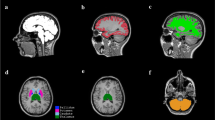
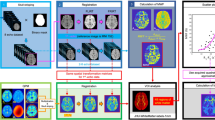
Abstract
The ability to dissociate axonal density in vivo from other microstructural properties is important for the diagnosis and treatment of neurologic disease, and new methods to do so are being developed. We investigated one such method—restricted diffusion imaging (RDI)—to see whether it can more accurately replicate histological axonal density patterns in the corpus callosum (CC) of adults and children compared to diffusion tensor imaging (DTI), neurite orientation dispersion and density imaging (NODDI), and generalized q-sampling imaging (GQI) methods. To do so, we compared known axonal density patterns defined by histology to diffusion-weighted imaging (DWI) scans of 840 healthy 20- to 40-year-old adults, and to DWI scans of 129 typically developing 7-month-old to 18-year-old children and adolescents. Contrast analyses were used to compare pattern similarities between the in vivo metric and previously published histological density models. We found that RDI was effective at mapping axonal density of small (Cohen’s d = 2.60) and large fiber sizes (Cohen’s d = 2.84) in adults. The same pattern was observed in the developing sample (Cohen’s d = 3.09 and 3.78, respectively). Other metrics, notably NODDI’s intracellular volume fraction in adults and GQI generalized fractional anisotropy in children, were also sensitive metrics. In conclusion, the study showed that the novel RDI metric is sensitive to density of small and large axons in adults and children, with both single- and multi-shell acquisition DWI data. Its effectiveness and availability to be used on standard as well as advanced DWI acquisitions makes it a promising method in clinical settings.








Similar content being viewed by others
Data availability
Data on adult participants were provided by the Human Connectome Project, WU-Minn Consortium (Principal Investigators: David Van Essen and Kamil Ugurbil; 1U54MH091657) funded by the 16 NIH Institutes and Centers that support the NIH Blueprint for Neuroscience Research; and by the McDonnell Center for Systems Neuroscience at Washington University. Data on child participants were from the Cincinnati MR Imaging of NeuroDevelopment (C-MIND) database, provided by the Pediatric Functional Neuroimaging Research Network (https://research.cchmc.org/c-mind/) and supported by a contract from the Eunice Kennedy Shriver National Institute of Child Health and Human Development (HHSN275200900018C).
References
Aboitiz F, Scheibel AB, Fisher RS, Zaidel E (1992) Fiber composition of the human corpus callosum. Brain Res 598:143–53. https://doi.org/10.1016/0006-8993(92)90178-c
Anderson DN, Duffley G, Vorwerk J, Dorval AD, Butson CR (2019) Anodic stimulation misunderstood: preferential activation of fiber orientations with anodic waveforms in deep brain stimulation. J Neural Eng 16:016026. https://doi.org/10.1088/1741-2552/aae590
Basser PJ, Mattiello J, LeBihan D (1994) MR diffusion tensor spectroscopy and imaging. Biophys J 66:259–67. https://doi.org/10.1016/S0006-3495(94)80775-1
Beaulieu C (2002) The basis of anisotropic water diffusion in the nervous system–a technical review. NMR Biomed 15:435–55. https://doi.org/10.1002/nbm.782
Bjornholm L et al (2017) Structural properties of the human corpus callosum: Multimodal assessment and sex differences. Neuroimage 152:108–118. https://doi.org/10.1016/j.neuroimage.2017.02.056
Broad RJ et al (2019) Neurite orientation and dispersion density imaging (NODDI) detects cortical and corticospinal tract degeneration in als. J Neurol Neurosurg Psychiatry 90:404–411. https://doi.org/10.1136/jnnp-2018-318830
Caminiti R et al (2013) Diameter, length, speed, and conduction delay of callosal axons in macaque monkeys and humans: comparing data from histology and magnetic resonance imaging diffusion tractography. J Neurosci 33:14501–11. https://doi.org/10.1523/JNEUROSCI.0761-13.2013
Chan AH et al (2008) Neural correlates of nouns and verbs in early bilinguals. Ann N Y Acad Sci 1145:30–40. https://doi.org/10.1196/annals.1416.000
Clarke J (1990) Interhemispheric functions in humans? Relationships between anatomical measures of the corpus callosum, behavioral laterality effects, and cognitive profiles. University of California, Los Angeles, Thesis
Cohen-Adad J et al (2008) Detection of multiple pathways in the spinal cord using q-ball imaging. Neuroimage 42:739–49. https://doi.org/10.1016/j.neuroimage.2008.04.243
Cook PA et al (2006) Camino: open-source diffusion-MRI reconstruction and processing
Daducci A et al (2014) Quantitative comparison of reconstruction methods for intra-voxel fiber recovery from diffusion MRI. IEEE Trans Med Imaging 33:384–99. https://doi.org/10.1109/TMI.2013.2285500
Daducci A et al (2015) Accelerated microstructure imaging via convex optimization (AMICO) from diffusion MRI data. Neuroimage 105:32–44. https://doi.org/10.1016/j.neuroimage.2014.10.026
Deluca GC, Ebers GC, Esiri MM (2004) The extent of axonal loss in the long tracts in hereditary spastic paraplegia. Neuropathol Appl Neurobiol 30:576–84. https://doi.org/10.1111/j.1365-2990.2004.00587.x
Descoteaux M, Angelino E, Fitzgibbons S, Deriche R (2007) Regularized, fast, and robust analytical q-ball imaging. Magn Reson Med 58:497–510. https://doi.org/10.1002/mrm.21277
D’Souza S, Ormond DR, Costabile J, Thompson JA (2019) Fiber-tract localized diffusion coefficients highlight patterns of white matter disruption induced by proximity to glioma. PLoS One 14:e0225323. https://doi.org/10.1371/journal.pone.0225323
Garyfallidis E et al (2014) Dipy, a library for the analysis of diffusion MRI data. Front Neuroinf 8. https://doi.org/10.3389/fninf.2014.00008
Genc S, Malpas CB, Holland SK, Beare R, Silk TJ (2017) Neurite density index is sensitive to age related differences in the developing brain. Neuroimage 148:373–380. https://doi.org/10.1016/j.neuroimage.2017.01.023
Genc S, Malpas CB, Ball G, Silk TJ, Seal ML (2018) Age, sex, and puberty related development of the corpus callosum: a multi-technique diffusion MRI study. Brain Struct Funct 223:2753–2765. https://doi.org/10.1007/s00429-018-1658-5
Grussu F et al (2017) Neurite dispersion: a new marker of multiple sclerosis spinal cord pathology? Ann Clin Transl Neurol 4:663–679. https://doi.org/10.1002/acn3.445
Harms RL, Fritz FJ, Tobisch A, Goebel R, Roebroeck A (2017) Robust and fast nonlinear optimization of diffusion MRI microstructure models. Neuroimage 155:82–96. https://doi.org/10.1016/j.neuroimage.2017.04.064
Hasan KM, Narayana PA (2006) Retrospective measurement of the diffusion tensor eigenvalues from diffusion anisotropy and mean diffusivity in DTI. Magn Reson Med 56:130–7. https://doi.org/10.1002/mrm.20935
Jespersen SN, Kroenke CD, Ostergaard L, Ackerman JJ, Yablonskiy DA (2007) Modeling dendrite density from magnetic resonance diffusion measurements. Neuroimage 34:1473–86. https://doi.org/10.1016/j.neuroimage.2006.10.037
Jones DK, Knosche TR, Turner R (2013) White matter integrity, fiber count, and other fallacies: the do’s and don’ts of diffusion MRI. Neuroimage 73:239–54. https://doi.org/10.1016/j.neuroimage.2012.06.081
Kumar R, Nguyen HD, Macey PM, Woo MA, Harper RM (2012) Regional brain axial and radial diffusivity changes during development. J Neurosci Res 90:346–55. https://doi.org/10.1002/jnr.22757
Kunz N et al (2014) Assessing white matter microstructure of the newborn with multi-shell diffusion MRI and biophysical compartment models. Neuroimage 96:288–99. https://doi.org/10.1016/j.neuroimage.2014.03.057
Lebel C, Beaulieu C (2009) Lateralization of the arcuate fasciculus from childhood to adulthood and its relation to cognitive abilities in children. Hum Brain Mapp 30:3563–73. https://doi.org/10.1002/hbm.20779
Lebel C, Deoni S (2018) The development of brain white matter microstructure. Neuroimage 182:207–218. https://doi.org/10.1016/j.neuroimage.2017.12.097
Mairal J, Bach F, Ponce J, Sapiro G (2010) Online learning for matrix factorization and sparse coding. J Mach Learn Res 11:19–60
Nir TM et al (2013) Effectiveness of regional DTI measures in distinguishing Alzheimer’s disease, mci, and normal aging. Neuroimage Clin 3:180–95. https://doi.org/10.1016/j.nicl.2013.07.006
Pecheva D et al (2018) Recent advances in diffusion neuroimaging: applications in the developing preterm brain. F1000Res. https://doi.org/10.12688/f1000research.15073.1
Qiu D, Tan LH, Zhou K, Khong PL (2008) Diffusion tensor imaging of normal white matter maturation from late childhood to young adulthood: voxel-wise evaluation of mean diffusivity, fractional anisotropy, radial and axial diffusivities, and correlation with reading development. Neuroimage 41:223–32. https://doi.org/10.1016/j.neuroimage.2008.02.023
R Core Team (2019) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Raffelt D et al (2012) Apparent fibre density: a novel measure for the analysis of diffusion-weighted magnetic resonance images. Neuroimage 59:3976–94. https://doi.org/10.1016/j.neuroimage.2011.10.045
Raffelt DA et al (2015) Connectivity-based fixel enhancement: whole-brain statistical analysis of diffusion mri measures in the presence of crossing fibres. Neuroimage 117:40–55. https://doi.org/10.1016/j.neuroimage.2015.05.039
Reyes-Haro D, Mora-Loyola E, Soria-Ortiz B, Garcia-Colunga J (2013) Regional density of glial cells in the rat corpus callosum. Biol Res 46:27–32. https://doi.org/10.4067/S0716-97602013000100004
Reynolds JE, Grohs MN, Dewey D, Lebel C (2019) Global and regional white matter development in early childhood. Neuroimage 196:49–58. https://doi.org/10.1016/j.neuroimage.2019.04.004
Riise J, Pakkenberg B (2011) Stereological estimation of the total number of myelinated callosal fibers in human subjects. J Anat 218:277–84. https://doi.org/10.1111/j.1469-7580.2010.01333.x
Ritchie JM (1982) On the relation between fibre diameter and conduction velocity in myelinated nerve fibres. Proc R Soc Lond B Biol Sci 217:29–35. https://doi.org/10.1098/rspb.1982.0092
Rosenthal R, Rosnow RL, Rubin DB (2000) Contrasts and effect sizes in behavioral research: a correlational approach (Cambridge University Press, New York, NY, US, 2000)
Sammartino F, Yeh FC, Krishna V (2019) Longitudinal analysis of structural changes following unilateral focused ultrasound thalamotomy. Neuroimage Clin 22:101754. https://doi.org/10.1016/j.nicl.2019.101754
Seehusen F, Baumgartner W (2010) Axonal pathology and loss precede demyelination and accompany chronic lesions in a spontaneously occurring animal model of multiple sclerosis. Brain Pathol 20:551–9. https://doi.org/10.1111/j.1750-3639.2009.00332.x
Sepehrband F et al (2015) Brain tissue compartment density estimated using diffusion-weighted MRI yields tissue parameters consistent with histology. Hum Brain Mapp 36:3687–702. https://doi.org/10.1002/hbm.22872
Song SK et al (2003) Diffusion tensor imaging detects and differentiates axon and myelin degeneration in mouse optic nerve after retinal ischemia. Neuroimage 20:1714–22. https://doi.org/10.1016/j.neuroimage.2003.07.005
Song SK et al (2005) Demyelination increases radial diffusivity in corpus callosum of mouse brain. Neuroimage 26:132–40. https://doi.org/10.1016/j.neuroimage.2005.01.028
Sotiropoulos SN et al (2013) Advances in diffusion MRI acquisition and processing in the human connectome project. Neuroimage 80:125–43. https://doi.org/10.1016/j.neuroimage.2013.05.057
Suzuki Y et al (2016) Estimation of the mean axon diameter and intra-axonal space volume fraction of the human corpus callosum: Diffusion q-space imaging with low q-values. Magn Reson Med Sci 15:83–93. https://doi.org/10.2463/mrms.2014-0141
Tallantyre EC et al (2009) Greater loss of axons in primary progressive multiple sclerosis plaques compared to secondary progressive disease. Brain 132:1190–9. https://doi.org/10.1093/brain/awp106
Taoka T et al (2020) White matter microstructural changes in tuberous sclerosis: Evaluation by neurite orientation dispersion and density imaging (NODDI) and diffusion tensor images. Sci Rep 10:436. https://doi.org/10.1038/s41598-019-57306-w
Tuch DS (2004) Q-ball imaging. Magn Reson Med 52:1358–72. https://doi.org/10.1002/mrm.20279
Veraart J, Sijbers J (2016) Diffusion kurtosis imaging. In: Diffusion tensor imaging: a practical handbook. Springer, New York
White NS, Leergaard TB, D’Arceuil H, Bjaalie JG, Dale AM (2013) Probing tissue microstructure with restriction spectrum imaging: histological and theoretical validation. Hum Brain Mapp 34:327–46. https://doi.org/10.1002/hbm.21454
Wilke M, Holland SK, Altaye M, Gaser C (2008) Template-o-matic: a toolbox for creating customized pediatric templates. Neuroimage 41:903–13. https://doi.org/10.1016/j.neuroimage.2008.02.056
Winston GP (2012) The physical and biological basis of quantitative parameters derived from diffusion MRI. Quant Imaging Med Surg 2:254–65. https://doi.org/10.3978/j.issn.2223-4292.2012.12.05
Winston GP et al (2014) Advanced diffusion imaging sequences could aid assessing patients with focal cortical dysplasia and epilepsy. Epilepsy Res 108:336–9. https://doi.org/10.1016/j.eplepsyres.2013.11.004
Yeh FC et al (2019) Automatic removal of false connections in diffusion MRI tractography using topology-informed pruning (tip). Neurotherapeutics 16:52–58. https://doi.org/10.1007/s13311-018-0663-y
Yeh FC, Verstynen TD (2016) Converting multi-shell and diffusion spectrum imaging to high angular resolution diffusion imaging. Front Neurosci 10:418. https://doi.org/10.3389/fnins.2016.00418
Yeh FC, Wedeen VJ, Tseng WY (2010) Generalized q-sampling imaging. IEEE Trans Med Imaging 29:1626–35. https://doi.org/10.1109/TMI.2010.2045126
Yeh FC, Liu L, Hitchens TK, Wu YL (2017) Mapping immune cell infiltration using restricted diffusion MRI. Magn Reson Med 77:603–612. https://doi.org/10.1002/mrm.26143
Zhang H, Hubbard PL, Parker GJ, Alexander DC (2011) Axon diameter mapping in the presence of orientation dispersion with diffusion MRI. Neuroimage 56:1301–15. https://doi.org/10.1016/j.neuroimage.2011.01.084
Zhang H, Schneider T, Wheeler-Kingshott CA, Alexander DC (2012) NODDI: practical in vivo neurite orientation dispersion and density imaging of the human brain. Neuroimage 61:1000–16. https://doi.org/10.1016/j.neuroimage.2012.03.072
Acknowledgements
Thank you to Adam Dick for help with figures, and to both reviewers for helpful comments on the earlier version of the manuscript.
Funding
This research was supported by NIH Grants R01MH112588 and R01DK119814 to P.G. and A.S.D.
Author information
Authors and Affiliations
Contributions
DG and ASD analyzed the data and wrote the initial draft. F-C Y provided advice on analyses of diffusion data and wrote the DSIStudio software. F-C Y and PG provided edits to the initial draft for the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest and have no relevant financial or non-financial interests to disclose.
Consent to participate.
Informed consent was obtained from all individual participants included in the study.
Code availability
Analyses used open source software. R code are available upon request.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Garic, D., Yeh, FC., Graziano, P. et al. In vivo restricted diffusion imaging (RDI) is sensitive to differences in axonal density in typical children and adults. Brain Struct Funct 226, 2689–2705 (2021). https://doi.org/10.1007/s00429-021-02364-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-021-02364-y