Abstract
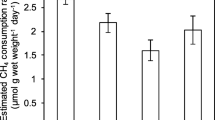
Methanotrophs are important microbial communities in coastal ecosystems. They reduce CH4 emission in situ, which is influenced by soil conditions. This study aimed to understand the differences in active aerobic methanotrophic communities in mangrove forest soils experiencing different inundation frequency, i.e., in soils from tidal mangroves, distributed at lower elevations, and from dwarf mangroves, distributed at higher elevations. Labeling of pmoA gene of active methanotrophs using DNA-based stable isotope probing (DNA-SIP) revealed that methanotrophic activity was higher in the dwarf mangrove soils than in the tidal mangrove soils, possibly because of the more aerobic soil conditions. Methanotrophs affiliated with the cluster deep-sea-5 belonging to type Ib methanotrophs were the most dominant methanotrophs in the fresh mangrove soils, whereas type II methanotrophs also appeared in the fresh dwarf mangrove soils. Furthermore, Methylobacter and Methylosarcina were the most important active methanotrophs in the dwarf mangrove soils, whereas Methylomonas and Methylosarcina were more active in the tidal mangrove soils. High-throughput sequencing of the 16S ribosomal RNA (rRNA) gene also confirmed similar differences in methanotrophic communities at the different locations. However, several unclassified methanotrophic bacteria were found by 16S rRNA MiSeq sequencing in both fresh and incubated mangrove soils, implying that methanotrophic communities in mangrove forests may significantly differ from the methanotrophic communities documented in previous studies. Overall, this study showed the feasibility of 13CH4 DNA-SIP to study the active methanotrophic communities in mangrove forest soils and revealed differences in the methanotrophic community structure between coastal mangrove forests experiencing different tide frequencies.






Similar content being viewed by others
References
Chmura GL, Anisfeld SC, Cahoon DR, Lynch JC (2003) Global carbon sequestration in tidal, saline wetland soils. Glob. Biogeochem. Cycles 17:1111. https://doi.org/10.1029/2002gb001917
Kennedy H, Bjork M (2009) Seagrass meadows. In: Laffoley D, Grimsditch G (eds) The management of natural coastal carbon sinks, vol 53. IUCN, Gland, pp. 23–30
Pidgeon E (2009) Carbon sequestration by coastal marine habitats: important missing sinks. In: Laffoley D, Grimsditch G (eds) The management of natural coastal carbon sinks, vol 53. IUCN, Gland, pp. 47–51
Shiau Y, Burchell MR, Krauss KW, Birgand F, Broome SW (2016) Greenhouse gas emissions from a created brackish marsh in eastern North Carolina. Wetlands 36:1009–1024. https://doi.org/10.1007/s13157-016-0815-y
IPCC (2013) Climate change 2013: the physical science basis. http://www.ipcc.ch/report/ar5/wg1/
Bridgham SD, Megonigal JP, Keller JK, Bliss NB, Trettin C (2006) The carbon balance of North American wetlands. Wetlands 26:889–916
Le Mer J, Roger P (2001) Production, oxidation, emission and consumption of methane by soils: a review. Eur. J. Soil Biol. 37:25–50. https://doi.org/10.1016/s1164-5563(01)01067-6
Megonigal JP, Schlesinger WH (2002) Methane-limited methanotrophy in tidal freshwater swamps. Glob. Biogeochem. Cycles 16:35-1–35-10. https://doi.org/10.1029/2001gb001594
Roslev P, King GM (1996) Regulation of methane oxidation in a freshwater wetland by water table changes and anoxia. FEMS Microbiol. Ecol. 19:105–115. https://doi.org/10.1111/j.1574-6941.1996.tb00203.x
Bowman JP, Sly LI, Nichols PD, Hayward AC (1993) Revised taxonomy of the methanotrophs - description of Methylobacter gen-nov, emendation of Methylococcus, validation of Methylosinus and Methylocystis species, and a proposal that the family Methylococcaceae includes only the group-I methanotrophs. Int. J. Syst. Bacteriol. 43:735–753
Gagliano AL, D'Alessandro W, Tagliavia M, Parello F, Quatrini P (2014) Methanotrophic activity and diversity of methanotrophs in volcanic geothermal soils at Pantelleria (Italy). Biogeosciences 11:5865–5875. https://doi.org/10.5194/bg-11-5865-2014
Hanson RS, Hanson TE (1996) Methanotrophic bacteria. Microbiol. Rev. 60:439–471
Wise MG, McArthur JV, Shimkets LJ (1999) Methanotroph diversity in landfill soil: isolation of novel type I and type II methanotrophs whose presence was suggested by culture-independent 16S ribosomal DNA analysis. Appl. Environ. Microbiol. 65:4887–4897
Hakemian AS, Rosenzweig AC (2007) The biochemistry of methane oxidation. Annu. Rev. Biochem. 76:223–241
McDonald IR, Bodrossy L, Chen Y, Murrell JC (2008) Molecular ecology techniques for the study of aerobic methanotrophs. Appl. Environ. Microbiol. 74:1305–1315. https://doi.org/10.1128/aem.02233-07
Knief C (2015) Diversity and habitat preferences of cultivated and uncultivated aerobic methanotrophic bacteria evaluated based on pmoA as molecular marker. Front. Microbiol. 6:1346. https://doi.org/10.3389/fmicb.2015.01346
Segarra KEA, Comerford C, Slaughter J, Joye SB (2013) Impact of electron acceptor availability on the anaerobic oxidation of methane in coastal freshwater and brackish wetland sediments. Geochim. Cosmochim. Acta 115:15–30. https://doi.org/10.1016/j.gca.2013.03.029
Adam P (2011) The energetics of mangrove forests. Austral Ecology 36:E18–E19. https://doi.org/10.1111/j.1442-9993.2010.02190.x
Duke NC, Ball MC, Ellison JC (1998) Factors influencing biodiversity and distributional gradients in mangroves. Glob. Ecol. Biogeogr. Lett. 7:27–47. https://doi.org/10.2307/2997695
Alongi DM, Pfitzner J, Trott LA, Tirendi F, Dixon P, Klumpp DW (2005) Rapid sediment accumulation and microbial mineralization in forests of the mangrove Kandelia candel in the Jiulongjiang estuary, China. Estuar. Coast. Shelf Sci. 63:605–618. https://doi.org/10.1016/j.ecss.2005.01.004
Chauhan R, Datta A, Ramanathan AL, Adhya TK (2015) Factors influencing spatio-temporal variation of methane and nitrous oxide emission from a tropical mangrove of eastern coast of India. Atmos. Environ. 107:95–106. https://doi.org/10.1016/j.atmosenv.2015.02.006
Wang H, Liao G, D’Souza M, Yu X, Yang J, Yang X, Zheng T (2015) Temporal and spatial variations of greenhouse gas fluxes from a tidal mangrove wetland in Southeast China. Environ. Sci. Pollut. Res. 23:1873–1885. https://doi.org/10.1007/s11356-015-5440-4
Gomes NCM, Cleary DFR, Pinto FN, Egas C, Almeida A, Cunha A, Mendonca-Hagler LCS, Smalla K (2010) Taking root: enduring effect of rhizosphere bacterial colonization in mangroves. PLoS One 5. https://doi.org/10.1371/journal.pone.0014065
Daebeler A, Bodelier PLE, Yan Z, Hefting MM, Jia Z, Laanbroek HJ (2014) Interactions between Thaumarchaea, Nitrospira and methanotrophs modulate autotrophic nitrification in volcanic grassland soil. The ISME Journal 8:2397–2410. https://doi.org/10.1038/ismej.2014.81
Jia ZJ, Cai ZC, Xu H, Li XP (2001) Effect of rice plants on CH4 production, transport, oxidation and emission in rice paddy soil. Plant Soil 230:211–221. https://doi.org/10.1023/a:1010366631538
Zheng Y, Huang R, Wang BZ, Bodelier PLE, Jia ZJ (2014) Competitive interactions between methane- and ammonia-oxidizing bacteria modulate carbon and nitrogen cycling in paddy soil. Biogeosciences 11:3353–3368. https://doi.org/10.5194/bg-11-3353-2014
He R, Wooller MJ, Pohlman JW, Catranis C, Quensen J, Tiedje JM, Leigh MB (2012) Identification of functionally active aerobic methanotrophs in sediments from an arctic lake using stable isotope probing. Environ. Microbiol. 14:1403–1419. https://doi.org/10.1111/j.1462-2920.2012.02725.x
Ho A, Angel R, Veraart AJ, Daebeler A, Jia Z, Kim SY, Kerckhof F-M, Boon N, Bodelier PLE (2016) Biotic interactions in microbial communities as modulators of biogeochemical processes: methanotrophy as a model system. Front. Microbiol. 7. https://doi.org/10.3389/fmicb.2016.01285
Sharp CE, Martínez-Lorenzo A, Brady AL, Grasby SE, Dunfield PF (2014) Methanotrophic bacteria in warm geothermal spring sediments identified using stable-isotope probing. FEMS Microbiol. Ecol. 90:92–102. https://doi.org/10.1111/1574-6941.12375
Cai Y, Zheng Y, Bodelier PLE, Conrad R, Jia Z (2016) Conventional methanotrophs are responsible for atmospheric methane oxidation in paddy soils. Nat. Commun. 7. https://doi.org/10.1038/ncomms11728
WRA (2005) Investigation of current status in Tanshui River system, Final Report. Water Resources Agency, Taiwan,
Chiu CY, Chou CH (1991) The distribution and influence of heavy-metals in mangrove forests of the Tamsui estuary in Taiwan. Soil Science and Plant Nutrition 37:659–669
Shiau Y, Dham V, Tian G, Chiu CY (2016) Factors influencing removal of sewage nitrogen through denitrification in mangrove soils. Wetlands 36:621–630. https://doi.org/10.1007/s13157-016-0770-7
Shiau Y-J, Lin M-F, Tan C-C, Tian G, Chiu C-Y (2017) Assessing N2 fixation in estuarine mangrove soils. Estuar. Coast. Shelf Sci. 189:84–89. https://doi.org/10.1016/j.ecss.2017.03.005
Lu L, Jia ZJ (2013) Urease gene-containing Archaea dominate autotrophic ammonia oxidation in two acid soils. Environ. Microbiol. 15:1795–1809. https://doi.org/10.1111/1462-2920.12071
Daebeler A, Abell GCJ, Bodelier PLE, Bodrossy L, Frampton DMF, Hefting MM, Laanbroek HJ (2012) Archaeal dominated ammonia-oxidizing communities in Icelandic grassland soils are moderately affected by long-term N fertilization and geothermal heating. Front. Microbiol. 3:352. https://doi.org/10.3389/fmicb.2012.00352
Caporaso JG, Kuczynski J, Stombaugh J, Bittinger K, Bushman FD, Costello EK, Fierer N, Pena AG, Goodrich JK, Gordon JI, Huttley GA, Kelley ST, Knights D, Koenig JE, Ley RE, Lozupone CA, McDonald D, Muegge BD, Pirrung M, Reeder J, Sevinsky JR, Turnbaugh PJ, Walters WA, Widmann J, Yatsunenko T, Zaneveld J, Knight R (2010) QIIME allows analysis of high-throughput community sequencing data. Nat. Methods 7:335–336. https://doi.org/10.1038/nmeth.f.303
Kozich J, Westcott S, Baxter N, Highlander S, Schloss P (2013) Development of a dual-index sequencing strategy and curation pipeline for analyzing amplicon sequence data on the MiSeq Illumina sequencing platform. Appl. Environ. Microbiol. 79:5112–5120
Dumont MG, Luke C, Deng Y, Frenzel P (2014) Classification of pmoA amplicon pyrosequences using BLAST and the lowest common ancestor method in MEGAN. Front. Microbiol. 5:34. https://doi.org/10.3389/fmicb.2014.00034
Caporaso JG, Lauber CL, Walters WA, Berg-Lyons D, Huntley J, Fierer N, Owens SM, Betley J, Fraser L, Bauer M, Gormley N, Gilbert JA, Smith G, Knight R (2012) Ultra-high-throughput microbial community analysis on the Illumina HiSeq and MiSeq platforms. The ISME Journal 6:1621–1624. https://doi.org/10.1038/ismej.2012.8
Wang Q, Garrity GM, Tiedje JM, Cole JR (2007) Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl. Environ. Microbiol. 73:5261–5267. https://doi.org/10.1128/aem.00062-07
Degelmann DM, Borken W, Drake HL, Kolb S (2010) Different atmospheric methane-oxidizing communities in European beech and Norway Spruce soils. Appl. Environ. Microbiol. 76:3228–3235. https://doi.org/10.1128/aem.02730-09
Hirayama H, Fuse H, Abe M, Miyazaki M, Nakamura T, Nunoura T, Furushima Y, Yamamoto H, Takai K (2013) Methylomarinum vadi gen. nov., sp nov., a methanotroph isolated from two distinct marine environments. Int. J. Syst. Evol. Microbiol. 63:1073–1082. https://doi.org/10.1099/ijs.0.040568-0
Cussler EL (1997) Diffusion: Mass transfer in fluid systems. Cambridge University Press, Cambridge
Cowie M, Watts H (1970) Diffusion of methane and chloromethanes in air. Can. J. Chem. 49:74–77
Hudson BD (1994) Soil organic-matter and available water capacity. J. Soil Water Conserv. 49:189–194
Manns HR, Berg AA (2014) Importance of soil organic carbon on surface soil water content variability among agricultural fields. J. Hydrol. 516:297–303. https://doi.org/10.1016/j.jhydrol.2013.11.018
Zhou Z, Chen J, Cao H, Han P, Gu J-D (2015) Analysis of methane-producing and metabolizing archaeal and bacterial communities in sediments of the northern South China Sea and coastal Mai Po Nature Reserve revealed by PCR amplification of mcrA and pmoA genes. Front. Microbiol. 5:789. https://doi.org/10.3389/fmicb.2014.00789
Yun J, Yu Z, Li K, Zhang H (2013) Diversity, abundance and vertical distribution of methane-oxidizing bacteria (methanotrophs) in the sediments of the Xianghai wetland, Songnen Plain, northeast China. J. Soils Sediments 13:242–252. https://doi.org/10.1007/s11368-012-0610-1
Yun J, Zhang H, Deng Y, Wang Y (2015) Aerobic methanotroph diversity in Sanjiang wetland, northeast China. Microb. Ecol. 69:567–576. https://doi.org/10.1007/s00248-014-0506-2
Henckel T, Jackel U, Conrad R (2001) Vertical distribution of the methanotrophic community after drainage of rice field soil. FEMS Microbiol. Ecol. 34:279–291. https://doi.org/10.1111/j.1574-6941.2001.tb00778.x
Lyle M (1983) The brown-green color transition in marine sediments: a marker of the Fe(III)-Fe(II) redox boundary. Limnol. Oceanogr. 28:1026–1033. https://doi.org/10.4319/lo.1983.28.5.1026
Chester R (1990) Marine geochemistry. Unwin Hyman Ltd., London,
Graham DW, Chaudhary JA, Hanson RS, Arnold RG (1993) Factors affecting competition between type-I and type-II methanotrophs in 2-organism, continuous-flow reactors. Microb. Ecol. 25:1–17
Bartlett KB, Bartlett DS, Harriss RC, Sebacher DI (1987) Methane emissions along a salt-marsh salinity gradient. Biogeochemistry 4:183–202
Krauss KW, Whitbeck JL (2012) Soil greenhouse gas fluxes during wetland forest retreat along the Lower Savannah River, Georgia (USA). Wetlands 32:73–81
Poffenbarger HJ, Needelman BA, Megonigal JP (2011) Salinity influence on methane emissions from tidal marshes. Wetlands 31:831–842
Liu H, Wu X, Li Z, Wang Q, Liu D, Liu G (2017) Responses of soil methanogens, methanotrophs, and methane fluxes to land-use conversion and fertilization in a hilly red soil region of southern China. Environ. Sci. Pollut. Res. 24:8731–8743. https://doi.org/10.1007/s11356-017-8628-y
Wagner-Riddle C, Thurtell GW, Kidd GE, Edwards GC, Simpson IJ (1996) Micrometeorological measurements of trace gas fluxes from agricultural and natural ecosystems. Infrared Phys. Technol. 37:51–58. https://doi.org/10.1016/1350-4495(95)00111-5
Stadmark J, Leonardson L (2005) Emissions of greenhouse gases from ponds constructed for nitrogen removal. Ecol. Eng. 25:542–551
Ho A, Luke C, Cao Z, Frenzel P (2011) Ageing well: methane oxidation and methane oxidizing bacteria along a chronosequence of 2000 years. Environ. Microbiol. Rep. 3:738–743. https://doi.org/10.1111/j.1758-2229.2011.00292.x
Krause S, Lüke C, Frenzel P (2012) Methane source strength and energy flow shape methanotrophic communities in oxygen-methane counter-gradients. Environ. Microbiol. Rep. 4:203–208. https://doi.org/10.1111/j.1758-2229.2011.00322.x
Zheng Y, Jia Z (2016) The application of biomarker genes for DNA/RNA-stable isotope probing of active methanotrophs responsible for aerobic methane oxidation in six paddy soils. Acta Pedol. Sin. 53:490–501. 10.11766/trxb201509020090
Ho A, Kerckhof F-M, Luke C, Reim A, Krause S, Boon N, Bodelier PLE (2013) Conceptualizing functional traits and ecological characteristics of methane-oxidizing bacteria as life strategies. Environ. Microbiol. Rep. 5:335–345. https://doi.org/10.1111/j.1758-2229.2012.00370.x
Shrestha M, Shrestha PM, Frenzel P, Conrad R (2010) Effect of nitrogen fertilization on methane oxidation, abundance, community structure, and gene expression of methanotrophs in the rice rhizosphere. The ISME Journal 4:1545–1556. https://doi.org/10.1038/ismej.2010.89
Steenbergh AK, Meima MM, Kamst M, Bodelier PLE (2010) Biphasic kinetics of a methanotrophic community is a combination of growth and increased activity per cell. FEMS Microbiol. Ecol. 71:12–22. https://doi.org/10.1111/j.1574-6941.2009.00782.x
Kalyuzhnaya MG (2016) Methylosarcina. In: Whitman WB (ed) Bergey's Manual of Systematics of Archaea and Bacteria. John Wiley & Sons, Inc., New Jersey. https://doi.org/10.1002/9781118960608.gbm01184.pub2
Bowman JP (2015) Methylobacter. In: Whitman WB (ed) Bergey’s Manual of Systematics of Archaea and Bacteria. John Wiley & Sons, Inc., New Jersey. https://doi.org/10.1002/9781118960608.gbm01179.pub2
Bowman JP (2015) Methylomonas. In: Whitman, WB (ed) Bergey’s Manual of Systematics of Archaea and Bacteria. John Wiley & Sons, Inc., New Jersey. https://doi.org/10.1002/9781118960608.gbm01183.pub2
Caldwell SL, Laidler JR, Brewer EA, Eberly JO, Sandborgh SC, Colwell FS (2008) Anaerobic oxidation of methane: mechanisms, bioenergetics, and theecology of associated microorganisms. Environ. Sci. Technol. 42:6791–6799
Knittel K, Boetius A (2009) Anaerobic oxidation of methane: progress with an unknown process. Annu. Rev. Microbiol. 63:311–334
Acknowledgements
We thank Ms. Yu-Shiuan Huang from the Biodiversity Research Center, Academia Sinica, Taipei, Taiwan, and Ms. Dongmei Wang from the Nanjing Soil Research Institute, Chinese Academy of Science, Nanjing, PRC, for the laboratory work.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Data Availability
All of the pmoA and 16S rRNA gene sequences obtained from Miseq sequencing have been deposited in NCBI with accession codes SRP111433.
Rights and permissions
About this article
Cite this article
Shiau, YJ., Cai, Y., Lin, YT. et al. Community Structure of Active Aerobic Methanotrophs in Red Mangrove (Kandelia obovata) Soils Under Different Frequency of Tides. Microb Ecol 75, 761–770 (2018). https://doi.org/10.1007/s00248-017-1080-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-017-1080-1