Abstract
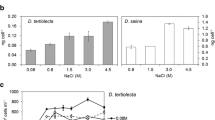
Diverse algae possess the ability to recover from extreme desiccation without forming specialized resting structures. Green algal genera such as Tetradesmus (Sphaeropleales, Chlorophyceae) contain temperate terrestrial, desert, and aquatic species, providing an opportunity to compare physiological traits associated with the transition to land in closely related taxa. We subjected six species from distinct habitats to three dehydration treatments varying in relative humidity (RH 5%, 65%, 80%) followed by short- and long-term rehydration. We tested the capacity of the algae to recover from dehydration using the effective quantum yield of photosystem II as a proxy for physiological activity. The degree of recovery was dependent both on the habitat of origin and the dehydration scenario, with terrestrial, but not aquatic, species recovering from dehydration. Distinct strains of each species responded similarly to dehydration and rehydration, with the exception of one aquatic strain that recovered from the mildest dehydration treatment. Cell ultrastructure was uniformly maintained in both aquatic and desert species during dehydration and rehydration, but staining with an amphiphilic styryl dye indicated damage to the plasma membrane from osmotically induced water loss in the aquatic species. These analyses demonstrate that terrestrial Tetradesmus possess a vegetative desiccation tolerance phenotype, making these species ideal for comparative omics studies.






Similar content being viewed by others
Data Availability
New DNA sequence data are available in NCBI GenBank. Raw physiological data and scripts for data analysis were deposited to DRYAD (doi:10.5061/dryad.sqv9s4n1t).
References
Oliver MJ, Zoltan T, Mishler BD (2000) The evolution of vegetative desiccation tolerance in land plants. Plant Ecol 151:85–100
Bewley JD (1979) Physiological aspects of desiccation tolerance. Annu Rev Plant Physiol 30:195–238
Oliver MJ, Farrant JM, Hilhorst HWM, Mundree Williams S, Bewley JB (2020) Desiccation tolerance: avoiding cellular damage during drying and rehydration. Annu Rev Plant Biol 71:435–460
Alpert P, Oliver MJ (2002) Drying without dying. In: Black M, Pritchard HW (eds) Desiccation and survival in plants: drying without dying. CABI Publishing, pp 3–44
Ligrone R, Duckett JG, Renzaglia KS (2012) Major transitions in the evolution of early land plants: a bryological perspective. Ann Bot 109(5):851–871
Holzinger A, Lütz K, Karsten U (2011) Desiccation stress causes structural and ultrastructural alterations in the aeroterrestrial green alga Klebsormidium crenulatum (Klebsormidiophyceae, Streptophyta) isolated from an alpine soil crust. J Phycol 47(3):591–602
Yobi A, Wone BWM, Xu W, Alexander DC, Guo L, Ryals JA, Oliver MJ, Cushman JC (2012) Comparative metabolic profiling between desiccation-sensitive and desiccation-tolerant species of Selaginella reveals insights into the resurrection trait. Plant J 72(6):983–999
Gao B, Li X, Zhang D, Liang Y, Yang H, Chen M, Zhang Y, Zhang J, Wood A (2017) Desiccation tolerance in bryophytes: the dehydration and rehydration transcriptomes in the desiccation-tolerant bryophyte Bryum argenteum. Sci Rep 7:7571
Peredo EL, Cardon ZG (2020) Shared up-regulation and contrasting down-regulation of gene expression distinguish desiccation-tolerant from intolerant green algae. Proceedings of the National Academy of Sciences of the United States of America 117(29):17438–17445
Aigner S, Glaser K, Arc E, Holzinger A, Schletter M, Karsten U, Kranner I (2020) Adaptation to aquatic and terrestrial environments in Chlorella vulgaris (Chlorophyta). Front Microbiol 11:585836
Farrant JM, Lehner A, Cooper K, Wiswedel S (2009) Desiccation tolerance in the vegetative tissues of the fern Mohria caffrorum is seasonally regulated. Plant J 57(1):65–79
Leliaert F, Smith DR, Moreau H, Herron MD, Verbruggen H, Delwiche CF, De Clerck O (2012) Phylogeny and molecular evolution of the green algae. Crit Rev Plant Sci 31(1):1–46
Lewis LA, Lewis PO (2005) Unearthing the molecular phylodiversity of desert soil green algae (Chlorophyta). Syst Biol 54(6):936–947
Cardon ZG, Gray DW, Lewis LA (2008) The green algal underground: evolutionary secrets of desert cells. BioScience 58(2):114–122
Rindi F, Allali HA, Lam DW, Lopez-Bautista JM (2009) An overview of the biodiversity and biogeography of terrestrial green algae. In: Rescigno V, Maletta S (eds) Biodiversity hotspots. Nova Science, New York, pp 105–122
Evans RD, Johansen JR (1999) Microbiotic crusts and ecosystem processes. Crit Rev Plant Sci 18:183–225
Belnap J, Lange OL (2001) Biological soil crusts: structure, function, and management. Springer, Berlin
Song G, Li X, Hui R (2017) Effect of biological soil crusts on seed germination and growth of an exotic and two native plant species in an arid ecosystem. PLoS One 12(10):e018583
Proctor MC, Smirnoff N (2000) Rapid recovery of photosystems on rewetting desiccation-tolerant mosses: chlorophyll fluorescence and inhibitor experiments. J Exp Bot 51(351):1695–1704
Gray DW, Lewis LA, Cardon ZG (2007) Photosynthetic recovery following desiccation of desert green algae (Chlorophyta) and their aquatic relatives. Plant Cell Environ 30(10):1240–1255
Büdel B, Darienko T, Deutschewitz K, Dojani S, Friedl T, Mohr KI, Salisch M, Reisser W, Weber B (2009) Southern African biological soil crusts are ubiquitous and highly diverse in drylands, being restricted by rainfall frequency. Soil Microbiol 57:229–247
Fučíková K, Lewis PO, Lewis LA (2014) Widespread desert affiliation of Trebouxiophycean algae (Trebouxiophyceae, Chlorophyta) including discovery of three new desert genera. Phycol Res 62(4):294–305
Mikhailyuk T, Glaser K, Tsarenko P, Demchenko U, Karsten E (2019) Composition of biological soil crusts from sand dunes of the Baltic sea coast, in the context of an integrative approach to the taxonomy of microalgae and cyanobacteria. Eur J Phycol 54(3):236–255
Terlova EF, Lewis LA (2019) A new species of Tetradesmus (Chlorophyceae, Chlorophyta) isolated from desert soil crust habitats in southwestern North America. Plant Fungal Syst 64(1):25–32
Cardon ZG, Peredo EL, Dohnalkova AC, Gershone HL, Bezanilla M (2018) A model suite of green algae within the Scenedesmaceae for investigating contrasting desiccation tolerance and morphology. J Cell Sci 131:jcs212233
Bartoškova H, Komenda J, Nauš J (1999) Functional changes of photosystem II in the moss Rhizomnium punctatum (Hedw.) induced by different rates of dark desiccation. J Plant Physiol 154:597–604
Donner A, Glaser K, Borchhardt N, Karsten U (2017) Ecophysiological response of dehydration and temperature in terrestrial Klebsormidium (Streptophyta) isolated from biological soil crusts in central European grasslands and forests. Microb Ecol 73(4):850–864
Cheng X, Lang I, Adeniji OS, Griffing L (2017) Plasmolysis–deplasmolysis causes changes in endoplasmic reticulum form, movement, flow, and cytoskeletal association. J Exp Biol 68(15):4075–4087
Wu N, Zhang YM, Downing A, Zhang J, Yang CH (2012) Membrane stability of the desert moss Syntrichia caninervis Mitt. during desiccation and rehydration. J Bryol 34(1):1–8
Li J, Li X, Zhang P (2014) Micro-morphology, ultrastructure and chemical composition changes of Bryum argenteum from a desert biological soil crust following one-year desiccation. Bryologist 117(30):232–240
Holzinger A, Herburger K, Kaplan F, Lewis LA (2015) Desiccation tolerance in the chlorophyte green alga Ulva compressa: does cell wall architecture contribute to ecological success? Planta 242(2):477–492
Hall J, Fučíková K, Lo C, Lewis L, Karol K (2010) An assessment of proposed DNA barcodes in freshwater green algae. Cryptogam Algol 31(4):529–555
Famá P, Wysor B, Kooistra W, Zuccarello GC (2012) Molecular phylogeny of the genus Caulerpa (Caulerpales, Chlorophyta) inferred from chloroplast tufA gene. J Phycol 38(5):1040–1050
McManus HA, Lewis LA (2011) Molecular phylogenetic relationships in the freshwater family Hydrodictyaceae (Sphaeropleales, Chlorophyceae), with an emphasis on Pediastrum duplex. J Phycol 47(1):152–163
White TJ, Bruns TD, Lee SB, Taylor JW (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ (eds) PCR Protocols: a guide to methods and applications. Academic Press, New York, pp 315–322
Schultz J, Müller T, Achtziger M, Seibel PN, Dandekar T, Wolf M (2006) The internal transcribed Spacer 2 Database — a web server for (not only) low level phylogenetic analyses. Nucleic Acids Res 34:704–707
Koetschan C, Hackl T, Müller T, Wolf M, Förster F, Schultz J (2012) ITS2 Database IV interactive taxon sampling for internal transcribed spacer 2 based phylogenies. Mol Phylogenet Evol 63(3):585–588
Lanfear R, Frandsen PB, Wright AM, Senfeld T, Calcott B (2017) PartitionFinder 2: new methods for selecting partitioned models of evolution for molecular and morphological phylogenetic analyses. Mol Biol Evol 34(3):772–773
Lanfear R, Calcott B, Ho SY, Guindon S (2012) PartitionFinder: combined selection of partitioning schemes and substitution models for phylogenetic analyses. Mol Biol Evol 29(6):1695–1701
Guindon S, Dufayard JF, Lefort V, Anisimova M, Hordijk W, Gascuel O (2010) New algorithms and methods to estimate maximum-likelihood phylogenies: assessing the performance of Phyml 3.0. Syst Biol 59(3):307–321
Akaike H (1998) Information theory and an extension of the maximum likelihood principle. In: Parzen E, Tanbe K, Kitagawa G (eds) Selected papers of Hirotugu Akaike. Springer New York, New York, pp 199–213
Ronquist F, Huelsenbeck JP (2003) MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 19(12):1572–1574
Miller MA, Pfeiffer W, Schwartz T (2010) Creating the CIPRES Science Gateway for inference of large phylogenetic trees, Gateway Computing Environments Workshop (GCE). New Orleans, LA 2010:1–8
Rambout A, Drummond AJ, Xie D, Baele G, Suchard MA (2018) Posterior summarization in Bayesian phylogenetics using Tracer 1.7. Syst Biol 67(5):901–904
Swofford D L (2003) PAUP*. Phylogenetic Analysis Using Parsimony (* and other methods). Version 4. Sinauer Associates, Sunderland.
Karsten U, Herburger K, Holzinger A (2014) Dehydration, temperature, and light tolerance in members of the aeroterrestrial green algal genus Interfilum (Streptophyta) from biogeographically different temperate soils. J Phycol 50(5):804–816
Fletchner VR, Johansen JR, Clark WH (1998) Algal composition of microbiotic crusts from the central desert in Baja California, Mexico. Great Basin Nat 58:295–311
Lewis LA, Flechtner VR (2005) Tetradesmus bajacalifornicus L. A. Lewis & Flechtner, sp. nov. and Tetradesmus deserticola L. A. Lewis & Flechtner, sp. nov. (Scenedesmaceae, Chlorophyta). Syst Biol 54:936–947
Banchi E, Candotto Carniel F, Montagner A, Petruzzellis F, Pichler G, Giarola V, Bartels D, Pallavicini A, Tretiach M (2018) Relation between water status and desiccation-affected genes in the lichen photobiont Trebouxia gelatinosa. Plant Physiol Biochem 120:189–197
Proctor MC, Oliver MJ, Wood AJ, Alpert P, Stark LR, Cleavitt NL, Mishler BD (2007) Desiccation-tolerance in bryophytes: a review. Bryologist 110(4):595–621
Koster KL, Balsamo RA, Espinoza C, Oliver MJ (2010) Desiccation sensitivity and tolerance in the moss Physcomitrella patens: assessing limits and damage. Plant Growth Regul 62:293–302
Karsten U, Herburger K, Holzinger A (2016) Living in biological soil crusts communities of African deserts – physiological traits of green algal Klebsormidium species (Streptophyta) to cope with desiccation, light and temperature gradients. J Plant Physiol 194:2–12
Pierangelini M, Rysanek D, Lang I, Adlassnig W, Holzinger A (2017) Terrestrial adaptation of green algae Klebsormidium and Zygnema (Charophyta) involves diversity in photosynthetic traits but not in CO2 acquisition. Planta 246(5):971–986
Pierangelini M, Glaser K, Mikhailyuk T, Karsten U, Holzinger A (2019) Light, dehydration but not temperature drive photosynthetic adaptations of basal streptophytes (Hormidiella, Streptosarcina and Streptofilum) living in terrestrial habitats. Microb Ecol 77:380–393
Farrant JM (2000) Comparison of mechanisms of desiccation tolerance among three angiosperm resurrection plants. Plant Ecol 151:29–39
Alpert P (2006) Constrains of tolerance: why are desiccation tolerant organisms so small or rare? J Exp Biol 209(9):1575–1584
Oliver MJ, Guo L, Alexander D, Ryals JA, Wone BWM (2011) A sister group contrast using untargeted global metabolomic analysis delineates the biochemical regulation underlying desiccation tolerance in Sporobolus stapfianus. Plant Cell 23:1231–1248
Rippin M, Becker B, Holzinger A (2017) Enhanced desiccation tolerance in mature cultures of the streptophytic green alga Zygnema circumcarinatum revealed by transcriptomics. Plant Cell Physiol 58(12):2067–2084
Domozych DS, Roberts R, Danyow C, Flitter B, Smith R, Providence K (2003) Plasmolysis, Hechtain strand formation, and localized membrane-wall adhesions in the desmid, Closterium acerosum (Chlorophyta). J Phycol 39(6):1194–1206
Becker B, Feng X, Yin Y, Holzinger A (2020) Desiccation tolerance in streptophyte algae and the algae to land plant transition: evolution of LEA and MIP protein families within the Viridiplantae. J Exp Bot 71(11):3270–3278
Ismagulova T, Chekanov K, Gorelova O, Baulina O, Semenova L, Selyakh I, Chivkunova O, Lobakova E, Karpova O, Solovchenko A (2018) A new subarctic strain of Tetradesmus obliquus – Part I: Identification and fatty acid profiling. J Appl Phycol 30:2737–2750
Starr RC (1955) Zygospore germination in Cosmarium botrytis var. subtumidum. Am J Bot 42(7):577–581
Evans JH (1958) Algae during dry periods: Part I. An investigation of the algae of five small ponds. J Ecol 46(1):149–167
Rengefors K, Karlsson I, Hansson L-A (1998) Algal cyst dormancy: a temporal escape from herbivory. Proc R Soc Lond 265(1403):1353–1358
Illing N, Denby KJ, Collett H, Shen A, Farrant JM (2005) The signature of seeds in resurrection plants: a molecular and physiological comparison of desiccation tolerance in seeds and vegetative tissues. Integr Comp Biol 45(5):771–787
Costa M-C, Artur MAS, Maia J, Jonkheer E, Derks MFL, Nijveen H, Williams B, Mundree SG, Jiménez-Gómez JM, Hesselink T, Schijlen EGWM, Ligterink W, Oliver MJ, Farrant JM, Hilhorst HWM (2017) A footprint of desiccation tolerance in the genome of Xerophyta viscosa. Nat Plants 3:17038
VanBuren R, Wai CM, Zhang Q, Song X, Edger PP, Bryant D, Michael TP, Mockler TC, Bartels D (2017) Seed desiccation mechanisms co-opted for vegetative desiccation tolerance in the resurrection grass Oropetium thomaeum. Plant Cell Environ 40(10):2292–2306
Acknowledgments
The authors thank Dr. X. Sun and Dr. M. Abril from the Bioscience Electron Microscopy Laboratory at University of Connecticut for their help in sample preparation and assistance with TEM, and C. O’Connell from the Advanced Light Microscopy Facility at UConn for assistance with fluorescence microscopy. We thank Dr. P. Lewis for advice about the ancestral states analyses, and Drs. B. Goffinet, N. Patel, J. Seemann, Y. Yuan, J. Wegrzyn, and two anonymous reviewers for their helpful comments.
Funding
This study was supported by the Austrian Science Fund (FWF) grant I 1951-B16 to A.H. The research stay of A.H. at the University of Connecticut was generously supported by a Fulbright Scholarship. TEM and CLSM imaging of cells was supported by 2017 UConn EEB Research Award (The Betty Foster Feingold Endowment for Ecology and Evolutionary Biology to the Department of Ecology and Evolutionary Biology).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflicts of interest.
Supplementary Information
ESM 1
(PDF 1740 kb)
Rights and permissions
About this article
Cite this article
Terlova, E.F., Holzinger, A. & Lewis, L.A. Terrestrial Green Algae Show Higher Tolerance to Dehydration than Do Their Aquatic Sister-Species. Microb Ecol 82, 770–782 (2021). https://doi.org/10.1007/s00248-020-01679-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-020-01679-3