, Quaggio JÁ (2001) Análise química para avaliação da fertilidade de solos tropicais = Chemical analysis for fertility assessment of tropical soils. Instituto Agronômico de Campinas, Campinas
Walkley AJ, Black IA (1934) Estimation of soil organic carbon by the chromic acid titration method. Soil Sci 37:29–38
Muyzer G (1993) Profiling of complex microbial populations by denaturing gradient gel electrophoresis analysis of polymerase chain reaction-amplified genes coding for 16S rRNA. AEM 59:695–700
Gardes M, Bruns TD (1993) ITS primers with enhanced specificity for basidiomycetes – application to the identification of mycorrhizae and rusts. Mol Ecol 2:113–118. https://doi.org/10.1111/j.1365-294X.1993.tb00005.x
Sakurai M, Wasaki J, Tomizawa Y, Shinano T, Osaki M (2008) Analysis of bacterial communities on alkaline phosphatase in soil supplied with organic matter. Soil Sci Plant Nutr 54:62–71. https://doi.org/10.1111/j.1747-0765.2007.00210.x
Vollú R, Cotta SR, Jurelevicius D, Leite DCDA, Parente CET, Malm O, Martins DC, Resende AV, Marriel IE, Seldin L (2018) Response of the bacterial communities associated with maize rhizosphere to poultry litter as an organomineral fertilizer. Front Environ Sci 6:118. https://doi.org/10.3389/fenvs.2018.00118
Gaiero JR, Tosi M, Bent E, Boitt G, Khosla K, Turner BL, Richardson AE, Condron LM, Dunfield KE (2021) Soil microbial communities influencing organic phosphorus mineralization in a coastal dune chronosequence in New Zealand. FEMS Microbiol Ecol 97:fiab034. https://doi.org/10.1093/femsec/fiab034
Bolyen E, Rideout JR, Dillon MR et al (2019) Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nat Biotechnol 37:852–857. https://doi.org/10.1038/s41587-019-0209-9
Callahan BJ, McMurdie PJ, Rosen MJ, Han AW, Johnson AJA, Holmes SP (2016) DADA2: high-resolution sample inference from Illumina amplicon data. Nat Methods 13:581–583. https://doi.org/10.1038/nmeth.3869
Katoh K (2002) MAFFT: a novel method for rapid multiple sequence alignment based on fast Fourier transform. Nucleic Acids Res 30:3059–3066. https://doi.org/10.1093/nar/gkf436
Bokulich NA, Kaehler BD, Rideout JR et al (2018) Optimizing taxonomic classification of marker-gene amplicon sequences with QIIME 2’s q2-feature-classifier plugin. Microbiome 6:1–17. https://doi.org/10.1186/s40168-018-0470-z
Quast C, Pruesse E, Yilmaz P, Gerken J, Schweer T, Yarka P, Peplies J, Glöckner FO (2013) The SILVA ribosomal RNA gene database project: improved data processing and web-based tools. Nucleic Acids Res 41:590–596. https://doi.org/10.1093/nar/gks1219
Nilsson RH, Larsson KH, Taylor AFS, Bengtsson-Palme J, Jeppsen TS, Schigel D, Kennedy P, Picard K, Glöckner FO, Tedersoo L, Saar I, Kõljalg U, Abarenkov K (2019) The UNITE database for molecular identification of fungi: handling dark taxa and parallel taxonomic classifications. Nucleic Acids Res 47:D259–D264. https://doi.org/10.1093/nar/gky1022
Douglas GM, Maffei VJ, Zaneveld JR et al (2020) PICRUSt2 for prediction of metagenome functions. Nat Biotechnol 38:685–688. https://doi.org/10.1038/s41587-020-0548-6
Trivedi P, Delgado-Baquerizo M, Trivedi C, Hu H, Anderson IC, Jeffries TC, Zhou J, Singh BK (2016) Microbial regulation of the soil carbon cycle: evidence from gene–enzyme relationships. ISME J 10:2593–2604. https://doi.org/10.1038/ismej.2016.65
Liang JL, Liu J, Jia P, Yang TT, Zeng QW, Zhang SC, Liao B, Shu WS, Li JT (2020) Novel phosphate-solubilizing bacteria enhance soil phosphorus cycling following ecological restoration of land degraded by mining. ISME J 14:1600–1613. https://doi.org/10.1038/s41396-020-0632-4
Nguyen NH, Song Z, Bates ST, Branco S, Tedersoo L, Menke J, Schilling js, Kennedy PG, (2016) FUNGuild: an open annotation tool for parsing fungal community datasets by ecological guild. Fungal Ecol 20:241–248. https://doi.org/10.1016/j.funeco.2015.06.006
McMurdie PJ, Holmes S (2014) Waste not, want not: why rarefying microbiome data is inadmissible. PLoS Comput Biol 10:e1003531. https://doi.org/10.1371/journal.pcbi.1003531
McMurdie PJ, Holmes S (2013) phyloseq: an R package for reproducible interactive analysis and graphics of microbiome census data. PLoS ONE 8:e61217. https://doi.org/10.1371/journal.pone.0061217
Bisanz JE (2019) Tutorial : integrating QIIME2 and R for data visualization and analysis using. January, 2021
Faith DP (1992) Conservation evaluation and phylogenetic diversity. Biol Conserv 61:1–10. https://doi.org/10.1016/0006-3207(92)91201-3
Anderso MJ (2001) A new method for non-parametric multivariate analysis of variance. Austral Ecol 26:32–46. https://doi.org/10.1111/j.1442-9993.2001.01070.pp.x
Oksanen J, Blanchet FG, Friendly M, Kindt R, Legendre P, Mcglinn D, Minchin PR, O’hara RB, Simpson GL, Solymos P, Henry M, Stevens H, Szoecs E, Maintainer HW (2019) Package “vegan” Title Community Ecology Package. Community Ecol Packag 2:1–297
Wickham, H. (2016) ggplot2: Create elegant data visualisations using the grammar of graphics. 2nd. Ed. Springer, ISSN 2197–5744. https://doi.org/10.1007/978-3-319-24277-4
Ramette A (2007) Multivariate analyses in microbial ecology. FEMS Microbiol Ecol 62:142–160. https://doi.org/10.1111/j.1574-6941.2007.00375.x
Robinson MD, McCarthy DJ, Smyth GK (2010) edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26:139–140. https://doi.org/10.1093/bioinformatics/btp616
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Ser B Methodol 57:289–300. https://doi.org/10.1111/j.2517-6161.1995.tb02031.x
Pollard KS, Dudoit S, van der Laan MJ (2005). Multiple testing procedures: R multtest package and applications to genomics. In Gentleman R, Carey V, Huber W, Irizarry R, Dudoit S (eds) Bioinformatics and Computational Biology Solutions Using R and Bioconductor. Springer.
R Core Team (2020) R: a language and environment for statistical computing. https://www.r-project.org/. Accessed Jan 2021
Rilling JI, Acuña JJ, Nannipieri P, Cassan F et al (2019) Current opinion and perspectives on the methods for tracking and monitoring plant growth-promoting bacteria. Soil Biol Biochem 130:205–219. https://doi.org/10.1016/j.soilbio.2018.12.012
Finkel OM, Castrillo G, Herrera Paredes S, Salas González I, Dangl JL (2017) Understanding and exploiting plant beneficial microbes. Curr Opin Plant Biol 38:155–163. https://doi.org/10.1016/j.pbi.2017.04.018
Kaminsky LM, Trexler RV, Malik RJ, Hockett KL, Bell TH (2019) The inherent conflicts in developing soil microbial inoculants. Trends Biotechnol 37:140–151. https://doi.org/10.1016/j.tibtech.2018.11.011
Romano I, Ventorino V, Pepe O (2020) Effectiveness of plant beneficial microbes: overview of the methodological approaches for the assessment of root colonization and persistence. Front Plant Sci 11:6. https://doi.org/10.3389/fpls.2020.00006
van Elsas JD, Chiurazzi M, Mallon CA, Elhottovã D, Krištůfek V, Salles JF (2012) Microbial diversity determines the invasion of soil by a bacterial pathogen. Proc Natl Acad Sci USA 109:1159–1164. https://doi.org/10.1073/pnas.1109326109
Liu J, Qi W, Li Q, Wang SG, Song C, Yuan XZ (2020) Exogenous phosphorus-solubilizing bacteria changed the rhizosphere microbial community indirectly. 3 Biotech 10:164. https://doi.org/10.1007/s13205-020-2099-4
Marks BB, Megías M, Ollero FJ et al (2015) Maize growth promotion by inoculation with Azospirillum brasilense and metabolites of Rhizobium tropici enriched on lipo-chitooligosaccharides (LCOs). AMB Expr 5:71. https://doi.org/10.1186/s13568-015-0154-z
Fukami J, Ollero FJ, Megías M, Hungria M (2017) Phytohormones and induction of plant-stress tolerance and defense genes by seed and foliar inoculation with Azospirillum brasilense cells and metabolites promote maize growth. AMB Expr 7:153. https://doi.org/10.1186/s13568-017-0453-7
Antunes JEL, Freitas ADS, Oliveira LMS et al (2019) Sugarcane inoculated with endophytic diazotrophic bacteria: effects on yield, biological nitrogen fixation and industrial characteristics. An Acad Bras Ciênc 91:e20180990. https://doi.org/10.1590/0001-3765201920180990
Bindraban PS, Dimkpa CO, Pandey R (2020) Exploring phosphorus fertilizers and fertilization strategies for improved human and environmental health. Biol Fertil Soils 56:299–317. https://doi.org/10.1007/s00374-019-01430-2
Gyaneshwar P, Kumar GN, Parekh LJ, Poole PS (2002) Role of soil microorganisms in improving P nutrition of plants. Plant Soil 245:133–143. https://doi.org/10.1023/A:1020663916259
Wu X, Ren L, Luo L, Zhang J, Zhang L, Huang H (2020) Bacterial and fungal community dynamics and shaping factors during agricultural waste composting with zeolite and biochar addition. Sustainability 12:7082. https://doi.org/10.3390/su12177082
Chen YP, Rekha PD, Arun AB, Shen FT, Lai WA, Young CC (2006) Phosphate solubilizing bacteria from subtropical soil and their tricalcium phosphate solubilizing abilities. Appl Soil Ecol 34:33–41. https://doi.org/10.1016/j.apsoil.2005.12.002
Vyas P, Gulati A (2009) Organic acid production in vitro and plant growth promotion in maize under controlled environment by phosphate-solubilizing fluorescent Pseudomonas. BMC Microbiol 9:174. https://doi.org/10.1186/1471-2180-9-174
Santos LF, Lana RP, Da Silva MCS, Veloso TGR, Kasuya MCM, Riberio KG (2020) Effective microorganisms inoculant: diversity and effect on the germination of palisade grass seeds. An Acad Bras Ciênc 92:e20180426. https://doi.org/10.1590/0001-3765202020180426
Trabelsi D, Mhamdi R (2013) Microbial inoculants and their impact on soil microbial communities: a review. Biomed Res Int 2013:863240. https://doi.org/10.1155/2013/863240
Li JT, Lu JL, Wang HY et al (2021) A comprehensive synthesis unveils the mysteries of phosphate-solubilizing microbes. Biol. https://doi.org/10.1111/brv.12779
Griffiths RI, Whiteley AS, O’Donnell AG, Bailey MJ (2003) Influence of depth and sampling time on bacterial community structure in an upland grassland soil. FEMS Microbiol Ecol 43:35–43. https://doi.org/10.1111/j.1574-6941.2003.tb01043.x
Allison SD, Martiny JBH (2008) Resistance, resilience, and redundancy in microbial communities. PNAS 105:11512–11519. https://doi.org/10.1073/pnas.0801925105
Griffiths BS, Philippot L (2013) Insights into the resistance and resilience of the soil microbial community. FEMS Microbiol Rev 37:112–129. https://doi.org/10.1111/j.1574-6976.2012.00343.x
Lourenço KS, Suleiman AKA, Pijl A et al (2018) Resilience of the resident soil microbiome to organic and inorganic amendment disturbances and to temporary bacterial invasion. Microbiome 6:142. https://doi.org/10.1186/s40168-018-0525-1
Yeoh YK, Paungfoo-Lonhienne C, Dennis PG et al (2015) The core root microbiome of sugarcanes cultivated under varying nitrogen fertilizer application. Environ Microbiol 18:1338–1351. https://doi.org/10.1111/1462-2920.12925
Niu H, Pang Z, Fallah N et al (2021) Diversity of microbial communities and soil nutrients in sugarcane rhizosphere soil under water soluble fertilizer. PLoS ONE 16:e0245626. https://doi.org/10.1371/journal.pone.0245626
Liang B, Wang LY, Mbadinga SM, Liu JF, Yang SZ, Gu JD, Mu BZ (2015) Anaerolineaceae and Methanosaeta turned to be the dominant microorganisms in alkanes-dependent methanogenic culture after long-term of incubation. AMB Express 5:37. https://doi.org/10.1186/s13568-015-0117-4
Aanderud ZT, Saurey S, Ball BA et al (2018) Stoichiometric shifts in soil C:N: P promote bacterial taxa dominance, maintain biodiversity, and deconstruct community assemblages. Front Microbiol 9:1401. https://doi.org/10.3389/fmicb.2018.01401
Cheng J, Chen Y, He T, Liao R, Liu R, Yi M, Huang L, Yang Z, Fu T, Li X (2017) Soil nitrogen leaching decreases as biogas slurry DOC/N ratio increases. Appl Soil Ecol 111:105–113. https://doi.org/10.1016/j.apsoil.2016.12.001
Mallon CA, Le Roux X, van Doorn GS, Dini-Andreote F, Poly F, Salles JF (2018) The impact of failure: unsuccessful bacterial invasions steer the soil microbial community away from the invader’s niche. ISME J 12:728–741. https://doi.org/10.1038/s41396-017-0003-y
Bernal, MP, Sommer, SG, Chadwick, D, Qing, C, Guoxue, L, Michel Jr, FC (2017) Current approaches and future trends in compost quality criteria for agronomic, environmental, and human health benefits. In Sparks DL (eds) Advances in agronomy. Academic Press, pp 43–233
Obermeier MM, Minarsch EML, Durai Raj AC, Rineau F, Schröder P (2020) Changes of soil-rhizosphere microbiota after organic amendment application in a Hordeum vulgare L. short-term greenhouse experiment. Plant Soil 455:489–506. https://doi.org/10.1007/s11104-020-04637-7
Büttner D, Bonas U (2010) Regulation and secretion of Xanthomonas virulence factors. FEMS Microbiol Rev 34:107–133. https://doi.org/10.1111/j.1574-6976.2009.00192.x
Silva LCC, Targino BN, Furtado MM, de Oliveira Pinto MA, Rodarte MP, Hungaro HM (2017) Xanthan: biotechnological production and applications. In Da Cruz JC, Sérvulo EFC, De Castro AM (eds) Microbial Production of Food Ingredients and Additives. Academic Press, pp 385–422
Diyarova DK (2016) The role of wood-decaying fungi in the carbon cycle of forest ecosystems and the main ecological factors. Eur Sci J 12:162–166. https://doi.org/10.19044/esj.2016.v12n10p%25p
Peralta RM, da Silva BP, Côrrea RCG, Kato CG, Seixas FAV, Bracht A (2017) Enzymes from basidiomycetes—peculiar and efficient tools for biotechnology. In Fogarty WM, Kelly CT (eds) Biotechnology of microbial enzymes. Academic Press, pp 119–149
Yang W, Jing X, Guan Y, Zhai C, Wang T, Shi D, Sun W, Gu S (2019) Response of fungal communities and co-occurrence network patterns to compost amendment in black soil of Northeast China. Front microbiol 10:1562. https://doi.org/10.3389/fmicb.2019.01562
Sun RB, Dsouza M, Gilbert JA, Guo XS, Wang DZ, Guo ZB, Ni Y, Chu H (2016) Fungal community composition in soils subjected to long-term chemical fertilization is most influenced by the type of organic matter. Environ Microbiol 18:5137–5150. https://doi.org/10.1111/1462-2920.13512
Amprayn KO, Rose MT, Kecskés M, Pereg L, Nguyen HT, Kennedy IR (2012) Plant growth promoting characteristics of soil yeast (Candida tropicalis HY) and its effectiveness for promoting rice growth. Appl Soil Ecol 61:295–299. https://doi.org/10.1016/j.apsoil.2011.11.009
Streletskii RA, Kachalkin AV, Glushakova AM, Demin VV, Chernov IY (2016) Quantitative determination of indole-3-acetic acid in yeasts using high performance liquid chromatography–tandem mass spectrometry. Microbiology 85:727–736. https://doi.org/10.1134/S0026261716060187
Fu SF, Sun PF, Lu HY, Wei JY, Xiao HS, Fang WT, Cheng BY, Chou JY (2016) Plant growth-promoting traits of yeasts isolated from the phyllosphere and rhizosphere of Drosera spatulata Lab. Fungal Biol 120:433–448. https://doi.org/10.1016/j.funbio.2015.12.006
Yurkov AM (2018) Yeasts of the soil–obscure but precious. Yeast 35:369–378. https://doi.org/10.1002/yea.3310
Andrews JH, Harris RF (1986) r- and K-selection and microbial ecology. In: Marshall KC (ed) Advances in Microbial Ecology. Springer, Boston, pp 99–147
Fierer N, Bradford MA, Jackson RB (2007) Toward an ecological classification of soil bacteria. Ecology 88:1354–1364. https://doi.org/10.1890/05-1839
Dickie IA (2007) Host preference, niches and fungal diversity. New Phytol 174:230–233. https://doi.org/10.1111/j.1469-8137.2007.02055.x
Ceci A, Pinzari F, Russo F, Maggi O, Persiani AM (2018) Saprotrophic soil fungi to improve phosphorus solubilisation and release: In vitro abilities of several species. Ambio 47:30–40. https://doi.org/10.1007/s13280-017-0972-0
Wang Z, Gao W, Liu X, Chen P, Lu W, Wang F, Li H, Sun Q, Zhang H (2019) Efficient production of polysaccharide by Chaetomium globosum CGMCC 6882 through co-culture with host plant Gynostemma pentaphyllum. Bioprocess Biosyst Eng 42:1731–1738. https://doi.org/10.1007/s00449-019-02169-8
Dogaris I, Vakontios G, Kalogeris E, Mamma D, Kekos D (2009) Induction of cellulases and hemicellulases from Neurospora crassa under solid-state cultivation for bioconversion of sorghum bagasse into ethanol. Ind Crops Prod 29:404–411. https://doi.org/10.1016/j.indcrop.2008.07.008
Pamidipati S, Ahmed A (2017) Degradation of lignin in agricultural residues by locally isolated fungus Neurospora discreta. Appl Biochem Biotechnol 181:1561–1572. https://doi.org/10.1007/s12010-016-2302-6
Koyama A, Wallenstein MD, Simpson RT, Moore JC (2013) Carbon-degrading enzyme activities stimulated by increased nutrient availability in arctic tundra soils. PLoS ONE 8:e77212. https://doi.org/10.1371/journal.pone.0077212
Nannipieri P, Giagnoni L, Landi L, Renella G (2011) Role of phosphatase enzymes in soil. In: Bunemman EK, Oberson A, Frossard E (eds) Phosphorus in action. Springer, Berlin, pp 215–243
Mickan BS, Abbott LK, Fan J, Hart MM, Siddique KH, Solaiman ZM, Jenkins SN (2018) Application of compost and clay under water-stressed conditions influences functional diversity of rhizosphere bacteria. Biol Fertil Soils 54:55–70. https://doi.org/10.1007/s00374-017-1238-5
Acknowledgements
We are grateful to the Sugar Cane Plant “Usina Estiva” for providing field and operational support, as well as the Company Baraúna Soluções Biológicas for partial funding of this project. We thank Denise L Mescolotti, Fernando Baldesin, Maiele C Santana MSc., Bruna AB Lopes MSc., and Arthur PA Pereira Ph.D. for technical assistance. EJBNC acknowledges a research productivity fellowship from CNPq (no. 305193/2016-3). FPM acknowledges FAPESP for its postdoctoral scholarship (no. 2019/27682-0). SRC and HPF acknowledge CAPES for their postdoctoral scholarship and master’s grant, respectively.
Funding
This work was supported by “Financiadora de Estudos e Projetos” (FINEP) (no. 01.13.0209.00) and “Fundação de Amparo à Pesquisa do Estado de São Paulo” (FAPESP) (no. 2016/18944–3).
Author information
Authors and Affiliations
Contributions
Conceptualization: GAEB, CML and EJBNC; data curation: AMMS, GAEB and CML; formal analysis: AMMS, GAEB, FPM and HPF; investigation: AMMS, YFR and SRC; supervision: EJBNC and SRC; writing-original draft: AMMS, GAEB, SRC and FPM; writing-review and editing: AMMS, GAEB, SRC, FPM, HPF and EJBNC.
Corresponding author
Ethics declarations
Ethics Approval
Not applicable.
Consent to Participate
The authors declare their consent to participate in the present work.
Consent for Publication
The authors declare their consent for the publication of the present work.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Silva, A.M.M., Estrada-Bonilla, G.A., Lopes, C.M. et al. Does Organomineral Fertilizer Combined with Phosphate-Solubilizing Bacteria in Sugarcane Modulate Soil Microbial Community and Functions?. Microb Ecol 84, 539–555 (2022). https://doi.org/10.1007/s00248-021-01855-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-021-01855-z