Abstract
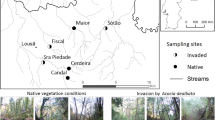
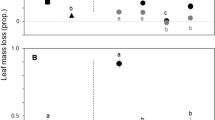
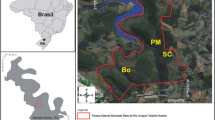
The invasion of native riparian forests by exotic tree species can lead to profound changes in the ecological integrity of freshwater ecosystems. We assessed litter decomposition of native (Alnus glutinosa and Quercus robur) and invasive (Acacia melanoxylon and Acacia dealbata) tree species, and associated microbial activity and community structure, after being immersed for conditioning in 3 reference and 3 “invaded” streams in Serra da Lousã (central Portugal) and used in microcosms simulating stream conditions. Litter decomposition differed among species, in the order: Al. glutinosa > Q. robur > (Ac. melanoxylon ~ Ac. dealbata). Alnus glutinosa litter decomposed faster probably because it was soft and had high nitrogen concentration for decomposers. Quercus robur litter decomposed slower most likely because it was tough and had high polyphenol and low nitrogen concentrations. Acacia melanoxylon litter was the toughest and had a thick cuticle that likely acted as a physical barrier for microbial colonization. In Ac. dealbata, the small-sized leaflets and high lignin concentration may have limited microbial litter decomposition. Litter decomposition was faster in “invaded” streams, probably because they were N-limited and increases in nitrogen concentration in water, promoted by Acacia species invasion, stimulated microbial activity on litter. The aquatic hyphomycete community structure differed among litter species and between stream types, further suggesting that microbes were sensitive to litter characteristics and water nutrient concentrations. Overall, the invasion of native riparian forests by Acacia species may affect microbial decomposer activity, thus altering important stream ecosystem processes, such as litter decomposition and nutrient cycles.





Similar content being viewed by others
References
Millennium Ecosystem Assessment (2005) Ecosystems and human well-being: synthesis. Island Press, Washington DC
Kominoski JS, Shah JJF, Canhoto C, Fischer DG, Giling DP, González E, Griffiths NA, Larrañaga A, LeRoy CJ, Mineau MM, McElarney YR, Shirley SM, Swan CM, Tiegs SD (2013) Forecasting functional implications of global changes in riparian plant communities. Front Ecol Environ 11(8):423–432. https://doi.org/10.1890/120056
Castro-Díez P, Pauchard A, Traveset A, Vilà M (2016) Linking the impacts of plant invasion on community functional structure and ecosystem properties. J Veg Sci 27:1233–1242. https://doi.org/10.1111/jvs.12429
Ferreira V, Koricheva J, Pozo J, Graça MAS (2016) A meta-analysis on the effects of changes in the composition of native forests on litter decomposition in streams. For Ecol Manag 364:27–38. https://doi.org/10.1016/j.foreco.2016.01.002
Wallace JB, Eggert SL, Meyer JL, Webster JR (1997) Multiple trophic levels of a forest stream linked to terrestrial litter inputs. Science 277(5322):102–104. https://doi.org/10.1126/science.277.5322.102
Graça MAS, Canhoto C (2006) Leaf litter processing in low order streams. Limnetica 25(1–2):1–10
Gessner MO, Chauvet E, Dobson M (1999) A perspective on leaf litter breakdown in streams. Oikos 85:377–384. https://doi.org/10.2307/3546505
Baldy V, Gessner MO, Chauvet E (1995) Bacteria, fungi and the breakdown of leaf litter in a large river. Oikos 74:93–102. https://doi.org/10.2307/3545678
Gessner MO, Chauvet E (1994) Importance of stream microfungi in controlling breakdown rates of leaf litter. Ecology 75(6):1807–1817. https://doi.org/10.2307/1939639
Ferreira V, Gulis V, Graça MAS (2006) Whole-stream nitrate addition affects litter decomposition and associated fungi but not invertebrates. Oecologia 149:718–729. https://doi.org/10.1007/s00442-006-0478-0
Schindler MH, Gessner MO (2009) Functional leaf traits and biodiversity effects on litter decomposition in a stream. Ecology 90(6):1641–1649. https://doi.org/10.1890/08-1597.1
Ferreira V, Encalada AC, Graça MAS (2012) Effects of litter diversity on decomposition and biological colonization of submerged litter in temperate and tropical streams. Freshw Sci 31(3):945–962. https://doi.org/10.1899/11-062.1
Gulis V, Suberkropp K (2003) Effect of inorganic nutrients on relative contributions of fungi and bacteria to carbon flow from submerged decomposing leaf litter. Microb Ecol 45:11–19. https://doi.org/10.1007/s00248-002-1032-1
Pereira A, Geraldes P, Lima-Fernandes E, Fernandes I, Cássio F, Pascoal C (2016) Structural and functional measures of leaf-associated invertebrates and fungi as predictors of stream eutrophication. Ecol Indic 69:648–656. https://doi.org/10.1016/j.ecolind.2016.05.017
Ferreira V, Castela J, Rosa P, Tonin AM, Boyero L, Graça MAS (2016) Aquatic hyphomycetes, benthic macroinvertebrates and leaf litter decomposition in streams naturally differing in riparian vegetation. Aquat Ecol 50:711–725. https://doi.org/10.1007/s10452-016-9588-x
Laitung B, Chauvet E (2005) Vegetation diversity increases species richness of leaf-decaying fungal communities in woodland streams. Arch Hydrobiol 164(2):217–235. https://doi.org/10.1127/0003-9136/2005/0164-0217
Lecerf A, Dobson M, Dang CK, Chauvet E (2005) Riparian plant species loss alters trophic dynamics in detritus-based stream ecosystems. Oecologia 146:432–442. https://doi.org/10.1007/s00442-005-0212-3
Ghate SD, Sridhar KR (2017) Endophytic aquatic hyphomycetes in roots of riparian tree species of two Western Ghat streams. Symbiosis 71:233–240. https://doi.org/10.1007/s13199-016-0435-6
Richardson DM, Rejmánek M (2011) Trees and shrubs as invasive alien species – a global review. Divers Distrib 17:788–809. https://doi.org/10.1111/j.1472-4642.2011.00782.x
Souza-Alonso P, Rodríguez J, González L, Lorenzo P (2017) Here to stay. Recent advances and perspectives about Acacia invasion in Mediterranean areas. Ann For Sci 74:55. https://doi.org/10.1007/s13595-017-0651-0
Le Maitre DC, Gaertner M, Marchante E et al (2011) Impacts of invasive Australian acacias: implications for management and restoration. Divers Distrib 17:1015–1029. https://doi.org/10.1111/j.1472-4642.2011.00816.x
ICNF (2019) IFN6 – Principais resultados – relatório sumário. Instituto da Conservação da Natureza e das Florestas, Lisboa
Presidência do Conselho de Ministros (2019) Decreto-Lei n.° 92/2019. Diário da República 130:3428–3442
Sheppard AW, Shaw RH, Sforza R (2006) Top 20 environmental weeds for classical biological control in Europe: a review of opportunities, regulations and other barriers to adoption. Weed Res 46:93–117. https://doi.org/10.1111/j.1365-3180.2006.00497.x
Goldstein CL, Williard KWJ, Schoonover JE (2009) Impact of an invasive exotic species on stream nitrogen levels in southern Illinois. J Am Water Resour Assoc 45:3–672. https://doi.org/10.1111/j.1752-1688.2009.00314.x
Atwood TB, Wiegner TN, Turner JP, MacKenzie RA (2010) Potential effects of an invasive nitrogen-fixing tree on a Hawaiian stream food web. Pac Sci 64(3):367–379. https://doi.org/10.2984/64.3.367
Mineau MM, Baxter CV, Marcarelli AM (2011) A non-native riparian tree (Elaeagnus angustifolia) changes nutrient dynamics in streams. Ecosystems 14:353–365. https://doi.org/10.1007/s10021-011-9415-0
Wiegner TN, Hughes F, Shizuma LM, Bishaw DK, Manuel ME (2013) Impacts of an invasive N2-fixing tree on Hawaiian stream water quality. Biotropica 45(4):409–418. https://doi.org/10.1111/btp.12024
Stewart SD, Young MB, Harding JS, Horton TW (2019) Invasive nitrogen-fixing plant amplifies terrestrial–aquatic nutrient flow and alters ecosystem function. Ecosystems 22:587–601. https://doi.org/10.1007/s10021-018-0289-2
Graça MAS, Poquet JM (2014) Do climate and soil influence phenotypic variability in leaf litter, microbial decomposition and shredder consumption? Oecologia 174:1021–1032. https://doi.org/10.1007/s00442-013-2825-2
Graça MAS, Bärlocher F, Gessner MO (2005) Methods to study litter decomposition. A Practical Guide, Springer, Dordrecht
Goering HK, Van Soest PJ (1970) Forage fiber analyses (apparatus, reagents, procedures, and some applications). Agricultural Research Service, Washington DC
APHA (1995) Standard methods for the examination of water and watershed. American Public Health Association, Washington DC
Gessner MO, Chauvet E (1993) Ergosterol-to-biomass conversion factors for aquatic hyphomycetes. Appl Environ Microbiol 59(2):502–507
Anderson MJ, Gorley RN, Clarke KR (2008) PERMANOVA+ for PRIMER: guide to software and statistical methods. PRIMER-E, Plymouth
Richardson JTE (2011) Eta squared and partial eta squared as measures of effect size in educational research. Educ Res Rev 6:135–147. https://doi.org/10.1016/j.edurev.2010.12.001
Lowe SR, Woodford DJ, Impson DN, Day JA (2008) The impact of invasive fish and invasive riparian plants on the invertebrate fauna of the Rondegat River, Cape Floristic Region, South Africa. Afr J Aquat Sci 33(1):51–62. https://doi.org/10.2989/AJAS.2007.33.1.6.390
Railoun MZ (2018) Impacts of the invasive tree Acacia mearnsii on riparian and instream aquatic environments in the Cape Floristic Region, South Africa. Dissertation, Stellenbosch University
Wiener KD (2018) Quantifying the impacts of invasive N2-fixing plants on relative sediment loads and nutrient concentrations in mountain streams of the Cape Floristic Region, South Africa. Dissertations, Stellenbosch University
Gessner MO, Swan CM, Dang CK, McKie BG, Bardgett RD, Wall DH, Hättenschwiler S (2010) Diversity meets decomposition. Trends Ecol Evol 25(6):372–380. https://doi.org/10.1016/j.tree.2010.01.010
Zhang M, Cheng X, Geng Q, Shi Z, Luo Y, Xu X (2019) Leaf litter traits predominantly control litter decomposition in streams worldwide. Glob Ecol Biogeogr 28:1469–1486. https://doi.org/10.1111/geb.12966
Ferreira V, Raposeiro PM, Pereira A, Cruz AM, Costa AC, Graça MAS, Gonçalves V (2016) Leaf litter decomposition in remote oceanic island streams is driven by microbes and depends on litter quality and environmental conditions. Freshw Biol 61:783–799. https://doi.org/10.1111/fwb.12749
Li AOY, Ng LCY, Dudgeon D (2009) Effects of leaf toughness and nitrogen content on litter breakdown and macroinvertebrates in a tropical stream. Aquat Sci 71:80–93. https://doi.org/10.1007/s00027-008-8117-y
Jabiol J, Lecerf A, Lamothe S, Gessner MO, Chauvet E (2019) Litter quality modulates effects of dissolved nitrogen on leaf decomposition by stream microbial communities. Microb Ecol 77:959–966. https://doi.org/10.1007/s00248-019-01353-3
Suberkropp K, Chauvet E (1995) Regulation of leaf breakdown by fungi in streams: influences of water chemistry. Ecology 76(5):1433–1445. https://doi.org/10.2307/1938146
Arroita M, Aristi I, Flores L, Larrañaga A, Díez J, Mora J, Romaní AM, Elosegi A (2012) The use of wooden sticks to assess stream ecosystem functioning: comparison with leaf breakdown rates. Sci Total Environ 440:115–122. https://doi.org/10.1016/j.scitotenv.2012.07.090
Gulis V, Suberkropp K (2003) Interactions between stream fungi and bacteria associated with decomposing leaf litter at different levels of nutrient availability. Aquat Microb Ecol 30:149–157
Duarte S, Pascoal C, Alves A, Correia A, Cássio F (2010) Assessing the dynamic of microbial communities during leaf decomposition in a low-order stream by microscopic and molecular techniques. Microbiol Res 165:351–362. https://doi.org/10.1016/j.micres.2009.06.002
Gulis V, Ferreira V, Graça MAS (2006) Stimulation of leaf litter decomposition and associated fungi and invertebrates by moderate eutrophication: implications for stream assessment. Freshw Biol 51:1655–1669. https://doi.org/10.1111/j.1365-2427.2006.01615.x
Pereira A, Trabulo J, Fernandes I, Pascoal C, Cássio F, Duarte S (2017) Spring stimulates leaf decomposition in moderately eutrophic streams. Aquat Sci 79:197–207. https://doi.org/10.1007/s00027-016-0490-3
Huryn AD, Butz Huryn VM, Arbuckle CJ, Tsomides L (2002) Catchment land-use, macroinvertebrates and detritus processing in headwater streams: taxonomic richness versus function. Freshw Biol 47:401–415. https://doi.org/10.1046/j.1365-2427.2002.00812.x
Fernandes I, Seena S, Pascoal C, Cássio F (2014) Elevated temperature may intensify the positive effects of nutrients on microbial decomposition in streams. Freshw Biol 59:2390–2399. https://doi.org/10.1111/fwb.12445
Gulis V, Suberkropp K (2003) Leaf litter decomposition and microbial activity in nutrient-enriched and unaltered reaches of a headwater stream. Freshw Biol 48:123–134. https://doi.org/10.1046/j.1365-2427.2003.00985.x
Rosemond AD, Benstead JP, Bumpers PM, Gulis V, Kominoski JS, Manning DWP, Suberkropp K, Wallace JB (2015) Experimental nutrient additions accelerate terrestrial carbon loss from stream ecosystems. Science 347:1142–1145. https://doi.org/10.1126/science.aaa1958
Canhoto C, Graça MAS (1996) Decomposition of Eucalyptus globulus leaves and three native leaf species (Alnus glutinosa, Castanea sativa and Quercus faginea) in a Portuguese low order stream. Hydrobiologia 333:79–85. https://doi.org/10.1007/BF00017570
Gulis V (2001) Are there any substrate preferences in aquatic hyphomycetes? Mycol Res 105:1088–1093. https://doi.org/10.1016/S0953-7562(08)61971-1
Ferreira V, Elosegi A, Gulis V, Pozo J, Graça MAS (2006) Eucalyptus plantations affect fungal communities associated with leaf-litter decomposition in Iberian streams. Arch Hydrobiol 166(4):467–490. https://doi.org/10.1127/0003-9136/2006/0166-0467
Laitung B, Pretty JL, Chauvet E, Dobson M (2002) Response of aquatic hyphomycete communities to enhanced stream retention in areas impacted by commercial forestry. Freshw Biol 47:313–323. https://doi.org/10.1046/j.1365-2427.2002.00801.x
Duarte S, Bärlocher F, Pascoal C, Cássio F (2016) Biogeography of aquatic hyphomycetes: current knowledge and future perspectives. Fungal Ecol 19:169–181. https://doi.org/10.1016/j.funeco.2015.06.002
Dang CK, Chauvet E, Gessner MO (2005) Magnitude and variability of process rates in fungal diversity-litter decomposition relationships. Ecol Lett 8:1129–1137. https://doi.org/10.1111/j.1461-0248.2005.00815.x
Acknowledgments
We thank Olímpia Sobral (CEF, University of Coimbra) for all the help in the field and laboratory, and Teresa Gonçalves and Daniela Antunes (CNC, University of Coimbra) for the use of the lyophilizer. Water nutrient analyses were performed by CACTI - Centro de Apoio Científico-Tecnolóxico á Investigación from the University of Vigo, Spain.
Funding
This study was financed by the Portuguese Foundation for Science and Technology (FCT) through the strategic project UID/MAR/04292/2019 granted to MARE and through the exploratory project IF/00129/2014 granted to Verónica Ferreira. Ana Pereira received a doctoral fellowship from FCT (SFRH/BD/118069/2016), financed by the European Social Fund through the Human Capital Operational Program (POCH) and by National Funds from MCTES.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
ESM 1
(PDF 245 kb)
Rights and permissions
About this article
Cite this article
Pereira, A., Ferreira, V. Invasion of Native Riparian Forests by Acacia Species Affects In-Stream Litter Decomposition and Associated Microbial Decomposers. Microb Ecol 81, 14–25 (2021). https://doi.org/10.1007/s00248-020-01552-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-020-01552-3