Abstract
Lake Dziani Dzaha (Mayotte Island, Indian Ocean) is a tropical thalassohaline lake which geochemical and biological conditions make it a unique aquatic ecosystem considered as a modern analogue of Precambrian environments. In the present study, we focused on the diversity of phytoplanktonic communities, which produce very high and stable biomass (mean2014–2015 = 652 ± 179 μg chlorophyll a L−1). As predicted by classical community ecology paradigms, and as observed in similar environments, a single species is expected to dominate the phytoplanktonic communities. To test this hypothesis, we sampled water column in the deepest part of the lake (18 m) during rainy and dry seasons for two consecutive years. Phytoplanktonic communities were characterized using a combination of metagenomic, microscopy-based and flow cytometry approaches, and we used statistical modeling to identify the environmental factors determining the abundance of dominant organisms. As hypothesized, the overall diversity of the phytoplanktonic communities was very low (15 OTUs), but we observed a co-dominance of two, and not only one, OTUs, viz., Arthrospira fusiformis (Cyanobacteria) and Picocystis salinarum (Chlorophyta). We observed a decrease in the abundance of these co-dominant taxa along the depth profile and identified the adverse environmental factors driving this decline. The functional traits measured on isolated strains of these two taxa (i.e., size, pigment composition, and concentration) are then compared and discussed to explain their capacity to cope with the extreme environmental conditions encountered in the aphotic, anoxic, and sulfidic layers of the water column of Lake Dziani Dzaha.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Recently, a new saline-alkaline lake was described on Mayotte Island in the West Indian Ocean, the Lake Dziani Dzaha. The initial data obtained on this crater lake showed characteristics distinct from those reported previously for saline-alkaline lakes [1]. Located at the bottom of a volcanic crater, this small (0.2 km2) lake has no hydrological connection with the surrounding Indian Ocean but nonetheless its water exhibits dissolved element ratios similar to those of seawater. This lake also has some specific physico-chemical characteristics such as a completely anoxic water column under a thin euphotic surface layer (maximum depth of 1.5 m), slightly alkaline (0.23 mol L−1), and has high sulfide concentration (2–6 mM) below a depth of 2.2 m during the rainy season. Based on these characteristics, Lake Dziani Dzaha is considered a modern thalassohaline analogue of Precambrian environment [2]. Moreover, due to the distance of Mayotte island from the African continent (≈ 500 km), this lake can be considered as an isolated ecosystem when compared to continental saline-alkaline lakes and especially those linked by hydrological connections, such as those of the Great Rift Valley [3] or the numerous crater lakes encountered in Ethiopia [4,5,6,7], Kenya [8,9,10,11,12], Uganda [13,14,15], or South Africa [16]. Local external influences also appear limited by the characteristics of the lake’s watershed that is only constituted by the slopes of the small volcanic crater (0.9 km2; Mathelin [17]) without any river or permanent human settlement, but with limited food crop cultivation. Therefore, Lake Dziani Dzaha is not significantly impacted by anthropogenic activities and its hydrological characteristics are mostly dependent on the seasonal climatology, which alternates dry and rainy seasons [1].
The initial biological studies of this lake [1, 18] revealed the following: (i) the absence of macroorganisms similar to those observed in most soda-lakes (birds, fishes, invertebrates, rotifers, and cladoceres), which are known to graze phytoplanktonic communities; (ii) a large amount of phytoplanktonic biomass is produced in a stable manner; and (iii) a very low number of phytoplanktonic taxa. However, these previous characterizations of the phytoplanktonic communities were done using traditional approaches such as microscopy and strain isolation, which are now recognized to have limitations (in terms of sampling depth and culture bias) and to provide an incomplete picture of the entire community [19,20,21]. For instance, several recent studies based on next-generation sequencing (NGS) have shown that the phytoplanktonic communities of saline-alkaline lakes from Kenya and Ethiopia [12, 22, 23] contained a higher richness than previously estimated by microscopic methods, with a number of taxa comparable to that of other aquatic ecosystems.
In this context, we wanted to build on the results from the above-mentioned study of Lake Dziani Dzaha and further characterize the phytoplanktonic community of this lake using a combination of NGS and more classical tools such as microscopy and flow cytometry (FCM).
Doing so, we want to answer the following questions: (i) What is the richness and the composition of the phytoplanktonic community in the Lake Dziani Dzaha? More specifically, we want to determine whether rare taxa exist, as reported in most, if not all, saline alkaline ecosystems, or if they are excluded due to the isolation of the lake and limited environmentally suitable niche for photosynthetic organisms. (ii) Do the drastic abiotic characteristics (high salinity, high pH, limited euphotic depth, anoxia, and sulfidic layer) coupled with high nutrient concentrations and the absence of large grazers lead to the dominance of only one phytoplanktonic species as would be expected according to ecological theories such as the “Killing the winner” hypothesis [24] or the “competitive exclusion” principle [25]?
Material and Methods
Study Site, Sampling, and Environmental Parameters
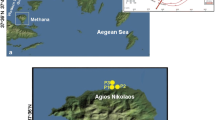
Lake Dziani Dzaha is a volcano crater lake located in Mayotte Island of the Comoros Archipelago, West Indian Ocean (12° 46′ 15.6″ S; 45° 17′ 19.2″ E). The lake surface area is approximately 0.25 km2 with a mean depth of approximately 4 m and a narrow 18-m-deep pit, located in the eastern part of the lake (Fig. 1). This tropical crater lake is characterized by high salinity (up to 70 psu) and alkalinity (0.23 mol L−1), conductivity between 77.1 and 79.7 mS and pH between 9 and 9.5 [1]. The lake is very turbid (Secchi transparency < 0.15 m), and the concentration of chlorophyll a (Chla) is very high (up to 875 μg L−1 in 2010–2011). The lake is also characterized by the absence of zooplankton, fishes, and lesser flamingos [1]. Actively growing stromatolites have been observed in the shallow waters of the lake shores [26].
Four sampling campaigns were conducted in April and October 2014 and in April and November 2015. The precipitation data for the corresponding sampling campaigns were obtained from the Meteo-France Meteorological Observation Station located near Lake Dziani Dzaha (http://www.infoclimat.fr). Discrete water sampling was carried out at the deepest point of the lake (18 m) (Fig. 1) using a horizontal 1.2-L Niskin bottle. Water samples were collected at 16 depths (0, 0.25, 0.5, 0.75, 1, 1.5, 2.5, 3.5, 5, 7, 9, 11, 13, 15, 17, 18 m) and the samples for biological and chemical analyses were processed in the field laboratory within 2 h. The vertical profile of salinity/conductivity, temperature, pH, dissolved O2, and redox potential were performed using YSI 6600 (CTD-O2-pH-redox) (YSI, Yellow Springs, OH, USA) and/or SDOT nke (O2 optode). The attenuation coefficient Kd was determined from discrete PAR measurement profiles using a SPAR nke (PAR). The total alkalinity was determined by Gran titration [27]. The content of soluble sulfide (∑S(-II); H2S), ammonium and ammonia (∑N(-III); NH4), and soluble-reactive phosphorus (SRP; PO43−) was determined colorimetrically in the field lab using the Aqualytic SpectroDirect spectrophotometer and Merck reagent kits. The concentration of Chla was analyzed after extraction using 96% ethanol by ultra-sonication in an ice bath for 30″, and further extraction was allowed overnight at 4 °C in dark. The extract was filtered and the filtrate was analyzed spectrophotometrically at 400–750 nm. The concentration of Chla was calculated according to Ritchie [28].
Cell Abundance
Picophytoplankton
Cell abundances were estimated for all the sampling depths, except for the 18-m depth, which contained a lot of sediment particles that disturb the flow cytometric analysis. Sub-samples (1.6 mL) of the lake water samples from each depth were preserved using 0.2-μm filtered formaldehyde solution (2% final concentration) and stored in liquid nitrogen (− 196 °C). They were further stored at − 80 °C before analysis using a FACSAria Flow cytometer (Becton Dickinson, San Jose, CA, USA) equipped with the HeNe air-cooled laser (633 nm, 20 mW). The cells excited at 633 nm were detected and enumerated according to their forward-angle light scatter (FALS) and right angle light scatter (RALS) properties, and red fluorescence from phycocyanin (660/20 nm) and chlorophyll (675/20 nm) pigments. Fluorescent beads (1–2 μm) were systematically added to each sample. True count beads (Becton Dickinson, San Jose, CA, USA) were added to determine the volume analyzed. List-mode files were analyzed using the BD FACSDiva software. This method discriminates autotrophic picoeukaryote by using their chlorophyll pigments [29]. Pure culture of Picocystis salinarum ALCP 144.1 (Algothèque Laboratoire Cryptogamie Paris, Museum National of Natural History) was used to confirm the flow cytometric signature of this autotrophic group.
Cyanobacteria
Cell abundance was estimated for the same sampling depths than for picophytoplankton. Sub-samples (9 mL) of the lake water samples were collected at each sampling depth for microscopic examination, identification, and measurement of cyanobacterial cells. The samples were fixed with 5% formaldehyde and the taxa were identified as described by Cellamare et al. [18]. The cyanobacterial count data were obtained by the Utermöhl method using an Eclipse TS100 inverted microscope at ×600 magnification (Nikon Instruments Inc., Melville, NY, USA) as described in Catherine et al. [30].
Functional Traits of Isolated Arthrospira fusiformis and Picocystis salinarum Strains
Culture Conditions and Cells Counting
Six clonal non-axenic strains of A. fusiformis PMC 851.14, 894.15 and 917.15 (Paris Museum Collection) and P. salinarum ALCP 144.1, 145.1 and 146.1, isolated from Lake Dziani Dzaha were used. Each strain was grown as batch cultures in 250-mL Erlenmeyer flasks containing 200 mL of Z8-salt medium [31], at 25 °C under 30 μmol photons m−2 s−1 provided by daylight fluorescent tubes (OSRAM Lumilux®, München, Germany), with a 16:8-h light/dark photoperiod. Sampling was performed at the end of exponential growth phase. The cell abundance was estimated by Malassez-based method for each culture as described by Catherine et al. [32]. Three replicates were counted for each culture.
Cell Size Estimation
Length of Arthrospira filaments were measured and counted by the Utermöhl method using an inverted microscope at ×600 magnification. Cell number per filament was then counted using an upright microscope (on at least 50 filaments), at ×1000 magnification. We applied the mean of cell number per filament length to each sample and Arthrospira filaments were then converted to cell abundance (mL−1). This method is described in the SOP Cyanobacterial samples: preservation, enumeration, and biovolume measurements [32].
Cell Pigment Concentrations
Four milliliters of each culture replicate were used for phycocyanin analysis as described by Yéprémian et al. [33]. For lipophilic pigments, 18 mL of each culture was filtered using GF/F Whatman filters, extracted with methanol, and analyzed according to Ras et al. [34]. The results are expressed as the amount of each pigment by cell volume (fg μm−3). The cell volume of the two species was computed from their respective sizes [18] using models for the specific cellular shape of each species [35].
DNA Extraction and Illumina Sequencing
NGS analysis was performed on samples collected at 0.5, 1, 2.5, 5, 11, 15, and 17 m depths. Water subsamples (20 mL) were filtered through 3- and 0.2-μm pore-size polycarbonate filters (Millipore, pressure < 10 kPa), and then stored at − 20 °C until nucleic acid extraction. The DNA was extracted separately from both the filters, using the Power Water DNA isolation kit (MoBio Laboratories) as described by the manufacturer for maximum yield. The DNA quality was analyzed by 1% (w/v) agarose gel electrophoresis and quantified using NanoDrop. The amplification of bacterial V3–V5 region of the 16S rRNA genes was performed in triplicate, using the universal primers 357F [36] and 926R [37] allowing the amplification of bacteria and eukaryotic chloroplast sequences, respectively. Additionally, the DNA was extracted without any biological matrix, and it was considered as a negative control to evaluate ambient contamination. High-throughput sequencing was achieved after pooling the PCR-triplicates and a multiplexing step, by HiSeq Rapid Run 300 bp PE technology using the Illumina HiSeq 2500 system (GATC Biotech, Konstanz, Germany).
Sequence Processing
The 16S rRNA paired-end reads obtained from both the filters were merged with a maximum of 10% mismatches in the overlap region using FLASh [38]. The denoising procedure was carried out by discarding reads containing ambiguous bases (N) or reads outside the range of expected length (e.g., 450–580 bp). After dereplication, the sequences were clustered using SWARM [39] that uses a local clustering threshold. Chimeric sequences were removed using VSEARCH [40]. Furthermore, the sequences representing < 0.005% of the total number of sequences [41] along with singletons were also removed. A preliminary taxonomic affiliation was performed with both RDP Classifier [42] and BLASTN+ [43] against the 128 SILVA database [44]. This procedure was automated in the FROGS pipeline [45]. The contaminant operational taxonomic units (OTUs) identified from the control samples were removed.
Taxonomic and Phylogenetic Affiliation
The affiliation of the phytoplanktonic OTUs was carried out using the National Center for Biotechnology Information Basic Local Alignment Search Tool (NCBI BLAST) tool on three databases: (i) GenBank sequence database (NCBI 1998), (ii) sequences of cyanobacterial PMC strains isolated from Lake Dziani Dzaha [18], and (iii) sequences in the PhytoRef database [46]. A phylogenetic approach was then used to create a 16S rRNA gene phylogeny with selected sequences based on the first affiliation obtained by similarity analysis (NCBI BLAST) and the reference databases (Genbank, PMC strains, and PhytoRef). Phylogenetic reconstruction was performed using the MEGA6 software with 1000 iterations, and according to three methods: neighbor joining (NJ), maximum likelihood (ML), and maximum parsimony (MP). The outgroups were selected based on the results of similar studies: (i) Rhodopseudomonas palustris for cyanobacteria and (ii) Aulacoseira ambigua (Ochrophyta), (iii) Vitrella brassicaformis (Myzozoa), and (iv) Euglena gracilis (Chlorophyta) for photosynthetic eukaryotes [47].
Data Analyses
Partial least square (PLS) regression was used to identify the environmental factors (Xi) affecting the abundance of P. salinarum and A. fusiformis (Yi), This method is particularly suited for analysis of data with several explanatory variables (Xi) and a limited number of observations (Yi), and in cases where there is multicollinearity among analyzed variables. In our case, we applied PLS as the data points were collected along a gradient and constituted non-statistically independent observations which resulted in significant correlations across the potential explanatory variables (such as temperature, salinity, H2S, and NH4+) [48, 49]. All the analyses were performed using the XLSTAT-Ecology software (Addinsoft, Paris, France).
Results
Environmental Characteristics of the Water Column
All four campaigns were characterized by high PAR values (2539–2787 μM m−2 s−1) at the lake surface. There were significant differences in rainfall between seasons: 1138–1056 mm during the rainy season (2014–2015, from November to March) and 53–154 mm during the dry season (2014–2015, from April to October). Several physico-chemical characteristics were clearly identified depending on the season and depth, along the water column (Fig. 2). The water column exhibited two distinct physico-chemical profiles depending on the sampling period:
-
At the end of the rainy season (April 2014 and 2015), we observed a stratified water column with two distinct layers well separated by physical and chemical clines. The surface layer (≤ 2.2 m) was characterized by weak light penetration (Kd = 11.6–12.7 m−1), temperatures between 30 °C and 33 °C, high level of oxygen saturation (15%–400%), low level of H2S (≤ 100 μM), high pH values (> 9.5 in 2014 and > 9.2 in 2015), and moderate salinity (35–45 psu). The intermediate layer (2.2–16 m) was characterized by the absence of light penetration, no oxygen, lower pH (9.5–8.9 in 2014 and 9.0–8.7 in 2015), and constant salinity (≈ 65 psu). Low NH4+ and PO43− concentrations (15 ± 14 and 23 ± 38 μM, respectively) were observed in the surface and intermediate layers during the stratified season.
-
At the end of the dry season (October 2014 and November 2015), light penetration was weak (Kd = 18.4 m−1), temperature was high, and oxygen saturation was observed at the surface of the lake (≤ 2.2 m). Furthermore, the salinity, pH, and H2S clines disappeared leading to a non-stratified water column (salinity ≈ 65 psu, pH ≈ 9.0, H2S ≈ 200 μM), from the surface to the deep permanent layers. Strong gradients of inorganic nutrients were also observed in the lake column, with low NH4+ and PO43− concentrations (15 ± 14 and 23 ± 38 μM, respectively) in the intermediate layer and very high and variable NH4+ and PO43−concentrations (2653 ± 2133 and 172 ± 114 μM, respectively) in the intermediate and bottom layers.
Changes in the main physico-chemical characteristics (temperature, salinity, soluble sulfide (H2S), and oxygen saturation (%O2) as a function of depth and sampling campaign. April 2014 and 2015: stratified water column, end of the rainy season. October 2014 and November 2015: non-stratified period, end of the dry season
During both seasons and all the sampling campaigns, a deep layer characterized by no light penetration, no oxygen, high salinity (≈ 70 psu), low pH values (< 9.0), and very high H2S concentration (> 2000 μM) was observed at the bottom of the water column (14–17 m).
Phytoplankton biomass, estimated by Chla concentration, also exhibited vertical gradients, with higher and less variable values observed in the upper layers (mean2014–2015 = 652 ± 179 μg L−1) than those in the bottom layers (mean2014–2015 = 171 ± 201 μg L−1).
Composition and Structure of Phytoplanktonic Communities
Taxonomic and Phylogenetic Affiliation of Phytoplanktonic OTUs
Using 16S rRNA genes amplicon, we obtained 455 16S rRNA-OTUs including bacteria, cyanobacteria, and eukaryotic plastids. The phytoplanktonic community was composed of 15 OTUs, with eight OTUs being affiliated to cyanobacteria and seven OTUs to eukaryotic plastids (Tables 1, S1, and S2).
Each cyanobacterial OTU was affiliated either to the genus or species level (Tables 1 and S1). The phylogeny of cyanobacteria (Fig. S1) showed that the eight observed OTUs belonged to the orders Oscillatoriales (A. fusiformis), Synechococcales (Family Synechococcaceae: Synechococcus, Family Leptolyngbyaceae: Sodalinema komarekii and Leptolyngbya spp. 1 to 4), and Pleurocapsales (Xenococcus sp.). Among these eight OTUs, two species (A. fusiformis and S. komarekii) have been already described in the isolated strains from Lake Dziani Dzaha by a polyphasic approach [18].
The seven eukaryotic plastid-related OTUs (Table S2) were distributed to three phyla (Fig. S2): (i) Ochrophyta, including three OTUs affiliated to the Cymbellaceae and Cymbellales (sp. 1 to 3), but the bootstrap scores did not support the inclusion of these OTUs to any genus or species and two OTUs to the Naviculaceae (sp.) and Naviculales (Gyrosigma sp.), (ii) Myzozoa, with one OTU closely related to Kryptoperidinium foliaceum within the order Peridiniales, and (iii) Chlorophyta, with one OTU affiliated to P. salinarum that forms a separate phylogenetic lineage within Prasinophyceae [14, 50].
Contribution of Cyanobacteria and Photosynthetic Eukaryotes to the Diversity of 16S rRNA Genes
Although the phytoplanktonic community represented only 3.3% of the total observed richness (15 OTUs among the 455 OTUs in the total dataset, data not shown), it nonetheless represented 52% of the total number of sequences (18,747,809 sequences). A. fusiformis (8,249,182 sequences) and P. salinarum (1,480,251 sequences) were the two most abundant taxa of the planktonic community in Lake Dziani Dzaha. Among the photosynthetic microorganisms, A. fusiformis and P. salinarum were, by far, the only dominant microorganisms, representing 99.99% of the total number of cyanobacterial and photosynthetic eukaryote sequences, respectively (Table 1). Photographs of these two dominant OTU representatives are presented in Fig. S3.
Changes in the Abundance of the Dominant Phytoplanktonic Taxa along the Water Column
The over-dominance of only two phytoplanktonic, P. salinarum and A. fusiformis, provided an opportunity to easily and accurately estimate their abundance by microscopy and flow cytometry, respectively. The observed mean abundances were high, with 1.44 × 106 and 7.78 × 105 cells mL−1 for A. fusiformis and P. salinarum, respectively (Table 2). Taking into account all the samples, the standardized number of sequences of these two taxa were significantly related to their abundance values estimated using FCM (Fig. S4). The relationship was linear for Picocystis but asymptotic for Arthrospira, probably as a consequence of the finite standardized number of analyzed sequences versus theoretically limitless number of cells counted using FCM.
The maximum values were recorded in the upper euphotic layer (Fig. 3; P. salinarum: 2.67 × 106 cells mL−1, April 2014; A. fusiformis: 4.66 × 106 cells mL−1, October 2014).
Large variations in the abundance of species were observed with depth (Fig. 3), with both the taxa exhibiting a decreasing trend with depth (Spearman rho = − 0.554, P < 10−3 for A. fusiformis and − 0.665, P < 10−4 for P. salinarum). However, we observed differences in these trends according to the stratification state of the water column (Fig. 3). During the stratified period (April 2014 and 2015), the abundance of both taxa showed a quasi-exponential decrease below the thin euphotic layer. During the non-stratified period (October 2014 and November 2015), small variations in the abundance of the two taxa were observed, notably between the surface and 14–16 m bottom layers, where the physico-chemical characteristics of the water column were homogeneous. Below this limit, the abundance of both the species exhibited a significant decrease. The relative decrease in taxa abundance between the top and bottom layers of the water column was very high and constant for A. fusiformis (93%–97%) and highly variable for P. salinarum (73%–99%). The lowest decrease in the abundance of P. salinarum was observed during the non-stratified period (78% in October 2014 and 73% in November 2015).
Relationships between the Abundance of the Dominant Photosynthetic Taxa and Physico-Chemical Characteristics of the Water Column
The results of the PLS regression models are provided in Table 3. The R2 associated with different models were very high, with higher values for P. salinarum than for A. fusiformis model. Among the four tested explanatory variables, temperature was the only variable that positively affected the abundance of P. salinarum and A. fusiformis, and showed relatively low standardized regression coefficient. The three other variables (salinity, log H2S, and log NH4+) negatively correlated with phytoplankton abundance. During the stratified and the non-stratified seasons, the explanatory variable with the highest coefficient value was salinity for P. salinarum, whereas, it was log H2S for A. fusiformis.
Pigment Composition of P. salinarum and A. fusiformis Isolated Strains
The results of the analysis of different pigment classes of the strains P. salinarum and A. fusiformis are presented in Table 4. Picocystis salinarum cells exhibited a higher diversity in their pigment composition than A. fusiformis (nine and five, respectively). Among the accessory pigments, specific carotenoids were exclusively detected in P. salinarum (neoxanthin, violaxanthin, antheraxanthin, and two unknown lipophilic pigments). Specific pigments also existed in A. fusiformis cells, including phycocyanin and two lipophilic pigments (zeaxanthin and an unknown pigment). Expressed as the amount of pigment by cellular volume unit, P. salinarum cells presented higher concentrations of both Chla and b (51.9 ± 12.8 and 21.1 ± 5.8 fg μm−3, respectively) than those of A. fusiformis (6.0 ± 4.9 and 0 fg μm−3, respectively). On the contrary and as expected, A. fusiformis had a high content of phycocyanin (51.1 ± 51.8 fg μm−3), which P. salinarum does not have.
Discussion
Drastic Environmental Changes in the Lake Dziani Dzaha Pelagic Ecosystem
Lake Dziani Dzaha is a tropical thalassohaline crater lake, with unique specific physico-chemical characteristics. The water of the Dziani Dzaha Lake probably originates from the nearby Indian Ocean with further modifications of its physico-chemical characteristics by precipitations, hydrothermal activity, precipitation of dissolved elements, and biogeochemical activity. This marine origin is original compared with that of most known other alkaline and saline lakes that are, for the great majority, inland water bodies whose salinity is due to dissolved continental minerals [1].
The hydrological characteristics of Lake Dziani Dzaha are mostly dependent on the seasonal climatology, which is based on alternating dry and rainy seasons. Low surface salinity and a halocline at a depth of 2.2 m can be attributed to the heavy rainfalls occurring during the rainy season in Mayotte Islands. During dry season, evaporation progressively increased the salinity of the surface layer, easing the mixing of water column and disappearance of the halocline. Therefore, the lake can be considered a monomictic lake (mixing once a year during the southern hemisphere winter) with special features in terms of oxygen content, i.e., permanent anoxia at a depth of approximately 2 m even in the absence of halocline. In summary, the water column in the deepest part of the lake undergoes important changes in its physicochemical characteristics, thus, defining several layers along the water column during both the seasons. These contrast layers, based on the physico-chemical characteristics, were observed between seasons from 2010 to 2015 [1] and this study. They define several environmental niches, which might have strong effects on phytoplanktonic diversity and distribution, along the water column.
The estimated phytoplanktonic biomass, expressed as mean chlorophyll a concentrations, was very high in 2014 (566 μg Chla L−1) and 2015 (692 μg Chla L−1), and in the same range than the ones already observed in 2010 (685 μg Chla L−1) and 2011 (702 μg Chla L−1) [1].
Diversity and Composition of Phytoplanktonic Community
In the present study, the richness of phytoplanktonic community in the lake was low, with only 15 taxa being detected, eight of them belonging to cyanobacteria and seven to photosynthetic eukaryotes. To the best of our best knowledge, no study on the phytoplanktonic diversity of thalassohaline lake have been carried out with metabarcoding and metagenomic approaches. The richness of phytoplanktonic communities of soda lakes located in the Kenyan and Ethiopian Rift Valleys, which are the most studied aquatic continental ecosystems close to Lake Dziani Dzaha, have been mainly evaluated through microscopic identification [5, 51,52,53]. The few studies based on next-generation sequencing (NGS) on the microbial communities of these lakes have focused on prokaryotic communities [12, 54]. The only NGS-based study of both prokaryotic and eukaryotic organisms in several Ethiopian Lakes was by Lanzén et al. [22]. However, the number of phytoplanktonic taxa that can be retrieved from their database was considerably lower than that of other studies on the same lakes but based on microscopy [5, 55].
Considering the microscopy-based richness estimation of phytoplanktonic communities of Kenyan and Ethiopian lakes, which range between 72 (Koka Lake; [5]) and seven (Abijata Lake; [5]), the richness of phytoplanktonic community in Lake Dziani Dzaha was in the lower range and it is among the lowest recorded in the saline-alkaline lakes in this part of the world. Among the crater lakes located in the same geographical and climatic area, a single study in Lake Oloidien (Kenya) reported the characterization of phytoplanktonic communities using NGS [12]. These authors observed up to 30 cyanobacteria and more than 15 eukaryotic taxa. However, Lake Oloidien differs greatly from Lake Dziani Dzaha in terms of salinity, which remains quite lower and more variable (< 4 ppt), the presence of zooplankton and the non-permanent flamingo populations, along with permanent human populations that rely on the lake water for different uses.
Among the 15 phytoplanktonic OTUs, the two significantly dominant ones were A. fusiformis and P. salinarum; both the species being commonly described as dominant phytoplanktonic taxa in equatorial soda lakes [54, 56,57,58]. Among the rare cyanobacteria OTUs, one corresponds to a new species (S. komarekii), which has been recently described in strains isolated from stromatolites, and it might be considered endemic to Lake Dziani Dzaha [18]. The other rare cyanobacterial OTUs (Leptolyngbya spp. 1–4, Synechococcus sp., and Xenococcus sp.) have also been reported with relatively high frequency in the stromatolites of the lake [26]. The fact that all these cyanobacterial species were detected with a very low frequency in the water column and a high frequency in stromatolites, implies that the preferential habitat of these species is probably the stromatolites, where Cyanobacteria have been shown to influence their shape and mineralogy [26].
With respect to the rare eukaryotic plastid-related OTUs, only Kryptoperidinium sp. was detected in stromatolites with a higher frequency than that in the water column [59]. Regarding Bacillariophyta, the assignation level of Cymbellaceae spp. clusters (Family) does not allow comparison with the composition of diatoms from other aquatic ecosystems. However, both Cymbellaceae species and Gyrosigma sp., affiliated with Gyrosigma fasciola, have already been observed in the Mozambique Channel [60]. Gyrosigma sp. has never been reported as a typical species of soda or thalassohaline lakes. Thus, it can be hypothesized that most of the rare eukaryotic species detected in Lake Dziani Dzaha are probably of marine origin. Finally, some common thalassohaline or soda lakes taxa (e.g., Chlorophyta such as Kirchneriella, Monoraphidium, Raphidocelis, Selenastrum, and Tetranephris [23] were not detected in Lake Dziani Dzaha.
Overall, these results support our initial hypothesis of very low taxonomic richness of phytoplanktonic community in the Lake Dziani Dzaha pelagic ecosystem. However, the two-dominant species (A. fusiformis and P. salinarum) were the same as those reported in most saline-alkaline lakes, whereas, the very rare OTUs observed in the water column appeared to originate either from stromatolites or surrounding marine environment. Different and complementary processes can explain the low richness. First, the geographical isolation of this insular crater lake from other alkaline-saline lakes avoids or significantly limits the movement of phytoplanktonic species, which might occur in continental lakes through dispersal agents (such as water, air, animal, and human) [61]. Second, the small size of the lake coupled with drastic environmental conditions in the water column limits the suitable niches for phytoplankton to the thin upper layer of the water column. Third, a main difference from other alkaline or thalassohaline lakes is the absence of grazers (zooplankton, fishes, and birds such as flamingos [62]). The absence of top-down control of phytoplanktonic populations might allow dominant species to establish and persist [63, 64]. Fourth, the large amount of phytoplanktonic biomass suggests that there is no nutrient limitation and implies a strong light attenuation, resulting in a competition for light among phytoplanktonic species leading to the dominance of the best competitors [65].
Only One Prokaryotic and One Eukaryotic Taxa Dominated the Phytoplanktonic Community from the Surface to Bottom Layers of the Water Column
Considering that the above-mentioned processes can explain the observed very low diversity, both competitive exclusion [66] and killing the winner theories [24, 67] predict that only one species can dominate and exclude the others. However, in Lake Dziani Dzaha, we observed the co-dominance of two distinct species in the same environmental niche, which, to the best of our knowledge, has not been reported in saline-alkaline lakes.
A first explanation of this unexpected result might be that co-occurrence is possible because of the distinct capacities of the two taxa to cope with light-limitation (Tables 4 and S3). Light limitation mostly refers to the quantitative aspect, but light is not a single resource, such as nutrients (e.g., NH4+ and PO43−). Light is composed of different wavelengths, and photosynthetic organisms have developed adaptations to use different regions of the photosynthetically active radiation (PAR) through a diverse range of pigments. Recently, Burson et al. [68] proposed an explanation for the unexpected co-occurrence of different phytoplanktonic species, where the best competitor for light is expected to dominate the phytoplanktonic community. In the two dominant taxa of Lake Dziani Dzaha, differences in their pigment composition were obvious (Table 4). For instance, if they share the ubiquitous Chla pigment with absorption peaks at 440 and 680 nm (blue and red regions of the spectrum), P. salinarum cells can exhibit higher concentration of Chla than A. fusiformis, given the potential better efficiency of P. salinarum to capture energy associated with Chla absorption wavelengths. The cyanobacteria A. fusiformis contains the accessory pigment phycocyanin, which has an absorption peak at 630 nm (orange region), whereas, P. salinarum contains Chlb with an absorption peak at 475 (blue-green region) and 650 nm (red region). Furthermore, P. salinarum also contains several other pigments, including diatoxanthin (451 and 479 nm) and monadoxanthin (448 and 475 nm), which enables them to efficiently use the blue-green part of the light spectrum.
A second explanation of the non-competitive access to light resources by these two taxa in the thin surface layer of the water column might be the significant difference in their size. To prevent sinking, the large filaments of A. fusiformis benefit from intracellular aerotopes, which regulate their buoyancy [69]. Due to their very small size, P. salinarum cells have a very low intrinsic sinking rate [70] that can be lowered by the viscosity of lake water where salinity is higher than that of seawater. Thus, these two taxa exhibited distinct but non-competitive approach to remain in the upper part of the euphotic layer, where they attained the maximum abundance.
Coupled with the low sinking property they confer to the phytoplanktonic cells [71], high surface/volume ratio (Table S3 and Fig. S3), as in picophytoplanktonic cells, compared with that of nano- or microphytoplanktonic cells, has also been proposed as a way to cope with light limitation [72]. In the present study, these two co-dominant taxa were also observed and enumerated in all the samples by both microscopy and flow cytometry. The abundance of these two-species showed that they mostly co-occur and exhibit a significant decrease with depth, especially when the water column is stratified. However, if we hypothesize that most, if not all, the cells of both taxa were produced in the top layer of the lake and then sank, it is remarkable that during non-stratified periods, approximately 25% of Picocystis cells were able to survive in the extreme hostile physico-chemical environments that prevail in the bottom layers of the water column. Contrarily, and irrespective of the water column structure, only a small percent of abundance of Arthrospira cells in the top layer was observed in these bottom layers. To the best of our knowledge, such differential behavior has not been reported by the studies on soda lakes (Table S3).
Factors Regulating the Dynamics of the Two Dominant Phytoplanktonic Taxa
As mentioned previously, it is probable that high PAR in the upper layer allowed active growth of the two photosynthetic taxa, but they also seem to maintain minimum growth when the PAR decreased. The ability to grow in low-light environments has been demonstrated for P. salinarum, with a positive growth at very low irradiance (0.6 μM photons m−2 s−1), showing its high potential for photoacclimatation and shade-adapted photosynthesis [73]. The high Chla pigment concentration, as observed in the isolated strains of the present study, might also help this species to optimize its ability to cope with very low light intensity [74]. Similarly, specific adaptive mechanisms to very low irradiance have been developed by Arthrospira, which can increase the cellular pigment concentrations [75].
From the upper layer to the bottom of the water column, the results of statistical modeling showed that the abundance of the two over-dominant taxa were significantly correlated with all the tested environmental factors. The abundance of P. salinarum and A. fusiformis positively correlated with the temperature. Temperatures of near or over 30 °C has been shown to potentially favor the growth of the two taxa [73,74,75,76]. The negative relationships between the abundance of the two taxa versus salinity, H2S, and NH4+ indicated that below the upper layer, they were both subjected to adverse conditions.
The negative relationship with H2S can result from the effect of anoxic conditions and/or the direct toxic effect of H2S. These two taxa might have different ways to cope with high levels of H2S, but the underlying mechanisms remain to be elucidated.
Growth and photosynthetic activity of P. salinarum strains were observed under anoxic conditions and after treatment with 100 μM Na2S with comparable rates than those measured under oxic conditions [73]. The results suggested that the ability to maintain oxygenic photosynthesis even under low light condition might allow the proximal environment of P. salinarum cells to be suboxic, thus, reducing the redox stress. However, below the surface layer of the lake, no light was available for photosynthesis and during the stratified season the H2S concentration was significantly above 1000 μM. The significant abundance of P. salinarum in the bottom layers of the lake suggests that they are highly efficient, but unknown processes must operate to maintain the cell integrity.
Different types of adaptations to H2S have been defined among cyanobacteria based on the differential sensitivity of photosystems II and I to sulfide, and the capacity to carry out anoxygenic photosynthesis [76, 77]. However, the described processes can only function if a minimum intensity of light is available [78]. In dark and in anoxic conditions, cyanobacteria have the capacity to perform fermentative metabolism coupled with sulfur reduction [79]. Dark and anaerobic conditions have been shown to enhance the survival of Oscillatoria terebreformis compared with that under aerobic conditions [74].
The negative relationships between the abundance of the two taxa and NH4+ content probably originated from the significant increase in its concentration in the bottom waters. The observed values were among the highest recorded in aquatic ecosystems [80], and therefore, might be toxic or sub-toxic for both the taxa. Additionally, under high temperature and pH conditions observed in the lake, the ratio between toxic free ammonia (NH3) versus ammonium (NH4+) might increase, further affecting cell growth and viability [81].
Salinity has also been reported by the model as a significant explanatory variable with a potential negative effect on the abundance of Picocystis and Arthrospira. The regression coefficients were relatively high for Picocystis, suggesting the higher sensitivity of Picocystis to salinity variations in the water column. However, the field observations of different studies suggest that P. salinarum is more tolerant to high salinity values (10–300 psu) than A. fusiformis (20–70 psu) [53]. In a laboratory experiment, Kebede [82] studied the growth of A. fusiformis strains subjected to a wide range of salinity and different salt compositions. They found an inverse relationship between salinity and growth, although the growth rate was still positive at a salinity of 90 psu (Table S3).
The apparent high sensitivity of P. salinarum to salinity in Lake Dziani Dzaha might be attributed to the conjunction of several adverse conditions that probably did not co-exist in the other ecosystems, where this species has been observed, and that have not been experimentally tested simultaneously.
Considering the above results, we can hypothesize that the co-existence of these two taxa in the same environmental niche is based on different adaptive features they might have to cope with light-limitation and adverse environmental conditions. To the best of our knowledge, this is the first in situ example of the role that niche differentiation in the light spectrum can play to complete the “nutrient-load hypothesis” [68]. However, one can ask why the coexistence of these two taxa was not reported in other saline-alkaline lakes, with the exception of one crater lake investigated by Krienitz et al. [14]. These authors provided the most probable explanation for the few reports of Picocystis as a co-occurrent species of cyanobacteria, writing that “This tiny protist is difficult to recognize and distinguish in field samples and is probably often neglected or wrongly identified.” During this study, we had the opportunity to use flow cytometry, which is currently the most efficient tool to enumerate picophytoplanktonic cells [83] and we also benefited from P. salinarum isolates to calibrate FCM analyses.
Regarding the ecological consequences of this co-existence, the two species might sustain two different trophic networks owing to their different morphological traits. The small size of P. salinarum might support a microbial loop [84] in the surface layer of the lake. Not only bacteria but also protozoans that have been detected in the lake [59] might benefit from the presence of these small phytoplanktonic cells either as a source of organic matter for aerobic heterotrophic bacteria or as a prey for nanoflagellates or protozoa. In contrast, due to their large size A. fusiformis filaments cannot be ingested by protozoans. By sedimentation, these filamentous organisms might act as a biological CO2 pump [85] and as the main source of (i) organic matter in the detrital bacterial loop of the anoxic bottom layer of the lake and (ii) long-term carbon stock stored in the sediment of the lake.
The presence of intact A. fusiformis and P. salinarum cells in the deepest layers of the lake, although representing only a small fraction of the abundance observed in the upper layer, implies that a part of the population of these two phylogenetically and morphologically distinct taxa was able to persist in extremely adverse conditions (anoxia, high H2S and NH4+ concentration, and no light). It is probable that the physiological status of the upper- and lower-layer cells is different. In the future, the survival behavior and physiological status of cells from isolated strains of these two taxa under controlled conditions should be evaluated. Furthermore, the transcriptome and metabolome of the two taxa along the depth profile should also be analyzed in order to understand the changes in certain metabolic pathways as a response to such extreme environmental conditions.
In conclusion, due to its geographical isolation, low anthropogenic pressure, thalassohaline signature associated with extreme physico-chemical conditions with a thin euphotic layer, and absence of grazers, Lake Dziani Dzaha is an excellent model to study the composition and functioning of a “simple” microbial aquatic ecosystem. By analyzing the phytoplanktonic community of this lake through high-depth sequencing approach, we verified the hypothesis that this community exhibits a very low richness and diversity like in other isolated and extreme aquatic ecosystems. Two OTUs (A. fusiformis and P. salinarum) appeared as the sole true pelagic phytoplanktonic OTUs in this lake. Furthermore, they not only appeared to form the minimal community that can be encountered in a present or “primitive” aquatic ecosystem, but also co-existed as a stable co-dominant consortium, at least according to the data collected during our four campaigns. Our findings are different from what is proposed by the classical community ecology hypotheses, according to which, in Lake Dziani Dzaha, only one species should have dominated the ecosystem. The two phylogenetically distinct OTUs (Cyanobacteria and Chlorophyta) showed distinct functional traits (pigment composition and size) that can explain their co-existence in the euphotic layer. Because of adverse environmental conditions that prevail below the surface layer, both the taxa exhibited a significant decrease in their abundances. Their behavior in the aphotic and anoxic water layers should be explored, and it constitutes one of the interesting challenges for future studies.
References
Leboulanger C, Agogué H, Bernard C, Bouvy M, Carré C, Cellamare M, Duval C, Fouilland E, Got P, Intertaglia L, Lavergne C, le Floc’h E, Roques C, Sarazin G (2017) Microbial diversity and cyanobacterial production in Dziani Dzaha crater lake, a unique tropical thalassohaline environment. PLoS One 12:1–28. https://doi.org/10.1371/journal.pone.0168879
Cadeau P (2017) Caractérisation des cycles géochimiques dans un analogue potentiel des océans à 2.2 Ga : le lac DzianiDzaha, Mayotte. http://www.ipgp.fr/fr/gis/these-de-pierre-cadeau. Accessed 7 Sep 2018
Fazi S, Butturini A, Tassi F, Amalfitano S, Venturi S, Vazquez E, Clokie M, Wanjala SW, Pacini N, Harper DM (2018) Biogeochemistry and biodiversity in a network of saline–alkaline lakes: implications of ecohydrological connectivity in the Kenyan Rift Valley. Ecohydrol. Hydrobiol. 18:96–106. https://doi.org/10.1016/j.ecohyd.2017.09.003
Wood, R. & Talling, J. (1988). Chemical and algal relationships in a salinity-series of Ethiopian inland waters. Hydrobiologia 15: 29–67. In: Chem. algal relationships a Salin. Ethiop. Inl. waters. http://www.sciepub.com/reference/79101. Accessed 23 Nov 2018
Kebede E, Willén K Phytoplankton in a salinity–alkalinity series of lakes in the Ethiopian Rift Valley Algol Stud, 89 (1998), pp. 63–96
Abate R, Kifle D, Lemma B, Gao Y (2014) Phytoplankton community and physico-chemical seasonality and changes in Lake Hora-Kilole, Ethiopia, a tropical crater lake. African J Aquat Sci 39:97–108. https://doi.org/10.2989/16085914.2013.867253
Lemma B (2003) LIMNOLOGICA ecological changes in two Ethiopian lakes caused by contrasting human intervention
Melack JM (1979) Photosynthesis and growth of Spirulina platensis (Cyanophyta) in an equatorial lake (Lake Simbi, Kenya)1. Limnol. Oceanogr. 24:753–760. https://doi.org/10.4319/lo.1979.24.4.0753
Finlay BJ, Curds CR, Bamforth SS, Bafort JM (1987) Ciliated protozoa and other microorganisms from two African Soda Lakes (Lake Nakuru and Lake Simbi, Kenya). Arch. Protistenkd. 133:81–91. https://doi.org/10.1016/S0003-9365(87)80041-6
Njuguna SG (1988) Nutrient-phytoplankton relationships in a tropical meromictic soda lake. Saline Lakes. Springer, Dordrecht, pp 15–28
Ballot A, Krienitz L, Kotut K, Wiegand C, Pflugmacher S (2005) Cyanobacteria and cyanobacterial toxins in the alkaline crater lakes Sonachi and Simbi, Kenya. Harmful Algae 4:139–150. https://doi.org/10.1016/J.HAL.2004.01.001
Luo W, Li H, Kotut K, Krienitz L (2017) Molecular diversity of plankton in a tropical crater lake switching from hyposaline to subsaline conditions: lake Oloidien, Kenya. Hydrobiologia 788:205–229. https://doi.org/10.1007/s10750-016-2998-x
Rumes B, Eggermont H, Verschuren D (2011) Distribution and faunal richness of Cladocera in western Uganda crater lakes. Hydrobiologia 676:39–56. https://doi.org/10.1007/s10750-011-0829-7
Krienitz L, Bock C, Kotut K, Luo W (2012) Picocystis salinarum (Chlorophyta) in saline lakes and hot springs of East Africa. Phycologia 51:22–32. https://doi.org/10.2216/11-28.1
Nkambo M, Bugenyi FW, Naluwayiro J, et al (2015) Planktonic and fisheries biodiversity of alkaline saline crater lakes of Western Uganda
Oberholster PJ, Botha A-M, Ashton PJ (2009) Appearance of new taxa: invertebrates, phytoplankton and bacteria in an alkaline, saline, meteorite crater lake, South Africa. Fundam Appl Limnol / Arch für Hydrobiol 174:271–282. https://doi.org/10.1127/1863-9135/2009/0174-0271
Mathelin E (2013) Les lacs insulaires du sud-ouest de l’océan indien, un enjeu pour la gestion durable de la ressource en eau. Université d'Orléans, Dissertation
Cellamare M, Duval C, Drelin Y, Djediat C, Touibi N, Agogué H, Leboulanger C, Ader M, Bernard C (2018) Characterization of phototrophic microorganisms and description of new cyanobacteria isolated from the saline-alkaline crater-lake Dziani Dzaha (Mayotte, Indian Ocean). FEMS Microbiol. Ecol. 94. https://doi.org/10.1093/femsec/fiy108
Eiler A, Drakare S, Bertilsson S, Pernthaler J, Peura S, Rofner C, Simek K, Yang Y, Znachor P, Lindström ES (2013) Unveiling distribution patterns of freshwater phytoplankton by a next generation sequencing based approach. PLoS One 8:1–10. https://doi.org/10.1371/journal.pone.0053516
Xiao X, Sogge H, Lagesen K, Tooming-Klunderud A, Jakobsen KS, Rohrlack T (2014) Use of high throughput sequencing and light microscopy show contrasting results in a study of phytoplankton occurrence in a freshwater environment. PLoS One 9:e106510. https://doi.org/10.1371/journal.pone.0106510
Johnson ZI, Martiny AC (2015) Techniques for quantifying phytoplankton biodiversity. Annu. Rev. Mar. Sci. 7:299–324. https://doi.org/10.1146/annurev-marine-010814-015902
Lanzén A, Simachew A, Gessesse A, Chmolowska D, Jonassen I, Øvreås L (2013) Surprising prokaryotic and eukaryotic diversity, community structure and biogeography of Ethiopian Soda Lakes. PLoS One 8:e72577. https://doi.org/10.1371/journal.pone.0072577
Luo W, Kotut K, Krienitz L (2013) Hidden diversity of eukaryotic plankton in the soda lake Nakuru, Kenya, during a phase of low salinity revealed by a SSU rRNA gene clone library. Hydrobiologia 702:95–103. https://doi.org/10.1007/s10750-012-1310-y
Thingstad TF, Lignell R (1997) Theoretical models for the control of bacterial growth rate, abundance, diversity and carbon demand. Aquat. Microb. Ecol. 13:19–27. https://doi.org/10.3354/ame013019
Tilman D, Kilham SS, Kilham P (1982) Phytoplankton community ecology: the role of limiting nutrients. Annu. Rev. Ecol. Syst. 13:349–372
Gérard E, De Goeyse S, Hugoni M et al (2018) Key role of Alphaproteobacteria and Cyanobacteria in the formation of stromatolites of Lake Dziani Dzaha (Mayotte, Western Indian Ocean). Front. Microbiol. 9. https://doi.org/10.3389/fmicb.2018.00796
Gran G, Dahlenborg H, Laurell S, Rottenberg M (1950) Determination of the equivalent point in potentiometric titrations. Acta Chem. Scand. 4:559–577. https://doi.org/10.3891/acta.chem.scand.04-0559
Ritchie RJ (2006) Consistent sets of spectrophotometric chlorophyll equations for acetone, methanol and ethanol solvents. Photosyn Res 89:27–41. https://doi.org/10.1007/s11120-006-9065-9
Bouvy M, Got P, Domaizon I, Pagano M, Leboulanger C, Bouvier C, Carré C, Roques C, Dupuy C (2016) Plankton communities in the five Iles Eparses (Western Indian Ocean) considered to be pristine ecosystems. Acta Oecol. 72:9–20. https://doi.org/10.1016/j.actao.2015.10.013
Catherine A, Selma M, Mouillot D, Troussellier M, Bernard C (2016) Patterns and multi-scale drivers of phytoplankton species richness in temperate peri-urban lakes. Sci. Total Environ. 559:74–83. https://doi.org/10.1016/j.scitotenv.2016.03.179
Rippka R (1988) Isolation and purification of cyanobacteria. Meth Enzym 167:3–27
Catherine A, Maloufi S, Congestri R, Viaggiu E, Pilkaityte R (2017) Cyanobacterial samples: preservation, enumeration, and biovolume measurements. In: Meriluoto J, Spoof L, Codd G (eds) Handbook of cyanobacterial monitoring and cyanotoxin analysis, edition : 1st. Wiley, Editors, pp 333–334
Yéprémian C, Catherine A, Congestri R, Bernard C, Elersek T, Pilkaityte R (2017) Chlorophyll a extraction and determination. In: Meriluoto J, Spoof L, Codd G (eds) Handbook of cyanobacterial monitoring and cyanotoxin analysis, edition: 1st. Wiley, Editors, pp 315–330
Ras J, Claustre H, Uitz J (2007) Spatial variability of phytoplankton pigment distributions in the subtropical South Pacific Ocean: comparison between in situ and predicted data. Biogeosci. Discuss. 4:3409–3451. https://doi.org/10.5194/bgd-4-3409-2007
Sun J, Liu D (2003) Geometric models for calculating cell biovolume and surface area for phytoplankton. J. Plankton Res. 25:1331–1346. https://doi.org/10.1093/plankt/fbg096
Schuurman T, de Boer RF, Kooistra-Smid AMD, van Zwet AA (2004) Prospective study of use of PCR amplification and sequencing of 16S ribosomal DNA from cerebrospinal fluid for diagnosis of bacterial meningitis in a clinical setting. J. Clin. Microbiol. 42:734–740. https://doi.org/10.1128/JCM.42.2.734-740.2004
Walters W, Hyde ER, Berg-Lyons D, Ackermann G, Humphrey G, Parada A, Gilbert JA, Jansson JK, Caporaso JG, Fuhrman JA, Apprill A, Knight R (2016) Improved bacterial 16S rRNA gene (V4 and V4-5) and fungal internal transcribed spacer marker gene primers for microbial community surveys. mSystems 1:e00009-15 . 10.1128/mSystems.00009-15, 1
Magoč T, Salzberg SL (2011) FLASH: fast length adjustment of short reads to improve genome assemblies. Bioinformatics 27:2957–2963. https://doi.org/10.1093/bioinformatics/btr507
Mahé F, Rognes T, Quince C, de Vargas C, Dunthorn M (2014) Swarm: robust and fast clustering method for amplicon-based studies. PeerJ 2:e593. https://doi.org/10.7717/peerj.593
Rognes T, Flouri T, Nichols B, Quince C, Mahé F (2016) VSEARCH: a versatile open source tool for metagenomics. PeerJ 4:e2584. https://doi.org/10.7717/peerj.2584
Bokulich NA, Subramanian S, Faith JJ, Gevers D, Gordon JI, Knight R, Mills DA, Caporaso JG (2013) Quality-filtering vastly improves diversity estimates from Illumina amplicon sequencing. Nat. Methods 10:57–59. https://doi.org/10.1038/nmeth.2276
Wang Q, Garrity GM, Tiedje JM, Cole JR (2007) Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl. Environ. Microbiol. 73:5261–5267. https://doi.org/10.1128/AEM.00062-07
Camacho C, Coulouris G, Avagyan V, Ma N, Papadopoulos J, Bealer K, Madden TL (2009) BLAST+: architecture and applications. BMC Bioinformatics 10:421. https://doi.org/10.1186/1471-2105-10-421
Pruesse E, Quast C, Knittel K, Fuchs BM, Ludwig W, Peplies J, Glockner FO (2007) SILVA: a comprehensive online resource for quality checked and aligned ribosomal RNA sequence data compatible with ARB. Nucleic Acids Res. 35:7188–7196. https://doi.org/10.1093/nar/gkm864
Escudié F, Auer L, Bernard M, Mariadassou M, Cauquil L, Vidal K, Maman S, Hernandez-Raquet G, Combes S, Pascal G (2017) FROGS: find, rapidly, OTUs with galaxy solution. Bioinformatics 34:1287–1294. https://doi.org/10.1093/bioinformatics/btx791
Decelle J, Romac S, Stern RF, Bendif EM, Zingone A, Audic S, Guiry MD, Guillou L, Tessier D, le Gall F, Gourvil P, Dos Santos AL, Probert I, Vaulot D, de Vargas C, Christen R (2015) PhytoREF: a reference database of the plastidial 16S rRNA gene of photosynthetic eukaryotes with curated taxonomy. Mol. Ecol. Resour. 15:1435–1445. https://doi.org/10.1111/1755-0998.12401
Kirchberger PC, Sefc KM, Sturmbauer C, Koblmüller S (2014) Outgroup effects on root position and tree topology in the AFLP phylogeny of a rapidly radiating lineage of cichlid fish. Mol. Phylogenet. Evol. 70:57–62. https://doi.org/10.1016/j.ympev.2013.09.005
Tenenhaus M (1998) La régression PLS: théorie et pratique. Editions TECHNIP
Wold S, Ruhe A, Wold H, Dunn WI (1984) The collinearity problem in linear regression. The partial least squares (PLS) approach to generalized inverses. SIAM J. Sci. Stat. Comput. 5:735–743. https://doi.org/10.1137/0905052
Guillou L, Eikrem W, Chrétiennot-Dinet M, Le Gall F (2004) Diversity of picoplanktonic prasinophytes assessed by direct nuclear SSU rDNA. Protist 155:193–214
Schagerl M, Oduor SO (2003) On the limnology of Lake Baringo (Kenya): II. Pelagic primary production and algal composition of Lake Baringo, Kenya. Hydrobiologia 506-509:297–303
Schagerl M, Oduor SO (2008) Phytoplankton community relationship to environmental variables in three Kenyan Rift Valley saline-alkaline lakes. Mar. Freshw. Res. 59:125–136. https://doi.org/10.1071/MF07095
Schagerl M, Burian A, Gruber-Dorninger M, Oduor SO, Kaggwa MN (2015) Algal communities of kenyan soda lakes with a special focus on Arthrospira Fusiformis. Fottea 15:245–257. https://doi.org/10.5507/fot.2015.012
Dadheech PK, Glöckner G, Casper P, Kotut K, Mazzoni CJ, Mbedi S, Krienitz L (2013) Cyanobacterial diversity in the hot spring, pelagic and benthic habitats of a tropical soda lake. FEMS Microbiol. Ecol. 85:389–401. https://doi.org/10.1111/1574-6941.12128
Ogato T, Kifle D (2017) Phytoplankton composition and biomass in tropical soda Lake Shala: seasonal changes in response to environmental drivers. https://doi.org/10.1111/lre.12169
Krienitz L, Dadheech PK, Kotut K (2013) Mass developments of the cyanobacteria Anabaenopsis and Cyanospira (Nostocales) in the soda lakes of Kenya: ecological and systematic implications. Hydrobiologia 703:79–93. https://doi.org/10.1007/s10750-012-1346-z
Sorokin DY, Berben T, Melton ED, Overmars L, Vavourakis CD, Muyzer G (2014) Microbial diversity and biogeochemical cycling in soda lakes. Extremophiles 18:791–809. https://doi.org/10.1007/s00792-014-0670-9
Kambura AK (2016) Metagenomic and metatranscriptomic analysis of bacterial, archaeal and fungal communities within the hot springs of lakes Magadi in Kenya ( Biotechnology ) University of Jomo Kenya
Hugoni M, Escalas A, Bernard C, et al (2018) Spatiotemporal variations in microbial diversity across the three domains of life in a tropical thalassohaline lake (Dziani Dzaha, Mayotte Island). 1–12 . https://doi.org/10.1111/mec.14901
Sournia A (1968) A checklist of planktonic diatoms and dino-flagellates from the Mozambique channel. Bull. Mar. Sci. 20:678–695
Padisák J, Vasas G, Borics G (2016) Phycogeography of freshwater phytoplankton: traditional knowledge and new molecular tools. Hydrobiologia 764:3–27
Schagerl M, Burian A (2016) The ecology of African Soda Lakes: driven by variable and extreme conditions
Pančić M, Kiørboe T (2018) Phytoplankton defence mechanisms: traits and trade-offs. Biol. Rev. 93:1269–1303
Leibold MA, Hall SR, Smith VH, Lytle DA (2017) Herbivory enhances the diversity of primary producers in pond ecosystems. Ecology 98:48–56. https://doi.org/10.1002/ecy.1636
Brauer VS, Stomp M, Huisman J (2012) The nutrient-load hypothesis: patterns of resource limitation and community structure driven by competition for nutrients and light. 179:721–740 . https://doi.org/10.1086/665650
Tilman D (1977) Resource competition between plankton algae: an experimental and theoretical approach. Ecology 58:338–348. https://doi.org/10.2307/1935608
Thingstad TF (2000) Elements of a theory for the mechanisms controlling abundance, diversity, and biogeochemical role of lytic bacterial viruses in aquatic systems. Limnol. Oceanogr. 45:1320–1328. https://doi.org/10.4319/lo.2000.45.6.1320
Burson A, Stomp M, Greenwell E, Grosse J, Huisman J (2018) Competition for nutrients and light: testing advances in resource competition with a natural phytoplankton community. Ecology 0:1–11 . https://doi.org/10.1002/ecy.2187, 99
Likens GE (2009) Biogeochemistry of inland waters: a derivative of encyclopedia of inland waters. Academic Press/Elsevier
Naselli-Flores L Fight on plankton! Or, phytoplankton shape and size as adaptive tools to get ahead in the struggle for life. Cryptogam Algol
Chindia JA, Figueredo CC (2018) Phytoplankton settling depends on cell morphological traits, but what is the best predictor ? Hydrobiologia 813:51–61. https://doi.org/10.1007/s10750-018-3505-3
Kruk C, Martínez A, Nogueira L, et al (2014) Morphological traits variability reflects light limitation of phytoplankton production in a highly productive subtropical estuary (Río de la Plata, South America ). https://doi.org/10.1007/s00227-014-2568-6
Roesler CS, Culbertson CW, Etheridge SM, Goericke R, Kiene RP, Miller LG, Oremland RS (2002) Distribution, production, and ecophysiology of Picocystis strain ML in Mono Lake, California. Limnol. Oceanogr. 47:440–452
Richardson LL, Castenholz RW (1987) Enhanced survival of the cyanobacterium Oscillatoria terebriformis in darkness under anaerobic conditions. Appl. Environ. Microbiol. 53:2151–2158
Schwaderer AS, Yoshiyama K, De Tezanos Pinto P et al (2011) Eco-evolutionary differences in light utilization traits and distributions of freshwater phytoplankton. Limnol. Oceanogr. 56:589–598. https://doi.org/10.4319/lo.2011.56.2.0589
Cohen Y, Jorgensen BB, Revsbech NP, Poplawski R (1986) Adaptation to hydrogen sulfide of oxygenic and anoxygenic photosynthesis among cyanobacteria. Appl. Environ. Microbiol. 51:398–407
Miller SR, Bebout BM (2004) Variation in sulfide tolerance of photosystem II in phylogenetically diverse cyanobacteria from sulfidic habitats. Appl. Environ. Microbiol. 70:736–744. https://doi.org/10.1128/AEM.70.2.736-744.2004
Castenholz RW, Utkilen HC (1984) Physiology of sulfide tolerance in a thermophilic Oscillatoria. Arch. Microbiol. 138:299–305. https://doi.org/10.1007/BF00410894
Stal LJ, Moezelaar R (1997) Fermentation in cyanobacteria. FEMS Microbiol. Rev. 21:179–211. https://doi.org/10.1111/j.1574-6976.1997.tb00350.x
Collos Y, Harrison PJ (2014) Acclimation and toxicity of high ammonium concentrations to unicellular algae. Mar. Pollut. Bull. 80:8–23. https://doi.org/10.1016/j.marpolbul.2014.01.006
Markou G, Depraetere O, Muylaert K (2016) Effect of ammonia on the photosynthetic activity of Arthrospira and chlorella: a study on chlorophyll fluorescence and electron transport. Algal Res. 16:449–457. https://doi.org/10.1016/j.algal.2016.03.039
Kebede E (1997) Response of Spirulina platensis (Arthrospira fusiformis) from Lake Chitu, Ethiopia, to salinity stress from sodium salts. J. Appl. Phycol. 9:551–558
Sosik HM, Olson RJ, Armbrust EV (2010) Flow cytometry in phytoplankton research. In: Chlorophyll a Fluorescence in aquatic sciences: methods and applications
Azam F, Fenchel T, Field J, Gray JS, Meyer-Reil LA, Thingstad F (1983) The ecological role of water column microbes in the sea. Mar. Ecol. Prog. Ser. 10:257–263. https://doi.org/10.2307/24814647
Longhurst AR, Glen Harrison W (1989) The biological pump: profiles of plankton production and consumption in the upper ocean. Prog. Oceanogr. 22:47–123. https://doi.org/10.1016/0079-6611(89)90010-4
Acknowledgments
The authors highly thank M. Bouvy, P. Cadeau, and E. Fouilland for their support during prospecting campaigns on Lake Dziani Dzaha. They also thanks J. Ras from the SAPIGH analytical platform of the Laboratoire d’Océanographie de Villefranche (CNRS-France) for lipophilic pigments analysis. Field permit was granted by the Conservatoire du Littoral et des Rivages Lacustres, Antenne Océan Indien, since Lake Dziani Dzaha is currently a protected water body with free public access but restricted activities, under the control of the French agency for littoral ecosystems conservation (http://www.conservatoire-du-littoral.fr/). The authors thank the anonymous referees for providing valuable suggestions that significantly improved the quality of the manuscript. The authors also would like to thank Editage (www.editage.com) for English language editing.
Funding
This work was supported by Agence Nationale de la Recherche (France, http://www.agence-nationale-recherche.fr, DZIANI project, grant no. ANR-13-BS06-0001), and Total Corporate Foundation (DZAHA project, grant no. C001493).
Author information
Authors and Affiliations
Contributions
CB, HA, MH, DJ, CL, MA, and MT have worked on the design of the experiments. CB, NV, CD, CC, PG, GS, DJ, CL, MA, VG, and CB have performed the experiments. MH, EA, MT, and CB have performed the data analysis. CB, MT, MH, MA, and AE have written the paper.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no competing interests.
Electronic supplementary material
Table S1
Cyanobacterial OTU affiliation obtained by alignment of 16S rDNA sequences with NCBI BLAST tool, Genbank Databases, and sequences from strains isolated from Lake Dziani Dzaha [18]. (DOCX 16 kb)
Table S2
Plastid-related OTU affiliation obtained by alignment of 16S rDNA plastids sequences with NCBI BLAST tool, Genbank Databases, and plastids sequences from Phytoref [46]. (DOCX 17 kb)
Table S3
Review of Arthrospira fusiformis (Cyanobacteria) and Picocystis salinarum (Chlorophyta) characteristics. (DOCX 44.4 kb)
Fig. S1
Cyanobacterial OTUs affiliation using consensus maximum likelihood phylogenetic tree based on 16S rRNA gene sequences of representative Cyanobacteria strains isolated from Dziani Dzaha (Cellamare et al., 2018) and Genbank. Numbers above branches indicate bootstrap support (> 50%) from 1000 replicates. Bootstrap values are given in the following order: neighbor-joining/maximum likelihood/maximum parsimony (NL/ML/MP). The cyanobacterial OTUs from Dziani Dzaha are indicated in bold (OTUs 1, 10, 23, 60, 105 et 450). (DOCX 114 kb)
Fig. S2
Eukaryotic plastid-related OTUs affiliation using consensus Maximum Parsimony tree based on 16S rRNA gene sequences of photosynthetic-eukaryotes chloroplasts of representative sequences of Genbank and Phytoref [46]. Numbers above branches indicate bootstrap support (> 50%) from 1000 replicates. Bootstrap values are given in the following order: neighbor-joining/maximum likelihood/maximum parsimony (NL/ML/MP). OTUs from Lake Dziani Dzaha are indicated in bold (OTUs 2, 46, 115, 248 et 269). (DOCX 93 kb)
Fig. S3
Light microscope micrographs of a environmental sample from Lake Dziani Dzaha (DZ-15-11 campaign) with Arthrospira fusiformis straight filaments and unicellular cells of Picocystis salinarum, b culture of Arthrospira fusiformis strain (PMC 851.14), c culture of Picocystis salinarum strain (ALCP 144.1) © C. Duval, MNHN. Scale bar = 10 μm. (DOCX 2904 kb)
Fig. S4
Comparison of normalized sequences number with the cells abundance (cells mL−1) for the two dominant taxa: Arthrospira fusiformis and Picocystis salinarum. Data all the four campaigns (n = 28) have been plotted. (DOCX 20 kb)
Rights and permissions
OpenAccess This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Bernard, C., Escalas, A., Villeriot, N. et al. Very Low Phytoplankton Diversity in a Tropical Saline-Alkaline Lake, with Co-dominance of Arthrospira fusiformis (Cyanobacteria) and Picocystis salinarum (Chlorophyta). Microb Ecol 78, 603–617 (2019). https://doi.org/10.1007/s00248-019-01332-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-019-01332-8