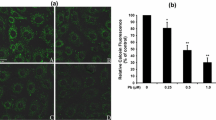
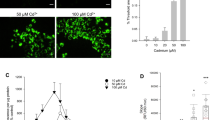
Abstract
The combined effects of lead (Pb) and cadmium (Cd) on primary cultures of rat proximal tubular (rPT) cells were studied. These cells were either treated with Pb acetate (0.5 and 1 μM) alone, Cd acetate (2.5 and 5 μM) alone, or a combination of Pb and Cd acetate, and then joint cytotoxicity was evaluated. The results showed that the combination of these two metals decreased cell viability and increased the number of apoptotic and necrotic cells and lactate dehydrogenase release synergistically. Simultaneously, increased intracellular reactive oxygen species, malondialdehyde, and calcium levels and decreased mitochondrial membrane potential, intracellular acidification, and inhibition of Na+, K+-, and Ca2+-ATPase activities were shown during the exposure. In addition, apoptotic morphological changes induced by these treatments in rPT cells were demonstrated by Hoechst 33258 staining. The apoptosis was markedly prevented by N-acetyl-l-cysteine, whereas necrosis was not affected. In summary, there was a synergistic cytototic effect of Pb combined with Cd on rPT cells. Cell death induced by Pb–Cd mixture was mediated by an apoptotic and a necrotic mechanism. Apoptotic death was the chief mechanism. Changes of intracellular events were intimately correlated with both oxidative stress and mitochondrial dysfunction, which promoted the development of apoptosis.








Similar content being viewed by others
References
Antonio Garcia T, Corredor L (2004) Biochemical changes in the kidneys after perinatal intoxication with lead and/or cadmium and their antagonistic effects when coadministered. Ecotoxicol Environ Saf 57:184–189
Bolduc JS, Denizeau F, Jumarie C (2004) Cadmium-induced mitochondrial membrane-potential dissipation does not necessarily require cytosolic oxidative stress: studies using rhodamine-123 fluorescence unquenching. Toxicol Sci 77:299–306
Brender JD, Suarez L, Felkner M, Gilani Z, Stinchcomb D, Moody K et al (2006) Maternal exposure to arsenic, cadmium, lead, and mercury and neural tube defects in offspring. Environ Res 101:132–139
Chakraborti T, Das S, Mondal M, Roychoudhury S, Chakraborti S (1999) Oxidant, mitochondria and calcium: an overview. Cell Signal 11:77–85
Dyatlov VA, Dyatlova OM, Parsons PJ, Lawrence DA, Carpenter DO (1998) Lipopolysaccharide and interleukin-6 enhance lead entry into cerebellar neurons: application of a new and sensitive flow cytometric technique to measure intracellular lead and calcium concentrations. Neurotoxicology 19:293–302
Ercal N, Treeratphan P, Hammond TC, Matthews RH, Grannemann N, Spitz D (1996) In vivo indices of oxidative stress in lead exposed C57BL/6 mice are reduced by treatment with meso-2,3-dimercaptosuccinic acid or N-acetyl cysteine. Free Radic Biol Med 21:157–161
Foster KA, Galeffi F, Gerich FJ, Turner DA, Müller M (2006) Optical and pharmacological tools to investigate the role of mitochondria during oxidative stress and neurodegeneration. Prog Neurobiol 79:136–171
Franco R, Sánchez-Olea R, Reyes-Reyes EM, Panayiotidis MI (2009) Environmental toxicity, oxidative stress and apoptosis: Ménage à trois. Mutat Res 674:3–22
Fujita T, Inoue H, Kitamura T, Sato N, Shimosawa T, Maruyama N (1998) Senescence marker protein-30 (SMP30) rescues cell death by enhancing plasma membrane Ca2+-pumping activity in HepG2 cells. Biochem Biophys Res Commun 250:374–380
Goyer RA (1989) Mechanisms of lead and cadmium nephrotoxicity. Toxicol Lett 46:153–162
Hirpara JL, Clément MV, Pervaiz S (2001) Intracellular acidification triggered by mitochondrial-derived hydrogen peroxide is an effector mechanism for drug-induced apoptosis in tumor cells. J Biol Chem 276:514–521
Kaufman DS, Goligorsky MS, Nord EP, Graber ML (1993) Perturbation of cell pH regulation by H2O2 in renal epithelial cells. Arch Biochem Biophys 302:245–254
Koh JY, Choi DW (1987) Quantitative determination of glutamate mediated cortical neuronal injury in cell culture by lactate dehydrogenase efflux assay. J Neurosci Methods 20:83–90
Kong IC, Bitton G, Koopman B, Jung KH (1995) Heavy metal toxicity testing in environmental samples. Rev Environ Contam Toxicol 142:119–147
Liu Z (2003) Lead poisoning combined with cadmium in sheep and horses in the vicinity of non-ferrous metal smelters. Sci Total Environ 309:117–126
Loghman-Adham M (1997) Renal effects of environmental and occupational lead exposure. Environ Health Perspect 105:928–938
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Matsuyama S, Llopis J, Deveraux QL, Tsien RY, Reed JC (2000) Changes in intramitochondrial and cytosolic pH: early events that modulate caspase activation during apoptosis. Nat Cell Biol 2:318–325
Morales AI, Vicente-Sánchez C, Sandoval JM, Egido J, Mayoral P, Arévalo MA et al (2006) Protective effect of quercetin on experimental chronic cadmium nephrotoxicity in rats is based on its antioxidant properties. Food Chem Toxicol 44:2092–2100
Muntner P, Menke A, Batuman V, Rabito FA, He J, Todd AC (2007) Association of tibia lead and blood lead with end-stage renal disease: a pilot study of African–Americans. Environ Res 104:396–401
Nolan CV, Shaikh ZA (1992) Lead nephrotoxicity and associated disorders: biochemical mechanisms. Toxicology 73:127–146
Nouwen EJ, Dauwe S, Van der Biest I, De Broe ME (1993) Stage- and segment-specific expression of cell-adhesion molecules N-CAM, A-CAM, and L-CAM in the kidney. Kidney Int 44:147–158
Nriagu JO, Pacyna JM (1988) Quantitative assessment of worldwide contamination of air, water and soils by trace metals. Nature 333:134–139
Oh SH, Lim SC (2006) A rapid and transient ROS generation by cadmium triggers apoptosis via caspase-dependent pathway in HepG2 cells and this is inhibited through N-acetylcysteine-mediated catalase upregulation. Toxicol Appl Pharmacol 212:212–223
Ott M, Gogvadze V, Orrenius S, Zhivotovsky B (2007) Mitochondria, oxidative stress and cell death. Apoptosis 12:913–922
Patra RC, Swarup D, Dwidedi SK (2001) Antioxidant effects of α tocopherol, ascorbic acid and l-methionine on lead-induced oxidative stress of the liver, kidney and brain in rats. Toxicology 162:81–88
Peñuelas J, Filella I (2002) Metal pollution in Spanish terrestrial ecosystems during the twentieth century. Chemosphere 46:501–505
Qin XJ, Li YN, Liang X, Wang P, Hai CX (2008) The dysfunction of ATPases due to impaired mitochondrial respiration in phosgene-induced pulmonary edema. Biochem Biophys Res Commun 367:150–155
Sen T, Sen N, Tripathi G, Chatterjee U, Chakrabarti S (2006) Lipid peroxidation associated cardiolipin loss and membrane depolarization in rat brain mitochondria. Neurochem Int 49:20–27
Thévenod F (2009) Cadmium and cellular signaling cascades: to be or not to be? Toxicol Appl Pharmacol 238:221–239
Üner N, Oruç E, Sevgiler Y (2005) Oxidative stress-related and ATPase effects of etoxazole in different tissues of Oreochromis niloticus. Environ Toxicol Pharmacol 20:99–106
Vermes I, Haanen C, Steffens-Nakken H, Reutelingsperger C (1995) A novel assay for apoptosis, flow cytometric detection of phosphatidylserine expression on early apoptotic cells using fluorescein-labeled Annexin V. J Immunol Methods 184:39–51
Walker LA, Simpson VR, Rockett L, Wienburg CL, Shore RF (2007) Heavy metal contamination in bats in Britain. Environ Pollut 148:483–490
Wang XQ, Xiao AY, Sheline C, Hyrc K, Yang A, Goldberg MP et al (2003) Apoptotic insults impair Na+, K+-ATPase activity as a mechanism of neuronal death mediated by concurrent ATP deficiency and oxidant stress. J Cell Sci 116:2099–2110
Wang L, Chen D, Cao J, Liu Z (2009a) Protective effect of N-acetylcysteine on experimental chronic cadmium nephrotoxicity in immature female rats. Hum Exp Toxicol 28(4):221–229
Wang L, Cao J, Chen D, Liu X, Lu H, Liu Z (2009b) Role of oxidative stress, apoptosis, and intracellular homeostasis in primary cultures of rat proximal tubular cells exposed to cadmium. Biol Trace Elem Res 127:53–68
Wang L, Chen D, Wang H, Liu Z (2009c) Effects of lead and/or cadmium on the expression of metallothionein in the kidney of rats. Biol Trace Elem Res 129(1–3):190–199
Wang L, Wang H, Hu M, Cao J, Chen D, Liu Z (2009d) Oxidative stress and apoptotic changes in primary cultures of rat proximal tubular cells exposed to lead. Arch Toxicol 83:417–427
Wang L, Li J, Li J, Liu Z (2010) Effects of lead and/or cadmium on the oxidative damage of rat kidney cortex mitochondria. Biol Trace Elem Res 137:69–78
Włostowski T, Krasowska A, Bonda E (2008) Joint effects of dietary cadmium and polychlorinated biphenyls on metallothionein induction, lipid peroxidation and histopathology in the kidneys and liver of bank voles. Ecotoxicol Environ Saf 69:403–410
Yang CF, Shen HM, Shen Y, Zhuang ZX, Ong CN (1997) Cadmium-induced oxidative cellular damage in human fetal lung fibroblasts (MRC-5) cells. Environ Health Perspect 105:712–716
Acknowledgements
This work was supported by the Shandong Provincial Natural Science Foundation of China (Grant No. ZR2010CQ014) and Science and Technology Innovation foundation for young scholars of Shandong Agricultural University in China (Grant No. 23690). Furthermore, Lin Wang and Heng Wang contributed equally to this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, L., Wang, H., Li, J. et al. Simultaneous Effects of Lead and Cadmium on Primary Cultures of Rat Proximal Tubular Cells: Interaction of Apoptosis and Oxidative Stress. Arch Environ Contam Toxicol 61, 500–511 (2011). https://doi.org/10.1007/s00244-011-9644-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00244-011-9644-4