Abstract
Purpose
The aim of the study is to explore interhemispheric homotopic functional connectivity alterations in systemic lupus erythematosus (SLE) patients with and without neuropsychiatric lupus (NPSLE and non-NPSLE, respectively) and their potential correlations with clinical characteristics and neuropsychological performance.
Methods
Based on resting-state functional MRI (rs-fMRI) data collected from SLE patients and matched healthy controls (HCs), the voxel-mirrored homotopic connectivity (VMHC) analysis was conducted to measure functional homotopy. Subsequently, correlations between altered functional homotopy and clinical/neuropsychological data were analyzed.
Results
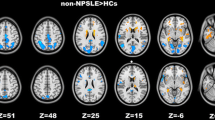
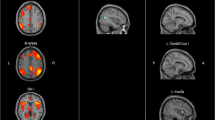
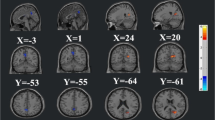
Compared with the HC group, both NPSLE and non-NPSLE groups showed attenuated homotopic connectivity in middle temporal gyrus (MTG), cuneus (CUN), middle occipital gyrus (MOG), angular gyrus (ANG), and postcentral gyrus (PoCG). NPSLE patients also exhibited decreased homotopic connectivity in inferior parietal gyrus (IPG) and middle frontal gyrus (MFG). Compared with non-NPSLE patients, NPSLE patients showed weaker interhemispheric homotopic functional connectivity in MOG. Decreased homotopic functional connectivity in PoCG, IPG, and MOG were associated with the anxiety state of SLE patients.
Conclusions
Our findings revealed attenuated functional homotopy in both NPSLE and non-NPSLE groups compared to the HC group, which appeared to be more severe in patients with comorbid neuropsychiatric lupus. Interhemispheric homotopy dysconnectivity may participate in the neuropathology of anxiety symptoms in SLE.





Similar content being viewed by others
References
Tsokos GC (2011) Systemic lupus erythematosus. N Engl J Med 365(22):2110–2121. https://doi.org/10.1056/NEJMra1100359
Ahn GY, Kim D, Won S, Song ST, Jeong HJ, Sohn IW, Lee S, Joo YB, Bae SC (2018) Prevalence, risk factors, and impact on mortality of neuropsychiatric lupus: a prospective, single-center study. Lupus 27(8):1338–1347. https://doi.org/10.1177/0961203318772021
The American College of Rheumatology nomenclature and case definitions for neuropsychiatric lupus syndromes (1999). Arthritis Rheum 42 (4):599–608. https://doi.org/10.1002/1529-0131(199904)42:4<599::AID-ANR2>3.0.CO;2-F
Zhang L, Fu T, Yin R, Zhang Q, Shen B (2017) Prevalence of depression and anxiety in systemic lupus erythematosus: a systematic review and meta-analysis. BMC Psychiatry 17(1):70. https://doi.org/10.1186/s12888-017-1234-1
Yue R, Gurung I, Long XX, Xian JY, Peng XB (2020) Prevalence, involved domains, and predictor of cognitive dysfunction in systemic lupus erythematosus. Lupus 29(13):1743–1751. https://doi.org/10.1177/0961203320958061
Moore E, Huang MW, Putterman C (2020) Advances in the diagnosis, pathogenesis and treatment of neuropsychiatric systemic lupus erythematosus. Curr Opin Rheumatol 32(2):152–158. https://doi.org/10.1097/BOR.0000000000000682
Schwartz N, Stock AD, Putterman C (2019) Neuropsychiatric lupus: new mechanistic insights and future treatment directions. Nat Rev Rheumatol 15(3):137–152. https://doi.org/10.1038/s41584-018-0156-8
Bonacchi R, Rocca MA, Ramirez GA, Bozzolo EP, Canti V, Preziosa P, Valsasina P, Riccitelli GC, Meani A, Moiola L, Rovere-Querini P, Manfredi AA, Filippi M (2020) Resting state network functional connectivity abnormalities in systemic lupus erythematosus: correlations with neuropsychiatric impairment. Mol Psychiatry. https://doi.org/10.1038/s41380-020-00907-z
Cao ZY, Wang N, Jia JT, Zhang HY, Shang SA, Hu JJ, Xu Y, Wu JT (2021) Abnormal topological organization in systemic lupus erythematosus: a resting-state functional magnetic resonance imaging analysis. Brain Imaging Behav 15(1):14–24. https://doi.org/10.1007/s11682-019-00228-y
Salvador R, Suckling J, Schwarzbauer C, Bullmore E (2005) Undirected graphs of frequency-dependent functional connectivity in whole brain networks. Philos Trans R Soc Lond B Biol Sci 360(1457):937–946. https://doi.org/10.1098/rstb.2005.1645
Stark DE, Margulies DS, Shehzad ZE, Reiss P, Kelly AM, Uddin LQ, Gee DG, Roy AK, Banich MT, Castellanos FX, Milham MP (2008) Regional variation in interhemispheric coordination of intrinsic hemodynamic fluctuations. J Neurosci 28(51):13754–13764. https://doi.org/10.1523/JNEUROSCI.4544-08.2008
Jiang W, Lei Y, Wei J, Yang L, Wei S, Yin Q, Luo S, Guo W (2019) Alterations of interhemispheric functional connectivity and degree centrality in cervical dystonia: a resting-state fMRI study. Neural Plast 2019:7349894. https://doi.org/10.1155/2019/7349894
Yu D, Yuan K, Bi Y, Luo L, Zhai J, Liu B, Li Y, Cheng J, Guan Y, Xue T, Bu L, Su S, Ma Y, Qin W, Tian J, Lu X (2018) Altered interhemispheric resting-state functional connectivity in young male smokers. Addict Biol 23(2):772–780. https://doi.org/10.1111/adb.12515
Nystedt J, Nilsson M, Jonsen A, Nilsson P, Bengtsson A, Lilja A, Latt J, Mannfolk P, Sundgren PC (2018) Altered white matter microstructure in lupus patients: a diffusion tensor imaging study. Arthritis Res Ther 20(1):21. https://doi.org/10.1186/s13075-018-1516-0
Zuo XN, Kelly C, Di Martino A, Mennes M, Margulies DS, Bangaru S, Grzadzinski R, Evans AC, Zang YF, Castellanos FX, Milham MP (2010) Growing together and growing apart: regional and sex differences in the lifespan developmental trajectories of functional homotopy. J Neurosci 30(45):15034–15043. https://doi.org/10.1523/JNEUROSCI.2612-10.2010
Guo W, Xiao C, Liu G, Wooderson SC, Zhang Z, Zhang J, Yu L, Liu J (2014) Decreased resting-state interhemispheric coordination in first-episode, drug-naive paranoid schizophrenia. Prog Neuropsychopharmacol Biol Psychiatry 48:14–19. https://doi.org/10.1016/j.pnpbp.2013.09.012
Liu Y, Guo W, Zhang Y, Lv L, Hu F, Wu R, Zhao J (2018) Decreased resting-state interhemispheric functional connectivity correlated with neurocognitive deficits in drug-naive first-episode adolescent-onset schizophrenia. Int J Neuropsychopharmacol 21(1):33–41. https://doi.org/10.1093/ijnp/pyx095
Liu HH, Wang J, Chen XM, Li JP, Ye W, Zheng J (2016) Interhemispheric functional and structural alterations and their relationships with alertness in unilateral temporal lobe epilepsy. Eur Rev Med Pharmacol Sci 20(8):1526–1536
Jia C, Ou Y, Chen Y, Li P, Lv D, Yang R, Zhong Z, Sun L, Wang Y, Zhang G, Guo H, Sun Z, Wang W, Wang Y, Wang X, Guo W (2020) Decreased resting-state interhemispheric functional connectivity in medication-free obsessive-compulsive disorder. Front Psychiatry 11:559729. https://doi.org/10.3389/fpsyt.2020.559729
Nasreddine ZS, Phillips NA, Bedirian V, Charbonneau S, Whitehead V, Collin I, Cummings JL, Chertkow H (2005) The Montreal cognitive assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc 53(4):695–699. https://doi.org/10.1111/j.1532-5415.2005.53221.x
Yu J, Li J, Huang X (2012) The Beijing version of the Montreal cognitive assessment as a brief screening tool for mild cognitive impairment: a community-based study. BMC Psychiatry 12:156. https://doi.org/10.1186/1471-244X-12-156
Hamilton M (1959) The assessment of anxiety states by rating. Br J Med Psychol 32(1):50–55. https://doi.org/10.1111/j.2044-8341.1959.tb00467.x
Hamilton M (1960) A rating scale for depression. J Neurol Neurosurg Psychiatry 23:56–62. https://doi.org/10.1136/jnnp.23.1.56
Guo W, Liu F, Dai Y, Jiang M, Zhang J, Yu L, Long L, Chen H, Gao Q, Xiao C (2013) Decreased interhemispheric resting-state functional connectivity in first-episode, drug-naive major depressive disorder. Prog Neuropsychopharmacol Biol Psychiatry 41:24–29. https://doi.org/10.1016/j.pnpbp.2012.11.003
Li J, Yuan Y, Wang M, Zhang J, Zhang L, Jiang S, Wang X, Ding J, Zhang K (2018) Decreased interhemispheric homotopic connectivity in Parkinson’s disease patients with freezing of gait: A resting state fMRI study. Parkinsonism Relat Disord 52:30–36. https://doi.org/10.1016/j.parkreldis.2018.03.015
Wang Y, Zhong S, Jia Y, Zhou Z, Zhou Q, Huang L (2015) Reduced interhemispheric resting-state functional connectivity in unmedicated bipolar II disorder. Acta Psychiatr Scand 132(5):400–407. https://doi.org/10.1111/acps.12429
Yu Y, Chen L, Wang Q, Hu L, Ding Q, Jia X, Yang X (2019) Altered amplitude of low-frequency fluctuations in inactive patients with nonneuropsychiatric systemic lupus erythematosus. Neural Plast 2019:9408612. https://doi.org/10.1155/2019/9408612
Inoue K, Nakanishi K, Hadoush H, Kurumadani H, Hashizume A, Sunagawa T, Ochi M (2013) Somatosensory mechanical response and digit somatotopy within cortical areas of the postcentral gyrus in humans: an MEG study. Hum Brain Mapp 34(7):1559–1567. https://doi.org/10.1002/hbm.22012
Kropf E, Syan SK, Minuzzi L, Frey BN (2019) From anatomy to function: the role of the somatosensory cortex in emotional regulation. Braz J Psychiatry 41(3):261–269. https://doi.org/10.1590/1516-4446-2018-0183
Minuzzi L, Syan SK, Smith M, Hall A, Hall GB, Frey BN (2018) Structural and functional changes in the somatosensory cortex in euthymic females with bipolar disorder. Aust N Z J Psychiatry 52(11):1075–1083. https://doi.org/10.1177/0004867417746001
Brady RO Jr, Masters GA, Mathew IT, Margolis A, Cohen BM, Ongur D, Keshavan M (2016) State dependent cortico-amygdala circuit dysfunction in bipolar disorder. J Affect Disord 201:79–87. https://doi.org/10.1016/j.jad.2016.04.052
Wang W, Peng Z, Wang X, Wang P, Li Q, Wang G, Chen F, Chen X, Liu S (2019) Disrupted interhemispheric resting-state functional connectivity and structural connectivity in first-episode, treatment-naive generalized anxiety disorder. J Affect Disord 251:280–286. https://doi.org/10.1016/j.jad.2019.03.082
Lu F, Wang M, Xu S, Chen H, Yuan Z, Luo L, Wang X, Zhang J, Dai J, Wang X, Chen H, Zhou J (2020) Decreased interhemispheric resting-state functional connectivity in male adolescents with conduct disorder. Brain Imaging Behav. https://doi.org/10.1007/s11682-020-00320-8
Pelphrey KA, Carter EJ (2008) Charting the typical and atypical development of the social brain. Dev Psychopathol 20(4):1081–1102. https://doi.org/10.1017/S0954579408000515
Whitney C, Jefferies E, Kircher T (2011) Heterogeneity of the left temporal lobe in semantic representation and control: priming multiple versus single meanings of ambiguous words. Cereb Cortex 21(4):831–844. https://doi.org/10.1093/cercor/bhq148
Xu J, Wang C, Xu Z, Li T, Chen F, Chen K, Gao J, Wang J, Hu Q (2020) Specific functional connectivity patterns of middle temporal gyrus subregions in children and adults with autism spectrum disorder. Autism Res 13(3):410–422. https://doi.org/10.1002/aur.2239
Blakemore SJ (2012) Development of the social brain in adolescence. J R Soc Med 105(3):111–116. https://doi.org/10.1258/jrsm.2011.110221
Zhang XD, Jiang XL, Cheng Z, Zhou Y, Xu Q, Zhang ZQ, Qi R, Luo S, Yun YS, Chen HJ, Kong X, Lu GM, Zhang LJ (2017) Decreased coupling between functional connectivity density and amplitude of low frequency fluctuation in non-neuropsychiatric systemic lupus erythematosus: a resting-stage functional MRI study. Mol Neurobiol 54(7):5225–5235. https://doi.org/10.1007/s12035-016-0050-9
Leslie B, Crowe SF (2018) Cognitive functioning in systemic lupus erythematosus: a meta-analysis. Lupus 27(6):920–929. https://doi.org/10.1177/0961203317751859
Yu H, Qiu X, Zhang YQ, Deng Y, He MY, Zhao YT, Zhai ZH (2019) Abnormal amplitude of low frequency fluctuation and functional connectivity in non-neuropsychiatric systemic lupus erythematosus: a resting-state fMRI study. Neuroradiology 61(3):331–340. https://doi.org/10.1007/s00234-018-2138-6
Liu S, Cheng Y, Xie Z, Lai A, Lv Z, Zhao Y, Xu X, Luo C, Yu H, Shan B, Xu L, Xu J (2018) A conscious resting state fMRI study in SLE patients without major neuropsychiatric manifestations. Front Psychiatry 9:677. https://doi.org/10.3389/fpsyt.2018.00677
Thomas LA, Brotman MA, Bones BL, Chen G, Rosen BH, Pine DS, Leibenluft E (2014) Neural circuitry of masked emotional face processing in youth with bipolar disorder, severe mood dysregulation, and healthy volunteers. Dev Cogn Neurosci 8:110–120. https://doi.org/10.1016/j.dcn.2013.09.007
Lai CH, Wu YT (2013) Decreased regional homogeneity in lingual gyrus, increased regional homogeneity in cuneus and correlations with panic symptom severity of first-episode, medication-naive and late-onset panic disorder patients. Psychiatry Res 211(2):127–131. https://doi.org/10.1016/j.pscychresns.2012.11.006
Zhu CM, Ma Y, Xie L, Huang JZ, Sun ZB, Duan SX, Lin ZR, Yin JJ, Le HB, Sun DM, Xu WC, Ma SH (2017) Spatial working memory impairment in patients with non-neuropsychiatric systemic lupus erythematosus: a blood-oxygen-level dependent functional magnetic resonance imaging study. J Rheumatol 44(2):201–208. https://doi.org/10.3899/jrheum.160290
Cao J, Chen X, Chen J, Ai M, Gan Y, Wang W, Lv Z, Zhang S, Zhang S, Wang S, Kuang L, Fang W (2016) Resting-state functional MRI of abnormal baseline brain activity in young depressed patients with and without suicidal behavior. J Affect Disord 205:252–263. https://doi.org/10.1016/j.jad.2016.07.002
Zhang R, Volkow ND (2019) Brain default-mode network dysfunction in addiction. Neuroimage 200:313–331. https://doi.org/10.1016/j.neuroimage.2019.06.036
Tomasi D, Volkow ND (2010) Functional connectivity density mapping. Proc Natl Acad Sci U S A 107(21):9885–9890. https://doi.org/10.1073/pnas.1001414107
Raichle ME (2015) The brain’s default mode network. Annu Rev Neurosci 38:433–447. https://doi.org/10.1146/annurev-neuro-071013-014030
Zhuo Z, Su L, Duan Y, Huang J, Qiu X, Haller S, Li H, Zeng X, Liu Y (2020) Different patterns of cerebral perfusion in SLE patients with and without neuropsychiatric manifestations. Hum Brain Mapp 41(3):755–766. https://doi.org/10.1002/hbm.24837
Hanly JG (2014) Diagnosis and management of neuropsychiatric SLE. Nat Rev Rheumatol 10(6):338–347. https://doi.org/10.1038/nrrheum.2014.15
Elkon KB, Santer DM (2012) Complement, interferon and lupus. Curr Opin Immunol 24(6):665–670. https://doi.org/10.1016/j.coi.2012.08.004
Acknowledgements
We sincerely thank all the participants in our study, and thank the Department of Radiology and the Department of Rheumatism and Immunology in the First Affiliated Hospital of Guangxi Medical University for their generous support. And we would like to express our gratitude to EditSprings (https://www.editsprings.com/) for the expert linguistic services provided.
Funding
This work was supported by grants from the Natural Science Foundation of Guangxi Province, China (2014GXNSFAA118192), and Foundation of Guangxi Educational Committee, China (ZD2014032).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, Yl., Jiang, Ml., Huang, LX. et al. Disrupted resting-state interhemispheric functional connectivity in systemic lupus erythematosus patients with and without neuropsychiatric lupus. Neuroradiology 64, 129–140 (2022). https://doi.org/10.1007/s00234-021-02750-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00234-021-02750-7