Abstract
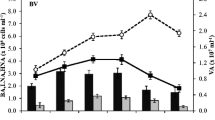
The effects of viral lysis and heterotrophic nanoflagellate grazing (top down forces) on prokaryotic mortality and their subsequent impact on their metabolism were estimated in the upper euphotic and deeper aphotic depth of 11 freshwater lakes located in the French Massif Central. The standing stocks of viruses (VA) and heterotrophic nanoflagellate (HNF) varied significantly (p < 0.05) with sampled depth. VA was substantially (twofold on an average) and significantly higher (p < 0.03) at the aphotic compared to euphotic depth, whereas the reverse was true (p < 0.02) for HNF. Among the prokaryote subgroup, high nucleic acid content prokaryotes explained for significant variability in the total VA and served as principle host target for viral proliferation. Like standing stocks, flagellate grazing and viral infection rates also followed similar patterns. In the investigated lakes, the mechanism for regulating prokaryotic production varied with sampled depth from grazing control in the euphotic to control due to viral lysis in the aphotic. We also tested the hypothesis of top down control on prokaryotic growth efficiency (PGE, which we used as an index of prokaryotic physiological and energetic status at the community level) at both depths. Overall, among the studied lakes, PGE varied widely (4–51 %) with significantly (p < 0.05) lower values in the aphotic (mean = 18 ± 4 %) than euphotic depth (mean = 32 ± 9 %). Contrasting observations on the top down control of PGE between sampled depths were observed. The presence of grazers was found to stimulate PGE at the euphotic, whereas viruses through their lytic infection had a strong negative impact on PGE at the aphotic depth. Such observed differences in PGE and the mechanism controlling prokaryotic production with depth could eventually have strong implication on carbon and nutrient flux patterns in the studied lakes.


Similar content being viewed by others
References
Biddanda BA, Ogdahl ML, Cotner JB (2001) Dominance of bacterial metabolism in oligotrophic relative to eutrophic waters. Limnol Oceanogr 46:730–739
Binder B (1999) Reconsidering the relationship between viral-induced bacterial mortality and frequency of infected cells. Aquat Microb Ecol 18:207–215
Bonilla-Findji O, Herndl G, Gattuso JP, Weinbauer MG (2009) Viral and flagellate control of prokaryotic production and community structure in offshore Mediterranean waters. Appl Environ Microbiol 75:4801–4812
Brussaard CPD (2004) Optimization of procedures for counting viruses by flow cytometry. Appl Environ Microbiol 70:1506–1513
Carignan R, Planas D, Vis C (2000) Planktonic production and respiration in oligotrophic Shield Lakes. Limnol Oceanogr 45:189–199
Caron DA (1983) Techniques for enumeration of heterotrophic and phototrophic nanoplankton using epifluorescent microscopy and comparison with other procedures. Appl Environ Microbiol 46:491–498
Carrias JF, Amblard C, Bourdier G (1996) Protistan bacterivory in an oligomesotrophic lake: importance of attached ciliates and flagellates. Microb Ecol 31:249–268
Colombet J, Sime-Ngando T, Cauchie HM, Fonty G, Hoffmann L, Demeure G (2006) Depth-related gradients of viral activity in Lake Pavin. Appl Environ Microbiol 72:4440–4445
del Giorgio PA, Cole JJ (1998) Bacterial growth efficiency in natural aquatic ecosystems. Annu Rev Ecol Syst 29:503–541
Fuhrman JA (1999) Marine viruses and their biogeochemical and ecological effects. Nature 399:541–548
Hewson J, Chow C, Fuhrman JA (2010) Ecological role of viruses in aquatic ecosystems. eLS. John Wiley and Sons, NY, USA. doi:10.1002/9780470015902.a0022546
Jansson M, Persson L, De Roos AM, Jones R, Tranvik LJ (2007) Terrestrial carbon and intraspecific size variation shape lake ecosystems. Trends Ecol Evol 22:316–322
Kritzberg ES, Cole JJ, Pace MM, Graneli W (2005) Does autochthonous primary production drive variability in bacterial metabolism and growth efficiency in lakes dominated by terrestrial inputs? Aquat Microb Ecol 38:103–111
Landry MR, Kirshtein J, Constantinou J (1995) A refined dilution technique for measuring the community grazing impact of microzooplankton, with experimental tests in the central equatorial Pacific. Mar Ecol Prog Ser 120:55–63
Lee S, Fuhrman JA (1987) Relationships between biovolume and biomass of naturally derived marine bacterioplankton. Appl Environ Microbiol 53:1298–1303
Lønborg C, Søndergaard M (2009) Microbial availability and degradation of dissolved organic carbon and nitrogen in two coastal areas. Estuar Coast Shelf Sci 81:513–520
Mahaffey C, Benitez-Nelson CR, Bidigare RR, Rii Y, Karl DM (2008) Nitrogen dynamics within a wind-driven eddy. Deep Sea Res II 55:1398–1411
Marie D, Brussaard C, Bratbak G, Vaulot D (1999) Enumeration of marine viruses in culture and natural samples by flow cytometry. Appl Environ Microbiol 65:45–52
Maurice C, Bouvier T, Comte J, Guillemette F, del Giorgio PA (2010) Seasonal variations of phage life strategies and bacterial physiological states in three northern temperate lakes. Environ Microbiol 12:628–641
Motegi C, Nagata T, Miki T, Weinbauer MG, Legendre L, Rassoulzadegan F (2009) Viral control of bacterial growth efficiency in marine pelagic environments. Limnol Oceanogr 54:1901–1910
Paul JH, Jiang S (2001) Lysogeny and transduction. In: Paul JH (ed) Methods in microbiology, vol 30. Academic, San Diego, pp 105–125
Pradeep Ram AS, Nishimura Y, TomaruY NK, Nagata T (2010) Seasonal variation in viral-induced mortality of bacterioplankton in the water column of a large mesotrophic lake (Lake Biwa, Japan). Aquat Microb Ecol 58:248–259
Pradeep Ram AS, Boucher D, Sime-Ngando T, Debroas D, Romagoux JC (2005) Phage bacteriolysis, protistan bacterivory potential, and bacterial production in a freshwater reservoir: coupling with temperature. Microb Ecol 50:64–72
Pradeep Ram AS, Sime-Ngando T (2008) Functional responses of prokaryotes and viruses to grazer effects and nutrient additions in freshwater microcosms. ISME J 2:498–509
Roland F, Cole JJ (1999) Regulation of bacterial growth efficiency in a large turbid estuary. Aquat Microb Ecol 20:31–38
SCOR-UNESCO (1966) Determination of photosynthetic pigments in sea water. Unesco, Paris
Sime-Ngando T, Mignot JP, Amblard C, Bourdier G, Devilettes C, Quilblier-Lloberas C (1996) Characterization of planktonic virus-like particles in a French mountain lake: methodological aspects and preliminary results. Ann Limnol 32:1–5
Smith EM, Prairie YT (2004) Bacterial metabolism and growth efficiency in lakes: the importance of phosphorus availability. Limnol Oceanogr 49:137–147
Suttle CA (2007) Marine viruses—major players in the global ecosystem. Nat Rev Microbiol 5:801–812
Thouvenot A, Richardot M, Debroas D, Devaux J (1999) Bacterivory of metazooplankton, ciliates, and flagellates in a newly flooded reservoir. J Plankton Res 21:1659–1679
Vrede K, Stensdotter U, Lindstrom ES (2003) Viral and bacterioplankton dynamics in two lakes with different humic contents. Microb Ecol 46:406–415
Weinbauer MG (2004) Ecology of prokaryotic viruses. FEMS Microbiol Rev 28:127–181
Weinbauer MG, Höfle MG (1998) Significance of viral lysis and flagellate grazing as factors controlling bacterioplankton production in a eutrophic lake. Appl Environ Microbiol 64:431–438
Weinbauer MG, Winter C, Höfle MG (2002) Reconsidering transmission electron microscopy-based estimates of viral infection of bacterioplankton using conversion factors derived from natural communities. Aquat Microb Ecol 27:103–110
Wetzel RG, Likens GE (1995) Limnological analysis, 2nd edn. Springer, New York
Wilhelm SW, Suttle CA (1999) Viruses and nutrient cycles in the sea. Bioscience 49:781–788
You L, Suthers PF, Yin J (2002) Effects of Escherichia coli physiology on growth of phage T7 in vivo and in silico. J Bacteriol 184:1888–1894
Zweifel UL, Norrman B, Hagström A (1993) Consumption of dissolved organic carbon by marine bacteria and demand for inorganic nutrients. Mar Ecol Prog Ser 101:23–32
Acknowledgments
SP was supported by PhD fellowship from the French Ministry of Research and Technology. We thank Dr. Jonathan Cole (Cary Institute of Ecosystem Studies, New York, USA) for his valuable discussion and constructive comments. We appreciate valuable comments and suggestions from two reviewers.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 1913 kb)
Rights and permissions
About this article
Cite this article
Ram, A.S.P., Palesse, S., Colombet, J. et al. Variable Viral and Grazer Control of Prokaryotic Growth Efficiency in Temperate Freshwater Lakes (French Massif Central). Microb Ecol 66, 906–916 (2013). https://doi.org/10.1007/s00248-013-0289-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-013-0289-x