Abstract
Background
Thickened corpus callosum is a rare finding and its pathophysiology is not well known. An anomalous supracallosal bundle has been depicted by fiber tracking in some cases but no diffusion tensor imaging metrics of thickened corpus callosum have been reported.
Objective
To use diffusion tensor imaging (DTI) in cases of thickened corpus callosum to help in understanding its clinical significance.
Materials and methods
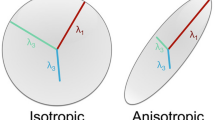
During a 7-year period five children (ages 6 months to 15 years) with thickened corpus callosum were studied. We determined DTI metrics of fractional anisotropy (FA), mean diffusivity, and axial (λ1) and radial (λ2, λ3) diffusivity and performed 3-D fiber tracking reconstruction of the thickened corpus callosum. We compared our results with data from the literature and 24 age-matched controls.
Results
Brain abnormalities were seen in all cases. All children had at least three measurements of corpus callosum thickness above the 97th percentile according to age. In all children 3-D fiber tracking showed an anomalous supracallosal bundle and statistically significant decrease in FA (P = 0.003) and λ1 (P = 0.001) of the corpus callosum compared with controls, but no significant difference in mean diffusivity and radial diffusivity.
Conclusion
Thickened corpus callosum was associated with abnormal bundles, suggesting underlying axonal guidance abnormality. DTI metrics suggested abnormal fiber compactness and density, which may be associated with alterations in cognition.



Similar content being viewed by others
References
Giedd JN, Blumenthal J, Jeffries NO et al (1999) Development of the human corpus callosum during childhood and adolescence: a longitudinal MRI study. Prog Neuropsychopharmacol Biol Psychiatry 23:571–588
Garel C, Cont I, Alberti C et al (2011) Biometry of the corpus callosum in children: MR imaging reference data. AJNR Am J Neuroradiol 32:1436–1443
Rollins NK (2013) Diffusion imaging of the congenitally thickened corpus callosum. AJNR Am J Neuroradiol 34:660–665
Lerman-Sagie T, Ben-Sira L, Achiron R et al (2009) Thick fetal corpus callosum: an ominous sign? Ultrasound Obstet Gynecol 34:55–61
Bindu PS, Taly AB, Sinha S et al (2010) Mega-corpus callosum, polymicrogyria, and psychomotor retardation syndrome. Pediatr Neurol 42:129–132
Göhlich-Ratmann G, Baethmann M, Lorenz P et al (1998) Megalencephaly, mega corpus callosum, and complete lack of motor development: a previously undescribed syndrome. Am J Med Genet 79:161–167
Hengst M, Tücke J, Zerres K et al (2010) Megalencephaly, mega corpus callosum, and complete lack of motor development: delineation of a rare syndrome. Am J Med Genet 152A:2360–2364
Dagli AI, Stalker HJ, Williams CA (2008) A patient with the syndrome of megalencephaly, mega corpus callosum and complete lack of motor development. Am J Med Genet 146A:204–207
Rypens F, Sonigo P, Aubry MC et al (1996) Prenatal MR diagnosis of a thick corpus callosum. AJNR Am J Neuroradiol 17:1918–1920
Pierson TM, Zimmerman RA, Tennekoon GI et al (2008) Mega-corpus callosum, polymicrogyria, and psychomotor retardation: confirmation of a syndromic entity. Neuropediatrics 39:123–127
Kivitie-Kallio SH, Autti TH, Salonen OH et al (1998) MRI of the brain in the Cohen syndrome: a relatively large corpus callosum in patients with mental retardation and microcephaly. Neuropediatrics 29:298–301
Sampaio A, Bouix S, Sousa N et al (2013) Morphometry of corpus callosum in Williams syndrome: shape as an index of neural development. Brain Struct Funct 218:711–720
Dubovsky EC, Booth TN, Vezina G et al (2001) MR imaging of the corpus callosum in pediatric patients with neurofibromatosis type 1. AJNR Am J Neuroradiol 22:190–195
Pöyhönen MH, Peippo MM, Valanne LK et al (2004) Hypertrichosis, hyperkeratosis, abnormal corpus callosum, mental retardation and dysmorphic features in three unrelated females. Clin Dysmorphol 13:85–90
Cavicchioni O, Gomes DM, Leroy B et al (2005) Prenatal diagnosis of de novo (7;19)(q11.2;q13.3) translocation associated with a thick corpus callosum and Wilms tumor of the kidneys. Prenat Diagn 25:876–878
Filippi CG, Watts R, Duy LA et al (2013) Diffusion-tensor imaging derived metrics of the corpus callosum in children with neurofibromatosis type I. AJR Am J Roentgenol 200:44–49
Koob M, Weingertner AS, Gasser B et al (2012) Thick corpus callosum: a clue to the diagnosis of fetal septopreoptic holoprosencephaly? Pediatr Radiol 42:886–890
Mitchell TN, Free SL, Merschhemke M et al (2003) Reliable callosal measurement: population normative data confirm sex-related differences. AJNR Am J Neuroradiol 24:410–418
Mori S, Crain BJ, Chacko VP et al (1999) Three-dimensional tracking of axonal projections in the brain by magnetic resonance imaging. Ann Neurol 45:265–269
Kadhim HJ, Bhide PG, Frost DO (1993) Transient axonal branching in the developing corpus callosum. Cereb Cortex 3:551–566
LaMantia AS, Rakic P (1994) Axon overproduction and elimination in the anterior commissure of the developing rhesus monkey. J Comp Neurol 340:328–336
Göçmen R, Oğuz KK (2008) Mega corpus callosum and caudate nuclei with bilateral hippocampal malformation. Diagn Interv Radiol 14:69–71
Raybaud C (2010) The corpus callosum, the other great forebrain commissures, and the septum pellucidum: anatomy, development, and malformation. Neuroradiology 52:447–477
Di Ieva A, Fathalla H, Cusimano MD et al (2014) The indusium griseum and the longitudinal striae of the corpus callosum. Cortex. doi:10.1016/j.cortex.2014.06.016 [Epub ahead of print]
Karol EA, Pandya DN (1971) The distribution of the corpus callosum in the rhesus monkey. Brain 94:471–486
LaMantia AS, Rakic P (1990) Axon overproduction and elimination in the corpus callosum of the developing rhesus monkey. J Neurosci 10:2156–2175
Elberger AJ (1994) Transitory corpus callosum axons projecting throughout developing rat visual cortex revealed by Dil. Cereb Cortex 4:279–299
Song SK, Sun SW, Ramsbottom MJ (2002) Dysmyelination revealed through MRI as increased radial (but unchanged axial) diffusion of water. Neuroimage 17:1429–1436
Tovar-Moll F, Moll J, de Oliveira-Souza R et al (2007) Neuroplasticity in human callosal dysgenesis: a diffusion tensor imaging study. Cereb Cortex 17:531–541
Hori A, Stan AC (2004) Supracallosal longitudinal fiber bundle: heterotopic cingulum, dorsal fornix or Probst bundle? Neuropathology 24:56–59
Poretti A, Meoded A, Rossi A et al (2013) Diffusion tensor imaging and fiber tractography in brain malformations. Pediatr Radiol 43:28–54
Zhang H, Schneider T, Wheeler-Kingshott CA et al (2012) NODDI: practical in vivo neurite orientation dispersion and density imaging of the human brain. Neuroimage 61:1000–1016
Kunz N, Zhang H, Vasung L et al (2014) Assessing white matter microstructure of the newborn with multi-shell diffusion MRI and biophysical compartment models. Neuroimage 96:288–299
Acknowledgments
We are grateful to Antoine Ponçet for his assistance in statistical analysis and to Seema Toso for her editorial assistance.
Conflicts of interest
None
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Merlini, L., Anooshiravani, M., Kanavaki, A. et al. Microstructural changes in thickened corpus callosum in children: contribution of magnetic resonance diffusion tensor imaging. Pediatr Radiol 45, 896–901 (2015). https://doi.org/10.1007/s00247-014-3242-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00247-014-3242-3