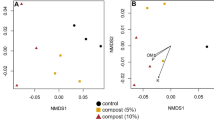
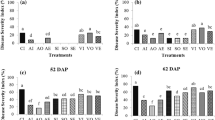
Abstract
Compost has been widely used in order to promote vegetation growth in post-harvested and burned soils. The effects on soil microorganisms were scarcely known, so we performed the microbial analyses in a wildfire area of the Taebaek Mountains, Korea, during field surveys from May to September 2007. Using culture-dependent and -independent methods, we found that compost used in burned soils influenced a greater impact on soil fungi than bacteria. Compost-treated soils contained higher levels of antifungal strains in the genera Bacillus and Burkholderia than non-treated soils. When the antifungal activity of Burkholderia sp. strain O1a_RA002, which had been isolated from a compost-treated soil, was tested for the growth inhibition of bacteria and fungi isolated from burned soils, the membrane-filtered culture supernatant inhibited 19/37 fungal strains including soil fungi, Eupenicillium spp. and Devriesia americana; plant pathogens, Polyschema larviformis and Massaria platani; an animal pathogen, Mortierella verticillata; and an unidentified Ascomycota. However, this organism only inhibited 11/151 bacterial strains tested. These patterns were compatible with the culture-independent DGGE results, suggesting that the compost used in burned soils had a greater impact on soil fungi than bacteria through the promotion of the growth of antifungal bacteria. Our findings indicate that compost used in burned soils is effective in restoring soil conditions to a state closer to those of nearby unburned forest soils at the early stage of secondary succession.




Similar content being viewed by others
Abbreviations
- DGGE:
-
Denaturing gradient gel electrophoresis
- C/N:
-
Ratio of carbon to nitrogen
- OM:
-
Organic matter
- EC:
-
Electrical conductivity
- ApCFU:
-
Approximate colony-forming unit
- PCR:
-
Polymerase chain reaction
- OD:
-
Optical density
- TS:
-
Tryptic soy
- CS:
-
Culture supernatant
- FAME:
-
Fatty acid methyl ester
References
Amaranthus MP, Trappe JM (1993) Effects of erosion on ecto- and VA-mycorrhizal inoculums potential of soil following forest fire in southwest Oregon. Plant Soil 150:41–49
Anderson IC, Campbell CD, Prosser JI (2003) Diversity of fungi in organic soils under a moorland—Scots pine (Pinus sylvestris L.) gradient. Environ Microbiol 5:1121–1132
APHA (2005) Standard methods for the examination of water and wastewater, 21st edn. American Public Health Association, Washington
Bevivino A, Peggion V, Chiarini L, Tabacchioni S, Cantale C, Dalmastri C (2005) Effect of Fusarium verticillioides on maize-root-associated Burkholderia cenocepacia populations. Res Microbiol 156:974–983
Bevivino A, Sarrocco S, Dalmastri C, Tabacchioni S, Cantale C, Chiarini L (1998) Characterization of a free-living maize-rhizosphere population of Burkholderia cepacia: effect of seed treatment on disease suppression and growth promotion of maize. FEMS Microbiol Ecol 27:225–237
Borneman J, Hartin RJ (2000) PCR primers that amplify fungal rRNA genes from environmental samples. Appl Environ Microbiol 66:4356–4360
Bowman DMJS, Balch JK, Artaxo P et al (2009) Fire in the Earth system. Science 324:481–484
Braun-Blanquet J (1932) Plant sociology: the study of plant communities. Fuller GD, Conard HS (transl.), McGraw-Hill, New York
Chiarini L, Bevivino A, Tabacchioni S, Dalmastri C (1998) Inoculation of Burkholderia cepacia, Pseudomonas fluorescens and Enterobacter sp. on Sorghum bicolor: Root colonization and plant growth promotion of dual strain inocula. Soil Biol Biochem 30:81–87
Choi SD, Chang YS, Park BK (2006) Increase in carbon emissions from forest fires after intensive reforestation and forest management programs. Sci Total Environ 372:225–235
Choromanska U, DeLuca TH (2002) Microbial activity and nitrogen mineralization in forest mineral soils following heating: evaluation of post-fire effects. Soil Biol Biochem 34:263–271
Crous PW, Braun U, Schubert K, Groenewald JZ (2007) Delimiting Cladosporium from morphologically similar genera. Stud Mycol 58:33–56
D’Acqui LP, Santi CA, Maselli F (2007) Use of ecosystem information to improve soil organic carbon mapping of a Mediterranean island. J Environ Qual 36:262–271
de Gruyter J, Aveskamp MM, Woudenberg JH, Verkley GJ, Groenewald JZ, Crous PW (2009) Molecular phylogeny of Phoma and allied anamorph genera: towards a reclassification of the Phoma complex. Mycol Res 113:508–519
Fernandez FA, Miller AN, Huhndorf SM, Lutzoni FM, Zoller S (2006) Systematics of the genus Chaetosphaeria and its allied genera: morphological and phylogenetic diversity in north temperate and neotropical taxa. Mycologia 98:121–130
Gardes M, Bruns TD (1993) ITS primers with enhanced specificity for basidiomycetes—application to the identification of mycorrhizae and rusts. Mol Ecol 2:113–118
Geiser DM, Gueidan C, Miadlikowska J, Lutzoni F, Kauff F, Hofstetter V, Fraker E, Schoch CL, Tibell L, Untereiner WA, Aptroot A (2006) Eurotiomycetes: Eurotiomycetidae and Chaetothyriomycetidae. Mycologia 98:1053–1064
Guerrero C, Gómez I, Moral R, Mataix-Solera J, Mataix-Beneyto J, Hernández T (2001) Reclamation of a burned forest soil with municipal waste compost: macronutrient dynamic and improved vegetation cover recovery. Biores Technol 76:221–227
Hart SC, DeLuca TH, Newman GS, MacKenzie MD, Boyle SI (2005) Post-fire vegetative dynamics as drivers of microbial community structure and function in forest soils. For Ecol Manage 220:166–184
Hayes VM, Wu Y, Osinga J, Mulder IM, van der Vlies P, Elfferich P, Buys CHCM, Hofstra RMW (1999) Improvements in gel composition and electrophoretic conditions for broad-range mutation analysis by denaturing gradient gel electrophoresis. Nucleic Acids Res 27:e29
Heydari A, Misaghi IJ (2004) Biocontrol activity of Burkholderia cepacia against Rhizoctonia solani in herbicide-treated soils. Plant Soil 202:109–116
Hicks RE, Amann RI, Stahl DA (1992) Dual staining of natural bacterioplankton with 4′,6-diamidino-2-phenylindole and fluorescent oligonucleotide probes targeting kingdom-level 16S rRNA sequences. Appl Environ Microbiol 58:2158–2163
Hopkins DW, MacNaughton SJ, O’Donnell AG (1991) A dispersion and differential centrifugation technique for representatively sampling microorganisms from soil. Soil Biol Biochem 23:217–225
James TY, Letcher PM, Longcore JE, Mozley-Standridge SE, Porter D, Powell MJ, Griffith GW, Vilgalys R (2006) A molecular phylogeny of the flagellated fungi (Chytridiomycota) and description of a new phylum (Blastocladiomycota). Mycologia 98:860–871
Jonsson L, Dalhberg A, Nilsson MC, Zackrisson O, Kårén O (1999) Ectomycorrhizal fungal communities in late-successional boreal forests, and their composition following wildfire. Mol Ecol 8:205–215
Kim KD, Lee EJ (2005) Potential tree species for use in the restoration of unsanitary landfills. Environ Manage 36:1–14
Klopatek CC, DeBano LF, Klopatek JM (1988) Effects of simulated fire on vesicular–arbuscular mycorrhizae in pinyon–juniper woodland soil. Plant Soil 109:245–249
Korb JE, Johnson NC, Covington WW (2003) Arbuscular mycorrhizal propagule densities respond rapidly to ponderosa pine restoration treatments. J Appl Ecol 40:101–110
Maldonado LA, Stach JEM, Pathom-aree W, Ward AC, Bull AT, Goodfellow M (2005) Diversity of cultivable actinobacteria in geographically widespread marine sediments. Antonie van Leeuwenhoek 87:11–18
Mazzola M (2004) Assessment and management of soil microbial community structure for disease suppression. Annu Rev Phytopathol 42:35–59
Morris SE, Moses TA (1987) Forest fire and the natural soil erosion regime in the Colorado Front Range. Annal Assoc Am Geogr 77:245–254
Muyzer G, De Waal EC, Uitterlinden AG (1993) Profiling of complex microbial populations by denaturing gradient gel electrophoresis analysis of polymerase chain reaction-amplified genes coding for 16S rRNA. Appl Environ Microbiol 59:695–700
Neary DG, Klopatek CC, DeBano LF, Ffolliott PF (1999) Fire effects on belowground sustainability: a review and synthesis. For Ecol Manage 122:51–71
Neufeld JD, Mohn WW (2005) Unexpectedly high bacterial diversity in Arctic tundra relative to boreal forest soils, revealed by serial analysis of ribosomal sequence Tags. Appl Environ Microbiol 71:5710–5718
Ojima DS, Schimel DS, Parton WJ, Owensby CE (1994) Long- and short-term effects of fire on nitrogen cycling in tallgrass prairie. Biogeochemistry 24:67–84
Packer A, Clay K (2000) Soil pathogens and spatial patterns of seedling mortality in a temperate tree. Nature 404:278–281
Pardini G, Gispert M, Dunjó G (2004) Relative influence of wildfire on soil properties and erosion processes in different Mediterranean environments in NE Spain. Sci Total Environ 328:237–246
Pare D, Van Cleve K (1993) Soil nutrient availability and relationships with aboveground biomass production on postharvested upland white spruce sites in interior Alaska. Can J Forest Res 23:1223–1232
Quan CS, Zheng W, Liu Q, Ohta Y, Fan SD (2006) Isolation and characterization of a novel Burkholderia cepacia with strong antifungal activity against Rhizoctonia solani. Appl Microbiol Biotechnol 72:1276–1284
Rodríguez H, Fraga R (1999) Phosphate solubilizing bacteria and their role in plant growth promotion. Biotechnol Adv 17:319–339
Sandhu GS, Kline BC, Stockman L, Roberts GD (1995) Molecular probes for the diagnosis of fungal infections. J Clin Microbiol 33:2913–2919
Shear CL, Davidson RW (1936) The life histories of Botryosphaeria melanops and Massaria platani. Mycologia 28:476–482
Shenoy BD, Jeewon R, Wang H, Amandeep K, Ho WH, Bhat DJ, Crous PW, Hyde KD (2010) Sequence data reveals phylogenetic affinities of fungal anamorphs Bahusutrabeeja, Diplococcium, Natarajania, Paliphora, Polyschema, Rattania and Spadicoides. Fungal Divers 44:161–169
Sigler WV, Turco RF (2002) The impact of chlorothalonil application on soil bacterial and fungal populations as assessed by denaturing gradient gel electrophoresis. Appl Soil Ecol 21:107–118
Smith NR, Kishchuk BE, Mohn WW (2008) Effects of wildfire and harvest disturbances on forest soil bacterial communities. Appl Environ Microbiol 74:216–224
Sparks RL (1996) Methods of soil analysis. Part 3. Chemical methods. Soil Science Society of America book series, no. 5. The Soil Science Society of America, Madison
Vaganov EA, Efremov SP, Onuchin AA (2006) International approaches to reduce anthropogenic greenhouse gas emissions. In: Lombardi S, Altunina LK, Beaubien SE (eds) Advances in the geological storage of carbon dioxide, NATO science series—IV, Earth and environmental sciences, vol 65. Springer, Netherlands, pp 17–34
Vilarino A, Arines J (1991) Numbers and viability of vesicular–arbuscular fungal propagules in field soil samples after a wildfire. Soil Biol Biochem 23:1083–1087
Vilgalys R, Hester M (1990) Rapid genetic identification and mapping of enzymatically amplified ribosomal DNA from several Cryptococcus species. J Bacteriol 172:4238–4246
Villar MC, Petrikova V, Díaz-Raviña M, Carballas T (2004) Recycling of organic wastes in burnt soils: combined application of poultry manure and plant cultivation. Waste Manage 24:365–370
Warnecke F, Amann R, Pernthaler J (2004) Actinobacterial 16S rRNA genes from freshwater habitats cluster in four distinct lineages. Environ Microbiol 6:242–253
Watanabe K, Kodama Y, Harayama S (2001) Design and evaluation of PCR primers to amplify bacterial 16S ribosomal DNA fragments used for community fingerprinting. J Microbiol Methods 44:253–262
Wu SH, Hibbett DS, Binder M (2001) Phylogenetic analyses of Aleurodiscus s.l. and allied genera. Mycologia 93:720–731
Yeager CM, Northup DE, Grow CC, Barns SM, Kuske CR (2005) Changes in nitrogen-fixing and ammonia-oxidizing bacterial communities in soil of a mixed conifer forest after wildfire. Appl Environ Microbiol 71:2713–2722
Zheng D, Alm EW, Stahl DA, Raskin L (1996) Characterization of universal small-subunit rRNA hybridization probes for quantitative molecular microbial ecology studies. Appl Environ Microbiol 62:4504–4545
Acknowledgements
We express thanks to Young-Gun Zo (Kyungsung University, Pusan, Republic of Korea) for the cluster analysis of DGGE gels. This study was partly supported by a research project (FE0500-1997-01) of Korea Forest Research Institute.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Table S1
Estimation of approximate colony-forming unit (ApCFU, cfu g−1 dry weight soil), specific growth rate constant (k, d−1), and a time constant (t ½, d) for the half-maximum colony count of cultivable microorganisms in burned soils and nearby unburned forest soils (PDF 76.0 kb)
Table S2
API-20E, API-20NE, API-ZYM, and API-Coryne test results for numerical characterization for the selected antifungal bacteria. All negative results are excluded (PDF 99.7 kb)
Table S3
Average percentages of FAME analyses for the selected antifungal bacteria (PDF 100 kb)
Rights and permissions
About this article
Cite this article
Kim, YH., Kim, I.S., Moon, E.Y. et al. High Abundance and Role of Antifungal Bacteria in Compost-Treated Soils in a Wildfire Area. Microb Ecol 62, 725–737 (2011). https://doi.org/10.1007/s00248-011-9839-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-011-9839-2