Abstract
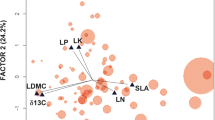
We tested the hypothesis that water stress and soil nutrient availability drive leaf-litter quality for decomposers and detritivores by relating chemical and physical leaf-litter properties and decomposability of Alnus glutinosa and Quercus robur, sampled together with edaphic parameters, across wide European climatic gradients. By regressing principal components analysis of leaf traits [N, P, condensed tannins, lignin, specific leaf area (SLA)] against environmental and soil parameters, we found that: (1) In Q. robur the condensed tannin and lignin contents increased and SLA decreased with precipitation, annual range of temperature, and soil N content, whereas leaf P increased with soil P and temperature; (2) In A. glutinosa leaves N, P, and SLA decreased and condensed tannins increased with temperature, annual range of temperature, and decreasing soil P. On the other hand, leaf P and condensed tannins increased and SLA decreased with minimum annual precipitation and towards sites with low temperature. We selected contrasting leaves in terms of quality to test decomposition and invertebrate consumption. There were intraspecific differences in microbial decomposition rates (field, Q. robur) and consumption by shredders (laboratory, A. glutinosa). We conclude that decomposition rates across ecosystems could be partially governed by climate and soil properties, affecting litter quality and therefore decomposers and detritivores. Under scenarios of global warming and increased nutrients, these results suggest we can expect species-specific changes in leaf-litter properties most likely resulting in slow decomposition with increased variance in temperatures and accelerated decomposition with P increase.





Similar content being viewed by others
References
Abelho M (2001) From litterfall to breakdown in streams: a review. Sci World J 1:656–680
Aerts R, Chapin F (2000) The mineral nutrition of wild plants revisited: reavaluation of processes and patterns. Adv Ecol Res 30:1–67
Allan D, Castillo M (2007) Stream ecology-structure and function of running waters. Springer, Dordrecht
Azevedo-Pereira HVS, Graça MAS, González JM (2006) Life history of Lepidostoma hirtum in an iberian stream and its role in organic matter processing. Hydrobiologia 559:183–192
Bakker MA, Carreño-Rocabado G, Poorter L (2011) Leaf economics traits predict litter decomposition of tropical plants and differ among land use types. Funct Ecol 25:473–483
Bärlocher F (2005) Leaf mass loss estimated by litter bag technique. In: Graça MAS, Bärlocher F, Gessner MO (eds) Methods to study litter decomposition: a practical guide. Springer, Dordrecht, pp 37–42
Boyero L et al (2011) Global distribution of a key trophic guild contrasts with common latitudinal diversity patterns. Ecology 92:1839–1848
Burnham KP, Anderson DR (2002) Model selection and multimodel inference: a practical information-theoretic approach. Springer, New York
Canhoto C, Graça MAS, Bärlocher F (2005) Feeding preferences. In: Graça MAS, Bärlocher F, Gessner MO (eds) Methods to study litter decomposition: a practical guide. Springer, Dordrecht, pp 297–302
Castro-Diez P, Villar-Salvador P, Perez-Rontome C, Maestro-Martinez M, Montserrat-Marti G (1998) Leaf morphology, leaf chemical composition and stem xylem characteristics in two Pistacia (Anacardiaceae) species along a climatic gradient. Flora 193:195–202
Chen Y, Han W, Tang L, Tang Z, Fang J (2011) Leaf nitrogen and phosphorus concentrations of woody plants differ in responses to climate, soil and plant growth form. Ecography 36
Cornwell WK, et al. (2008) Plant species traits are the predominant control on litter decomposition rates within biomes worldwide. Ecol Lett 11:1065–1071
Cunningham SA, Summerhayes B, Westoby M (1999) Evolutionary divergences in leaf structure and chemistry, comparing rainfall and soil nutrient gradients. Ecol Monogr 69:569–588
Doran JW, Jones AJ (1996) Methods for assessing soil quality. SSSA, Madison
Eaton AD, Clesceri LS, Greenberg AE (1995) (eds) Standard methods for the examination of water and wastewater, 19th edn. American Public Health Association, Washington, DC
Ferreira V, Encalada AC, Graça MAS (2012) Effects of litter diversity on decomposition and biological colonization of submerged litter in temperate and tropical streams. Freshwater Sci 31:945–962
Gessner MO, Steiner D (2005) Acid butanol assay for Proanthocyanidins (condensed Tannins). In: Graça MAS, Bärlocher F, Gessner MO (eds) Methods to study litter decomposition: a practical guide. Springer, Dordrecht, pp 107–113
Goering HK, Van Soest PJ (1970) Forage fiber analysis (apparatus, reagents, procedures and some applications). Agricultural handbook. US Department of Agriculture, Washington, DC, pp 1-2
Gonçalves JF, Graca MAS, Callisto M (2006) Leaf-litter break-down in 3 streams in temperate, Mediterranean, and tropical Cerrado climates. J N Am Benthol Soc 25:344–355
Gonçalves JF, Graca MAS, Callisto M (2007) Litter decomposition in a Cerrado savannah stream is retarded by leaf toughness, low dissolved nutrients and a low density of shredders. Freshwater Biol 52:1440–1451
Graça MAS (2001) The role of invertebrates on leaf litter decomposition in streams: a review. Int Rev Hydrobiol 86:383–393
Graça MAS, Cressa C (2010) Leaf quality of some tropical and temperate tree species as food resource for stream shredders. Int Rev Hydrobiol 95:27–41
Graça MAS, Ferreira RCF, Coimbra CN (2001) Litter processing along a stream gradient: the role of invertebrates and decomposers. J N Am Benthol Soc 20:408–420
Gulis V, Ferreira V, Graça MAS (2006) Stimulation of leaf litter decomposition and associated fungi and invertebrates by moderate eutrophication: implications for stream assessment. Freshwater Biol 51:1655–1669
Han WX, Fang J, Reich P, Woodward FI, Wang Z (2011) Biogeography and variability of eleven mineral elements in plant leaves across gradients of climate, soil and plant functional type in China. Ecol Lett 14:788–796
Hijmans RJ, Cameron SE, Parra JL, Jones PG, Jarvis A (2005) Very high resolution interpolated climate surfaces for global land areas. Int J Climatol 25:1965–1978
Intergovernmental Panel on Climate Change (IPCC) (2007) Climate change 2007: synthesis report. Contribution of Working Groups I, II and III to the fourth assessment report of the Intergovernmental Panel on Climate Change. http://www.ipcc.ch/publications
Kaspari M, Garcia M, Harms K, Santana M, Wright S, Yavitt J (2008) Multiple nutrients limit litterfall and decomposition in a tropical forest. Ecol Lett 11:35–43
Krauss GJ, Sole M, Krauss G, Schlosser D, Wesenberg D, Barlocher F (2011) Fungi in freshwaters: ecology, physiology and biochemical potential. FEMS Microbiol Rev 35:620–651
Lecerf A, Chauvet E (2008) Intraspecific variability in leaf traits strongly affects alder leaf decomposition in a stream. Basic Appl Ecol 9:598–605
LeRoy CJ, Whitham TG, Wooley SC, Marks JC (2007) Within-species variation in foliar chemistry influences leaf-litter decomposition in a Utah river. J N Am Benthol Soc 26:426–438
LeRoy CJ, Wooley SC, Lindroth RL (2012) Genotype and soil nutrient environment influence aspen litter chemistry and in-stream decomposition. Freshwater Sci 31:1244–1253
Makkonen M et al (2012) Highly consistent effects of plant litter identity and functional traits on decomposition across a latitudinal gradient. Ecol Lett 15:1033–1041
Ordoñez JC, van Bodegom PM, Witte J-PM, Wright IJ, Reich PB, Aerts R (2009) A global study of relationships between leaf traits, climate and soil measures of nutrient fertility. Global Ecol Biogeogr 18:137–149
Orfanoudakis M, Wheeler CT, Hooker JE (2010) Both the arbuscular mycorrhizal fungus Gigaspora rosea and Frankia increase root system branching and reduce root hair frequency in Alnus glutinosa. Mycorrhiza 20:117–126
Osier TL, Lindroth RL (2001) Effects of genotype, nutrient availability, and defoliation on aspen phytochemistry and insect performance. J Chem Ecol 27:1289–1313
Pallardy SE (2008) Physiology of woody plants. Academic Press, Amsterdam
Pennings S, Carefoot T, Siska E, Chase M, Page T (1998) Feeding preferences of a generalist salt-marsh crab: relative importance of multiple plant traits. Ecology 79:1968–1979
Quinn GP, Keough MJ (2002) Experimental design and data analysis for biologists. Cambridge University Press, Cambridge
R Development Core Team (2008) R a language and environment for statistical computing. The R project for statistical computing. http://www.r-project.org
Reich PB, Oleksyn J (2004) Global patterns of plant leaf N and P in relation to temperature and latitude. Proc Natl Acad Sci USA 101:11001–11006
Rockström J et al (2009) A safe operating space for humanity. Nature 461:472–475
SAS (2007) JMP version 70 http://www.jmp.com
Schweitzer JA et al (2008) From genes to ecosystems: the genetic basis of condensed tannins and their role in nutrient regulation in a Populus model system. Ecosystems 11:1005–1020
StatSoft (2005) STATISTICA version 71 http://www.statsoft.com
Trabucco A, Zomer RJ (2009) Global Aridity Index (Global-Aridity) and Global Potential Evapo-Transpiration (Global-PET) Geospatial database. CGIAR Consortium for Spatial Information. Published online, available from the CGIAR-CSI GeoPortal at: http://www.csi.cgiar.org
Vitousek PM et al (1997) Human alteration of the global nitrogen cycle: sources and consequences. Ecol Appl 7:737–750
Wantzen KM, Yule CM, Mathooko JM, Pringle C (2008) Organic matter processing in tropical streams. In: Dudgeon D (ed) Tropical stream ecology. Elsevier, Amsterdam
Waring BG (2012) A meta-analysis of climatic and chemical controls on leaf litter decay rates in tropical forests. Ecosystems 15:999–1009
Woodward G et al (2012) Continental-scale effects of nutrient pollution on stream ecosystem functioning. Science 336:1438–1440
Wright IJ, Reich PB, Westoby M (2001) Strategy shifts in leaf physiology, structure and nutrient content between species of high- and low-rainfall and high- and low-nutrient habitats. Funct Ecol 15:423–434
Acknowledgments
We thank our colleagues across Europe for kindly sending us litter and soil samples: Adolfo Cordero (Spain), Amílcar Teixeira (Portugal), Andreas Bruder (Switzerland), Anna Romaní (Spain), Antonella Pasquale (Italy), Aranzazu Marcotegui (Spain), Arturo Elosegi (Spain), Bjorn Schaller (Germany), Brendan McKie (Sweden), Carlos Alexandre (Portugal), Cláudia Pascoal (Portugal), Christophe Piscart (France), Claus-G. Schimming (Germany), Daniela Dieter (Germany), Dean Jacobsen (Denmark), Ágostonné Szabó Edit (Hungary), Fernando Cobo (Spain), Franca Sangiorgio (Italy), Ian Dodkins (Portugal), Isabel Muñoz (Spain), Jan Herrmann (Sweden), Jeanette Schlief (Germany), Jessica Hines (Germany), Jesús Casas (Spain), Joerg Schaller (Germany), José Díez (Spain), Juanita Mora (Spain), Julia Toja (Spain), Julien Cornut (France), Kamila Fricová (Czech Republic), Loreto Rossi (Italy), Loraine Maltby (UK), Luz Boyero (Spain), Marcos González (Spain), Maria Leichtfried (Austria), Dinka Mária (Hungary), Marija (Lithuania), Mark Gessner (Germany), Maruxa Alvarez (Spain), Melanie Fletcher (UK), Mike Dobson (UK), Nuno Formigo (Portugal), Nuria Bonada (Spain), Ondřej Simon (Czech Republic), Pedro Anastácio (Portugal), Pierre Marmonier (France), Rosa Fristad (Norway), Rui Cortes (Portugal), Salomé Almeida (Portugal), Samantha Huges (Portugal), Simone Varandas (Portugal), Sujitra Kosol (UK), Tadeuz Fleituch (Poland) and Verónica Ferreira (Portugal). We thank Maria Manuela Costa (Laboratório Química Agrícola e Ambiental) for chemical analysis of the soil samples, and Cristina Grela Docal (IMAR) for leaf N and P analysis and general laboratory assistance for other chemical analyses of leaves. The contribution of Robert Hall and two anonymous reviewers is gratefully acknowledged. This research was funded by the IMAR and the FCT through the project POCI/BIA-BDE/58297/2004.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Robert O. Hall.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Graça, M.A.S., Poquet, J.M. Do climate and soil influence phenotypic variability in leaf litter, microbial decomposition and shredder consumption?. Oecologia 174, 1021–1032 (2014). https://doi.org/10.1007/s00442-013-2825-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-013-2825-2