Abstract
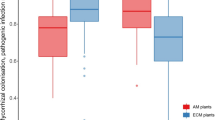
Fungal diversity in the rhizosphere of healthy and diseased clonal black spruce (Picea mariana) plants was analyzed with regard to nursery production chronosequences. The four key production stages were sampled: mother plants (MP), 8-week-old cuttings (B + 0), second-year cuttings (B + 1), and third-year cuttings (B + 2). A total of 45 fungal taxa were isolated and identified based on cultural, phenotypic, and molecular characters. Members of phylum Ascomycota dominated, followed by Basidiomycota and Zygomycota. Diagnosis characters and distance analysis of the internal transcribed spacer rDNA sequences allowed the identification of 39 ascomycetous taxa. Many belong to the order Hypocreales, families Hypocreaceae and Nectriaceae, which contain many clusters of potentially pathogenic taxa (Cylindrocladium, Fusarium, and Neonectria) and are also ecologically associated with antagonistic taxa (Chaetomium, Hypocrea, Microsphaeropsis, Penicillium, Paecilomyces, Verticillium, Trichoderma, and Sporothrix). This is also the first report of a Cylindrocladium canadense association with disease symptoms and relation with Pestalotiopsis, Fusarium, Exserochilum, Rhizoctonia, and Xenochalara fungal consortia. Both production chronosequence and plant health considerably influenced fungal taxa assemblages. Unweighted pair-group arithmetic average clustering showed that isolates from MP, B + 0, and B + 1 plant rhizospheres clustered together within healthy or diseased health classes, whereas isolates from healthy and diseased B + 2 plants clustered together. Canonical correspondence analysis revealed substantial alteration in community assemblages with regard to plant health and yielded a principal axis direction that regrouped taxa associated with diseased plant rhizosphere soil, whereas the opposite axis direction was associated with healthy plants. Two diversity indices were defined and applied to assess the fungal taxa contribution (Tc) and persistence (Pi) throughout the production.





Similar content being viewed by others
References
Anderson, IC, Campbell, CD, Prosser, JI (2003) Diversity of fungi in organic soils under a moorland—Scots pine (Pinus sylvestris L.) gradient. Environ Microbiol 5: 1121–1132
Asigbu, FO, Kacprzak, M, Daniel, G, Johansson, M, Stenlid, J, Manka, M (1999) Biochemical interactions of conifer seedling roots with Fusarium spp. Can J Microbiol 45: 923–935
Avis, TJ, Hamelin, RC, Bélanger, RR (2001) Approaches to molecular characterization of fungal biocontrol agents: some case studies. Can J Plant Pathol 23: 8–12
Axelrood, PE, Neumann, M, Trotter, D, Radley, R, Shrimpton, G, Dennis, J (1995) Seedborne Fusarium on Duglas-fir: pathogenicity and seed stratification method to decrease Fusarium contamination. New For 9: 35–51
Bais, HP, Park, SW, Weir, TL, Callaway, RM, Vivanco, MJ (2004) How plants communicate using the underground information superhighway. Trends Plant Sci 9: 26–32
Bloomberg, WJ (1981) Disease caused by Fusarium in forest nurseries. In: Nelson, PE, Toussoun, TA, Cook, RJ (Eds.) Fusarium: Disease, Biology and Taxonomy, Pennsylvania State University Press, University Park, pp 178–187
Canadian Council of Forest Ministers (2005) Compendium of Canadian Forestry Statistics, http://www.nfdp.ccfm.org
Capieau, K (2004) Biological control of grey mould in Swedish forest nurseries. Ph.D. Thesis, SLU, Acta Universitatis agriculturae Sueciae. Silvestria Vol. 325
Casgrain, P, Legendre, P (2001) The R package for multivariate and spatial analysis. Département de sciences biologiques, Université de Montréal, Québec
Colas, F, Tousignant, D, Mercier S (2003) Recent research in tree reproduction in Quebec. XII World Forestry Congress, Quebec-Canada. MRNFP-Research Note, http://www.mrnf.gouv.qc.ca/english/publications/forest/publications/Colas-A.pdf
Cordell, CE, Anderson, RL, Hoffard, WH, Landis, TD, Smith, RS Jr, Toko, HV (1989) Cylindrocladium USDA Forest Service, Diseases: Forest Nursery Pests No. 680: Agriculture Handbook. United States Government Printing, Washington DC
Crous, PW (2005) Impact of molecular phylogenetics on the taxonomy and diagnostics of fungi. EPPO Bull 35: 47–51
Crous, P, Wingfield, MJ (1994) A monograph of Cylindrocladium, including anamorph of Calonectria. Mycotaxon 51: 341–435
Duchesne, LC, Peterson, RL, Ellis, BE (1987) Interaction between the ectomycorrhizal fungus Paxillus involutus and Pinus resinosa induces resistance of Fusarium oxysporum. Can J Bot 66: 558–562
Dumas, MT, Strunz, GM, Boyonoski, NW, Finlay, HJ (1996) In vitro interactions between Cylindrocladium floridanum and species of Trichoderma. Can J Plant Pathol 18: 325–329
Dumas, MT, Greifenhagen, S, Halicki-Hayden, G, Meyer, TR (1998) Effect of seedbed steaming on Cylindrocladium floridanum soil microbes and the development of white pine seedlings. Phytoprotection 79: 35–43
Dumroese, RK, James, RL, Wemmy, D (2000) An assessment of Cylindrocarpon on container western white pine seedlings after outplanting. West J Appl For 15: 5–7
Enebek, SA, Wei, G, Kloepper, JW (1998) Effects of plant growth-promoting Rhizobacteria on loblolly and slach pine seedlings. For Sci 44: 139–144
Filion, M, Hamelin, RC, Bernier, L, St-Arnaud, M (2004) Molecular profiling of rhizosphere microbial communities associated with healthy and diseased black spruce (Picea mariana) seedlings grown in a nursery. Appl Environ Microbiol 70: 3541–3551
Garbeva, P, van Veen, JA, van Elsas, JD (2004) Microbial diversity in soil: selection of microbial populations by plant soil type and implications for disease suppressiveness. Ann Rev Phytopathol 42: 243–270
Gardes, M, Bruns, TD (1993) ITS primers with enhanced specificity for basidiomycetes—application to the identification of mycorrhiza and rusts. Mol Ecol 2: 113–118
Hadi, S (1974) Epidemiology and control of Cylindrocladium stem canker and root rot of conifers in nurseries. Ph.D. Thesis, University of Wisconsin
Hamelin, RC, Bérubé, P, Gignac, M, Bourassa, M (1996) Identification of root rot fungi in nursery seedlings by nested multiplex PCR. Appl Environ Microbiol 62: 4026–4031
Holdenrieder, O, Sieber, TN (1992) Fungal associations of serially washed healthy non-mycorrhizal roots of Picea abies. Mycol Res 96: 151–156
Innes, L (1993) Moyen de lutte contre la pourriture racinaire dans les pépinières forestières. MRN/RNC, Insectes et Maladies des arbres, St-Foy, Québec
James, RL, Dumroese, RK, Wenny, DL (1995) Fusarium proliferatum is common, aggressive pathogen of container-grow conifer seedlings. Phytopathology 85: 1129
Jeng, RS, Dumas, M, Liu, FH, Hubbes, M (1997) DNA analysis of Cylindrocladium floridanum isolates from selected forest nurseries. Mycol Res 101: 285–291
Juzwick, J, Honhart, C, Choung, N (1988) Cylindrocladium root rot in Ontario bare-root nurseries: estimate of spruce seedling losses. Can J For Res 18: 1493–1496
Kang, JC, Crous, PW, Schoch, CL (2001) Species concepts in the Cylindrocladium floridanum and Cy. spathiphylli complexes (Hypocreaceae) based on multi-allelic sequence data, sexual compatibility and morphology. Syst Appl Microbiol 24: 206–217
Keiko, Y, Yukiharu, F, Yasuyuki, H, Tadashi, Y, Satoshi, T (2001) Penicillium fungi from Picea glehnii seeds protect the seedlings from damping-off. New Phytol 152: 521–531
Kernaghan, G, Sigler, L, Khasa, D (2003) Mycorrhizal and root edophytic fungi of containerized Picea glauca seedlings assessed by rDNA sequence analysis. Microb Ecol 45: 128–136
Kope, HH, Alexrood, PE, Sutherland, J, Reddy, MS (1996) Prevalence and incidence of the root-inhabiting fungi Fusarium, Cylindrocarpon, and Pythium, on container grown douglas-fir and spruce seedlings in British Columbia. New For 12: 55–67
Lilja, A, Poteri, M, Vuorinen, M, Kurkela, T, Hantula, J (2005) Cultural and PCR-based identification of the two most common fungi from canker container-grown Norway spruce seedlings. Can J For Res 35: 432–439
Mazzola, M (2004) Assessment and management of soil microbial community structure for disease suppression. Ann Rev Phytopathol 42: 35–59
Meyer, TR, Irvine, M, Harvey, EM, McDonough, T (1993) Integrated Pest Management in Canadian Forest Nurseries—Current Perspectives and Future Opportunities. USDA Forest Service, Rocky Mountain Forest Range Experimental Station, General Report, RM-243. USDA Forest Service, Washington DC
Morin, C, Samson, J, Dessureault, M (1999) Protection of black spruce seedlings against Cylindrocladium root rot with ectomycorrhizal fungi. Can J Bot 77: 169–174
Nicoletti, R, Stefano, MD, Stefano, SD, Trincone, A, Marziano, F (2004) Antagonism against Rhizoctonia solani and fungitoxic metabolite production by some Penicillium isolates. Mycopathologia 158: 465–474
Paulitz, TC, Bélanger, RR (2001) Biological control in greenhouse systems. Ann Rev Phytopathol 39: 103–133
Punja, ZK, Utkhede, RS (2003) Using fungi and yeasts to manage vegetable crop diseases. Trends Biotechnol 21: 400–407
Rosenzweig, ML (1995) Species Diversity in Space and Time. Cambridge University Press, Princeton
Sabuquillo, P, Cal, AD, Melgarejo, P (2006) Biocontrol of tomato wilt by Penicillium oxalicum formulations in different crop conditions. Biol Control 37: 256–265
Saunders, JE, Juzwik, J, Hutchinson, R (1992) Outplanting survival of Cylindrocladium root rot affected black spruce seedlings. Can J For Res 2: 1204–1207
Sneath, PH, Sokal, RR (1973) Numerical Taxonomy: The Principles and Practice of Numerical Classification. WH Freeman, San Francisco
St-Arnaud, M, Vujanovic, V (2007) Effect of the arbuscular mycorrhizal symbiosis on plant diseases and pests. In: Hamel, C, Plenchette, C (Eds.) Mycorrhizae in Crop Production: Applying Knowledge, Haworth Press, Binghampton. In press
Summerbell, RC (1989) Microfungi associated with mycorrhizal mantle and adjacent microhabitats within the rhizosphere of black spruce. Can J Bot 67: 1085–1095
Sutherland, JR, Lock, W, Farris, SH (1981) Sirococcus blight: a seed born disease of container grown seedlings in coastal British Columbia forest nurseries. Can J Bot 59: 559–562
Sutherland, JR, Shrimpton, GM, Sturrock, RN (1989) Diseases and Insects in British Columbia Forest Seedling Nurseries. FRDA Report, ISSN 0835-0752. FRDA, Victoria
Swofford, DL (2000) PAUP*. Phylogenetic Analysis Using Parsimony (and other methods), Version 4,0b10. Sinauer Associates, Sunderland
ter Braak, CJF, Šmilauer, P (2002) CANOCO. Reference Manual and CanoDraw for Windows User’s Guide: Software for Canonical Community Ordination, (version 4.5). Microcomputer Power, Ithaca
Thompson, JD, Gibson, TJ, Plewniak, F, Jeanmougin, F, Higgins, DG (1997) The ClustalX windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res 24: 4876–4882
Tousignant, D, Périnet, P, Rioux, M (1996) Black spruce cutting propagation at the Pépinière de Saint-Modeste. Gouvernement du Québec, Ministère des Ressources naturelles, Quebec
Vey, A, Hoagland, R, Butt, T (2001) Toxic metabolites of fungal biocontrol agents. In: Butt, T, Jackson, C, Magan, N (Eds.) Fungi as Biocontrol Agents: Progress, Problems and Potential, CAB International, Wallingford, pp 311–346
Vujanovic, V, St-Arnaud, M (2005) Root rot of black spruce caused by Cylindrocladium canadense in eastern North America. Plant Dis 89: 204
Vujanovic, V, St-Arnaud, M, Neumann, P (2000) Susceptibility of cones and seeds to fungal infection in a pine (Pinus spp.) collection. For Pathol 30: 305–320
Whipps, JM (2001) Microbial interactions and biocontrol in rhizosphere. J Exp Bot 52: 487–511
White, TJ, Bruns, T, Lee, S, Taylor, JW (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis, MA, Gelfand, DH, Sninsky, JJ, White, TJ (Eds.) PCR Protocols: Guide to Methods and Applications, Academic, New York, pp 315–322
Yergeau, E, Filion, M, Vujanovic, V, St-Arnaud, M (2005) A PCR-denaturing gradient gel electrophoresis (DGGE) approach to assess Fusarium diversity in asparagus. J Microbiol Methods 60: 143–154
Yung, C-H, Crowley, DE (2000) Rhizosphere microbial community structures in relation to root location and plant iron nutritional status. Appl Environ Microbiol 66: 345–351
Acknowledgment
We thank the NSERC-funded Biocontrol Network for financial support of this research. We also want to thank E. Zimmerman for critical review of the manuscript, and the “Centre de bouturage de Saint-Modeste,” Gouvernement du Québec-MRNF, for kindly giving permission to sample and providing technical assistance.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Vujanovic, V., Hamelin, R.C., Bernier, L. et al. Fungal Diversity, Dominance, and Community Structure in the Rhizosphere of Clonal Picea mariana Plants Throughout Nursery Production Chronosequences. Microb Ecol 54, 672–684 (2007). https://doi.org/10.1007/s00248-007-9226-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-007-9226-1