Abstract
Purpose
On account of the potential inhibition of OATP1B1 (organic anion transporting polypeptide) by angiotensin II receptor blockers (ARBs) and the effects of SLCO1B1 (solute carrier organic anion transporter family member) polymorphism, the aim of current study is to assess the impact of ARBs on the pharmacokinetics (PK) and pharmacodynamics (PD) of repaglinide in Chinese healthy volunteers with different SLCO1B1 genotypes.
Methods
The in vitro study was conducted on irbesartan, valsartan, olmesartan, and losartan by using HEK293 cells transfected with OATP1B1. Data on drug interactions between repaglinide and irbesartan from 21 healthy Chinese-Han male volunteers were collected and analyzed.
Results
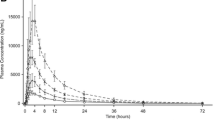
IC50 from in vitro study suggested irbesartan was the most potent inhibitor of OATP1B1 transporter. Clinical data from single dose of repaglinide indicated SLCO1B1 c.521 T>C polymorphism influenced the PK and PD of repaglinide in healthy Chinese-Han male volunteers. In subjects with SLCO1B1 c.521 TT genotype, irbesartan comedication increased the exposure of repaglinide. In details, the peak plasma concentration [Cmax] increased 84% (P = 0.003) and the area under the curve of plasma concentration 0–8 h [AUC0–8] increased 34% (P = 0.004), while the minimum blood glucose concentration [Cmin] decreased 33.8% (P = 0.005). No significant change was observed in repaglinide exposure in subjects with SLCO1B1 c.521 TC genotype in presence or absence of irbesartan.
Conclusion
SLCO1B1 c.521 T>C polymorphism affects the PK of repaglinide in Chinese population. Irbesartan increased repaglinide exposure in subjects with SLCO1B1 c.521 TT genotype, but not SLCO1B1 c.521 TC genotype.


Similar content being viewed by others
References
Hatorp V (2002) Clinical pharmacokinetics and pharmacodynamics of repaglinide. Clin Pharmacokinet 41(7):471–483
Kirchheiner J, Roots I, Goldammer M, Rosenkranz B, Brockmoller J (2005) Effect of genetic polymorphisms in cytochrome p450 (CYP) 2C9 and CYP2C8 on the pharmacokinetics of oral antidiabetic drugs: clinical relevance. Clin Pharmacokinet 44(12):1209–1225 eng
Bidstrup TB, Bjornsdottir I, Sidelmann UG, Thomsen MS, Hansen KT (2003) CYP2C8 and CYP3A4 are the principal enzymes involved in the human in vitro biotransformation of the insulin secretagogue repaglinide. Br J Clin Pharmacol 56(3):305–314
Niemi M, Backman JT, Kajosaari LI, Leathart JB, Neuvonen M, Daly AK et al (2005) Polymorphic organic anion transporting polypeptide 1B1 is a major determinant of repaglinide pharmacokinetics. Clin Pharmacol Ther 77(6):468–478 eng
Kajosaari LI, Laitila J, Neuvonen PJ, Backman JT (2005) Metabolism of repaglinide by CYP2C8 and CYP3A4 in vitro: effect of fibrates and rifampicin. Basic Clin Pharmacol Toxicol 97(4):249–256
Gan J, Chen W, Shen H, Gao L, Hong Y, Tian Y et al (2010) Repaglinide-gemfibrozil drug interaction: inhibition of repaglinide glucuronidation as a potential additional contributing mechanism. Br J Clin Pharmacol 70(6):870–880 eng
Zhu J, Song M, Tan HY, Huang LH, Huang ZJ, Liu C, Fu ZM, Huang YY, Tan ZR, Chen XP, Yuan H, Yang GP (2013) Effect of pitavastatin in different SLCO1B1 backgrounds on repaglinide pharmacokinetics and pharmacodynamics in healthy Chinese males. Pak J Pharm Sci 26(3):577–584
Kalliokoski A, Backman JT, Kurkinen KJ, Neuvonen PJ, Niemi M (2008) Effects of gemfibrozil and atorvastatin on the pharmacokinetics of repaglinide in relation to SLCO1B1 polymorphism. Clin Pharmacol Ther 84(4):488–496
Niemi M, Schaeffeler E, Lang T, Fromm MF, Neuvonen M, Kyrklund C, Backman JT, Kerb R, Schwab M, Neuvonen PJ, Eichelbaum M, Kivistö KT (2004) High plasma pravastatin concentrations are associated with single nucleotide polymorphisms and haplotypes of organic anion transporting polypeptide-C (OATP-C, SLCO1B1). Pharmacogenetics 14(7):429–440
Kalliokoski A, Backman JT, Neuvonen PJ, Niemi M (2008) Effects of the SLCO1B1*1B haplotype on the pharmacokinetics and pharmacodynamics of repaglinide and nateglinide. Pharmacogenet Genomics 18(11):937–942 eng
He J, Qiu Z, Li N, Yu Y, Lu Y, Han D, Li T, Zhao D, Sun W, Fang F, Zheng J, Fan H, Chen X (2011) Effects of SLCO1B1 polymorphisms on the pharmacokinetics and pharmacodynamics of repaglinide in healthy Chinese volunteers. Eur J Clin Pharmacol 67(7):701–707
Xiang Q, Cui YM, Zhao X, Yan L, Zhou Y (2012) The influence of MDR1 G2677T/a genetic polymorphisms on the pharmacokinetics of repaglinide in healthy Chinese volunteers. Pharmacology 89(1–2):105–110
Taylor J (2013) 2013 ESH/ESC guidelines for the management of arterial hypertension. Eur Heart J 34(28):2108–2109
Karlgren M, Ahlin G, Bergstrom CA, Svensson R, Palm J, Artursson P (2012) In vitro and in silico strategies to identify OATP1B1 inhibitors and predict clinical drug-drug interactions. Pharm Res 29(2):411–426
Wang X, Wolkoff AW, Morris ME (2005) Flavonoids as a novel class of human organic anion-transporting polypeptide OATP1B1 (OATP-C) modulators. Drug Metab Dispos 33(11):1666–1672 eng
Wu LX, Guo CX, Chen WQ, Yu J, Qu Q, Chen Y, Tan ZR, Wang G, Fan L, Li Q, Zhang W, Zhou HH (2012) Inhibition of the organic anion-transporting polypeptide 1B1 by quercetin: an in vitro and in vivo assessment. Br J Clin Pharmacol 73(5):750–757
Wu LX, Guo CX, Qu Q, Yu J, Chen WQ, Wang G, Fan L, Li Q, Zhang W, Zhou HH (2012) Effects of natural products on the function of human organic anion transporting polypeptide 1B1. Xenobiotica 42(4):339–348
Hirano M, Maeda K, Shitara Y, Sugiyama Y (2006) Drug-drug interaction between pitavastatin and various drugs via OATP1B1. Drug Metab Dispos 34(7):1229–1236 eng
Ito K, Iwatsubo T, Kanamitsu S, Ueda K, Suzuki H, Sugiyama Y (1998) Prediction of pharmacokinetic alterations caused by drug-drug interactions: metabolic interaction in the liver. Pharmacol Rev 50(3):387–412
Zhang W, He YJ, Han CT, Liu ZQ, Li Q, Fan L, Tan ZR, Zhang WX, Yu BN, Wang D, Hu DL, Zhou HH (2006) Effect of SLCO1B1 genetic polymorphism on the pharmacokinetics of nateglinide. Br J Clin Pharmacol 62(5):567–572
Qiu X, Wang Z, Wang B, Zhan H, Pan X, Xu RA (2014) Simultaneous determination of irbesartan and hydrochlorothiazide in human plasma by ultra high performance liquid chromatography tandem mass spectrometry and its application to a bioequivalence study. J Chromatogr B Analyt Technol Biomed Life Sci 957:110–115
Cho SK, Yoon JS, Lee MG, Lee DH, Lim LA, Park K, Park MS, Chung JY (2011) Rifampin enhances the glucose-lowering effect of metformin and increases OCT1 mRNA levels in healthy participants. Clin Pharmacol Ther 89(3):416–421
Oswald S, Konig J, Lutjohann D, Giessmann T, Kroemer HK, Rimmbach C et al (2008) Disposition of ezetimibe is influenced by polymorphisms of the hepatic uptake carrier OATP1B1. Pharmacogenet Genomics 18(7):559–568 eng
Tirona RG, Leake BF, Wolkoff AW, Kim RB (2003) Human organic anion transporting polypeptide-C (SLC21A6) is a major determinant of rifampin-mediated pregnane X receptor activation. J Pharmacol Exp Ther 304(1):223–228 eng
Kameyama Y, Yamashita K, Kobayashi K, Hosokawa M, Chiba K (2005) Functional characterization of SLCO1B1 (OATP-C) variants, SLCO1B1*5, SLCO1B1*15 and SLCO1B1*15+C1007G, by using transient expression systems of HeLa and HEK293 cells. Pharmacogenet Genomics 15(7):513–522 eng
Ho RH, Tirona RG, Leake BF, Glaeser H, Lee W, Lemke CJ, Wang Y, Kim RB (2006) Drug and bile acid transporters in rosuvastatin hepatic uptake: function, expression, and pharmacogenetics. Gastroenterology 130(6):1793–1806
Iwai M, Suzuki H, Ieiri I, Otsubo K, Sugiyama Y (2004) Functional analysis of single nucleotide polymorphisms of hepatic organic anion transporter OATP1B1 (OATP-C). Pharmacogenetics 14(11):749–757
Katz DA, Carr R, Grimm DR, Xiong H, Holley-Shanks R, Mueller T, Leake B, Wang Q, Han L, Wang PG, Edeki T, Sahelijo L, Doan T, Allen A, Spear BB, Kim RB (2006) Organic anion transporting polypeptide 1B1 activity classified by SLCO1B1 genotype influences atrasentan pharmacokinetics. Clin Pharmacol Ther 79(3):186–196
Funding
This work was supported by the National Natural Science Foundation of China (81373490, 81573463, 81573508), National Key Research and Development Programs (2016YFC1306900, 2016YFC0905002, 2016YFC1201805), The Strategy-Oriented Special Project of Central South University in China (ZLXD2017003), and Hunan Natural Science Foundation (2017JJ3464).
Author information
Authors and Affiliations
Contributions
Zhao-QianLiu, Guo-Ping Yang, and Qi Pei conceived and designed the experiments; Qi Pei, Yi Zheng, Jun-Yan Liu, and Mi Luo analyzed the plasma concentrations. Qi Pei, Cheng-Xian Guo, Jun-Yan Liu, and Ji-Ye Yin performed the in vitro research. Qi Pei and Xi Li analyzed the data. Qi Pei, Xi Li, Ji-Ye Yin, and Zhao-Qian Liu wrote the paper. Shi-Kun Liu, Pan Xie, and Hong-Hao Zhou reviewed and edited the manuscript. All authors have read and approved the manuscript.
Corresponding authors
Ethics declarations
The study protocol was reviewed and approved by the ethics committee of the Institute of Clinical Pharmacology, Central South University (Changsha, Hunan, P. R. China).
Electronic supplementary material
ESM 1
(DOCX 646kb)
Rights and permissions
About this article
Cite this article
Pei, Q., Liu, JY., Yin, JY. et al. Repaglinide-irbesartan drug interaction: effects of SLCO1B1 polymorphism on repaglinide pharmacokinetics and pharmacodynamics in Chinese population. Eur J Clin Pharmacol 74, 1021–1028 (2018). https://doi.org/10.1007/s00228-018-2477-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-018-2477-6