Abstract
Purpose
Osteoporosis is a chronic disease of the bone, whose incidence increases progressively with aging. The main consequences of osteoporosis are fragility fractures, which have considerable medical, social, and economic implications. Adequate treatment of osteoporosis must be considered as a compelling public health intervention. Bisphosphonates (BPs) represent the most significant advance in this field in the past decade, and they are widely used in the treatment of osteoporosis. However, evidence for their effectiveness is limited to secondary prevention, whereas their effect in primary prevention is uncertain and needs further investigation.
Methods
Using administrative data collected in the “Biphosphonates Efficacy-Safety Tradeoff” (BEST) study, a nested case-control study was conducted by including 56,058 participants, aged 55 years who were started on oral BPs from 2003 to 2005. Cases were the 1,710 participants who were hospitalized for osteoporotic fractures until 2007. Up to 20 controls were randomly selected for each case. Conditional logistic regression model was used to estimate odds ratio of fracture associated with categories of treatment duration.
Results
Compared with participants assuming BPs for less than 1 year, those who remained on therapy for at least 2 years had a 21 % (95 % confidence interval (CI) 7 to 33 %) fracture risk reduction.
Conclusion
This study provides evidence that BPs, dispensed for primary prevention of osteoporotic fractures, are associated with a reduced risk of osteoporotic fractures after at least 2 years of treatment.
Similar content being viewed by others
References
Reginster JY, Burlet N (2006) Osteoporosis: a still increasing prevalence. Bone 38:S4–S9
Piscitelli P, Gimigliano F, Gatto S, Marinelli A, Gimigliano A, Marinelli P, Chitano G, Greco M, Di Paola L, Sbenaglia E, Benvenuto M, Muratore M, Quarta E, Calcagnile F, Colì G, Borgia O, Forcina B, Fitto F, Giordano A, Distante A, Rossini M, Angeli A, Migliore A, Guglielmi G, Guida G, Brandi ML, Gimigliano R, Iolascon G (2010) Hip fractures in Italy: 2000-2005 extension study. Osteoporos Int 21:1323–1330
Tarantino U, Capone A, Planta M, D'Arienzo M, Letizia Mauro G, Impagliazzo A, Formica A, Pallotta F, Patella V, Spinarelli A, Pazzaglia U, Zarattini G, Roselli M, Montanari G, Sessa G, Privitera M, Verdoia C, Corradini C, Feola M, Padolino A, Saturnino L, Scialdoni A, Rao C, Iolascon G, Brandi ML, Piscitelli P (2010) The incidence of hip, forearm, humeral, ankle, and vertebral fragility fractures in Italy: results from a 3-year multicenter study. Arthritis Res Ther 12:R226
Piscitelli P, Iolascon G, Gimigliano F, Muratore M, Camboa P, Borgia O, Forcina B, Fitto F, Robaud V, Termini G, Rini GB, Gianicolo E, Faino A, Rossini M, Adami S, Angeli A, Distante A, Gatto S, Gimigliano R, Guida G, SIOMMMS study group; CERSUM research group (2007) Incidence and costs of hip fractures compared to acute myocardial infarction in the Italian population: a 4-year survey. Osteoporos Int 18:211–219
Sampalis JS, Adachi JD, Rampakakis E, Vaillancourt J, Karellis A, Kindundu C (2012) Long-term impact of adherence to oral bisphosphonates on osteoporotic fracture incidence. J Bone Miner Res 27:202–210
McCormack JP, Loewen P (2007) Adding "value" to clinical practice guidelines. Can Fam Physician 53:1326–1327
Gullberg B, Johnell O, Kanis JA (1997) World-wide projections for hip fracture. Osteoporos Int 7:407–413
Cauley JA, Thompson DE, Ensrud KC, Scott JC, Black D (2000) Risk of mortality following clinical fractures. Osteoporos Int 11:556–561
Center JR, Nguyen TV, Schneider D, Sambrook PN, Eisman JA (1999) Mortality after all major types of osteoporotic fracture in men and women: an observational study. Lancet 353:878–882
Keene GS, Parker MJ, Pryor GA (1993) Mortality and morbidity after hip fractures. BMJ 307:1248–1250
Sahni M, Guenther HL, Fleisch H, Collin P, Martin TJ (1993) Bisphosphonates act on rat bone resorption through the mediation of osteoblasts. J Clin Invest 91:2004–2011
Harris ST, Watts NB, Genant HK, McKeever CD, Hangartner T, Keller M, Chesnut CH 3rd, Brown J, Eriksen EF, Hoseyni MS, Axelrod DW, Miller PD (1999) Effects of risedronate treatment on vertebral and nonvertebral fractures in women with postmenopausal osteoporosis: a randomized controlled trial. Vertebral Efficacy With Risedronate Therapy (VERT) Study Group. JAMA 282:1344–1352
Reginster J, Minne HW, Sorensen OH, Hooper M, Roux C, Brandi ML, Lund B, Ethgen D, Pack S, Roumagnac I, Eastell R (2000) Randomized trial of the effects of risedronate on vertebral fractures in women with established postmenopausal osteoporosis. Vertebral Efficacy with Risedronate Therapy (VERT) Study Group. Osteoporos Int 11:83–91
Black DM, Cummings SR, Karpf DB, Cauley JA, Thompson DE, Nevitt MC, Bauer DC, Genant HK, Haskell WL, Marcus R, Ott SM, Torner JC, Quandt SA, Reiss TF, Ensrud KE (1996) Randomised trial of effect of alendronate on risk of fracture in women with existing vertebral fractures. Fracture Intervention Trial Research Group. Lancet 348:1535–1541
Liberman UA, Weiss SR, Bröll J, Minne HW, Quan H, Bell NH, Rodriguez-Portales J, Downs RW Jr, Dequeker J, Favus M (1995) Effect of oral alendronate on bone mineral density and the incidence of fractures in postmenopausal osteoporosis. The Alendronate Phase III Osteoporosis Treatment Study Group. N Engl J Med 333:1437–1443
Pols HA, Felsenberg D, Hanley DA, Stepán J, Muñoz-Torres M, Wilkin TJ, Qin-sheng G, Galich AM, Vandormael K, Yates AJ, Stych B (1999) Multinational, placebo-controlled, randomized trial of the effects of alendronate on bone density and fracture risk in postmenopausal women with low bone mass: results of the FOSIT study. Fosamax International Trial Study Group. Osteoporos Int 9:461–468
Cummings SR, Black DM, Thompson DE, Applegate WB, Barrett-Connor E, Musliner TA, Palermo L, Prineas R, Rubin SM, Scott JC, Vogt T, Wallace R, Yates AJ, LaCroix AZ (1998) Effect of alendronate on risk of fracture in women with low bone density but without vertebral fractures: results from the Fracture Intervention Trial. JAMA 280:2077–2082
McClung MR, Geusens P, Miller PD, Zippel H, Bensen WG, Roux C, Adami S, Fogelman I, Diamond T, Eastell R, Meunier PJ, Reginster JY, Hip Intervention Program Study Group. Hip Intervention Program Study Group (2001) Effect of risedronate on the risk of hip fracture in elderly women. Hip Intervention Program Study Group. N Engl J Med 344:333–340
Wells GA, Cranney A, Peterson J, Boucher M, Shea B, Robinson V, Coyle D, Tugwell P (2008) Alendronate for the primary and secondary prevention of osteoporotic fractures in postmenopausal women. Cochrane Database Syst Rev 1:CD001155
Wells G, Cranney A, Peterson J, Boucher M, Shea B, Robinson V, Coyle D, Tugwell P (2008) Risedronate for the primary and secondary prevention of osteoporotic fractures in postmenopausal women. Cochrane Database Syst Rev 1:CD004523
Di Bari M, Balzi D, Roberts AT, Barchielli A, Fumagalli S, Ungar A, Bandinelli S, De Alfieri W, Gabbani L, Marchionni N (2010) Prognostic stratification of older persons based on simple administrative data: development and validation of the "Silver Code," to be used in emergency department triage. J Gerontol A Biol Sci Med Sci 65:159–164
Corrao G, Conti V, Merlino L, Catapano AL, Mancia G (2010) Results of a retrospective database analysis of adherence to statin therapy and risk of nonfatal ischemic heart disease in daily clinical practice in Italy. Clin Ther 32:300–310
Corrao G, Zambon A, Bertù L, Botteri E, Leoni O, Contiero P (2004) Lipid lowering drugs prescription and the risk of peripheral neuropathy: an exploratory case-control study using automated databases. J Epidemiol Community Health 58:1047–1051
Gini R, Francesconi P, Mazzaglia G, Cricelli I, Pasqua A, Gallina P, Brugaletta S, Donato D, Donatini A, Marini A, Zocchetti C, Cricelli C, Damiani G, Bellentani M, Sturkenboom MC, Schuemie MJ (2013) Chronic disease prevalence from Italian administrative databases in the VALORE project: a validation through comparison of population estimates with general practice databases and national survey. BMC Public Health 13:15
Degli Esposti L, Saragoni S, Batacchi P, Benemei S, Geppetti P, Sturani A, Buda S, Degli Esposti E (2012) Adherence to statin treatment and health outcomes in an Italian cohort of newly treated patients: results from an administrative database analysis. Clin Ther 34:190–199
Charlson ME, Charlson RE, Peterson JC, Marinopoulos SS, Briggs WM, Hollenberg JP (2008) The Charlson comorbidity index is adapted to predict costs of chronic disease in primary care patients. J Clin Epidemiol 61:1234–1240
Chandler JM, Zimmerman SI, Girman CJ, Martin AR, Hawkes W, Hebel JR, Sloane PD, Holder L, Magaziner J (2000) Low bone mineral density and risk of fracture in white female nursing home residents. JAMA 284:972–977
Marshall D, Johnell O, Wedel H (1996) Meta-analysis of how well measures of bone mineral density predict occurrence of osteoporotic fractures. BMJ 312:1254–1259
Black DM, Cummings SR, Genant HK, Nevitt MC, Palermo L, Browner W (1992) Axial and appendicular bone density predict fractures in older women. J Bone Miner Res 7:633–638
Greenland S (2001) Sensitivity analysis, Monte Carlo risk analysis, and Bayesian uncertainty assessment. Risk Anal 21:579–583
Corrao G, Nicotra F, Parodi A, Zambon A, Soranna D, Heiman F, Merlino L, Mancia G (2012) External adjustment for unmeasured confounders improved drug-outcome association estimates based on health care utilization data. J Clin Epidemiol 65:1190–1199
Blouin J, Dragomir A, Moride Y, Ste-Marie LG, Fernandes JC, Perreault S (2008) Impact of noncompliance with alendronate and risedronate on the incidence of nonvertebral osteoporotic fractures in elderly women. Br J Clin Pharmacol 66:117–127
Silverman SL, Watts NB, Delmas PD, Lange JL, Lindsay R (2007) Effectiveness of bisphosphonates on nonvertebral and hip fractures in the first year of therapy: the risedronate and alendronate (REAL) cohort study. Osteoporos Int 18:25–34
Caro JJ, Ishak KJ, Huybrechts KF, Raggio G, Naujoks C (2004) The impact of compliance with osteoporosis therapy on fracture rates in actual practice. Osteoporos Int 15:1003–1008
Siris ES, Harris ST, Rosen CJ, Barr CE, Arvesen JN, Abbott TA, Silverman S (2006) Adherence to bisphosphonate therapy and fracture rates in osteoporotic women: relationship to vertebral and nonvertebral fractures from 2 US claims databases. Mayo Clin Proc 81:1013–1022
Park-Wyllie LY, Mamdani MM, Juurlink DN, Hawker GA, Gunraj N, Austin PC, Whelan DB, Weiler PJ, Laupacis A (2011) Bisphosphonate use and the risk of subtrochanteric or femoral shaft fractures in older women. JAMA 305:783–789
Cramer JA, Gold DT, Silverman SL, Lewiecki EM (2007) A systematic review of persistence and compliance with bisphosphonates for osteoporosis. Osteoporos Int 18:1023–1031
Scotti L, Arfè A, Zambon A, Merlino L, Corrao G (2014) Cost-effectiveness of enhancing adherence with oral bisphosphonates treatment in osteoporotic women: an empirical approach based on healthcare utilisation databases. BMJ Open 4:e003758
Strom BL (2005) Overview of automated databases in pharmacoepidemiology. In: Pharmacoepidemiology (4th edn). Strom BL (ed). Wiley, New York, pp 219-22
Weycker D, Macarios D, Edelsberg J, Oster G (2007) Compliance with osteoporosis drug therapy and risk of fracture. Osteoporos Int 18:271–277
McCombs JS, Thiebaud P, McLaughlin-Miley C, Shi J (2004) Compliance with drug therapies for the treatment and prevention of osteoporosis. Maturitas 48:271–287
Johnell O, Gullberg B, Kanis JA (1997) The hospital burden of vertebral fracture in Europe: a study of national register sources. Osteoporos Int 7:138–144
Tamblyn R, Reid T, Mayo N, McLeod P, Churchill-Smith M (2000) Using medical services claims to assess injuries in the elderly: sensitivity of diagnostic and procedure codes for injury ascertainment. J Clin Epidemiol 53:183–194
Huybrechts KF, Ishak KJ, Caro JJ (2006) Assessment of compliance with osteoporosis treatment and its consequences in a managed care population. Bone 38:922–928
Corrao G, Ghirardi A, Segafredo G, Zambon A, Della Vedova G, Lapi F, Cipriani F, Caputi AP, Vaccheri A, Gregori D, Gesuita R, Vestri A, Staniscia D, Mazzaglia G, and Di Bari M, on behalf of the BEST Investigators (2014) User-only design for assessing drug effectiveness in clinical practice: application to bisphosphonates and secondary prevention of fracture. Pharmacoepidemiol Drug Saf. doi:10.1002/pds.3650
Acknowledgments
This study was almost entirely funded by a research grant from the AIFA—the Italian Medicines Agency—(AIFA grant FARM06R9YY), Rome, Italy. Data analyses were performed at the Unit of Biostatistics and Epidemiology, Department of Statistics, University of Milano-Bicocca, with grants from the Italian Minister for University and Research (’Fondo d’Ateneo per la Ricerca’ portion, year 2010). The authors thank Mrs. Ann Georgina Rhodes for editorial assistance. Author’s roles: study design: GM and GC. Data collection: LS, FL, AlV, DG, RG, AnV, TS. Data analysis: AG, AZ. Data interpretation: AG, AZ, MDB, and GC. Drafting manuscript: AG, AZ, MDB, and GC. Revising manuscript content: AG, MDB and GC. Approving final version of manuscript: AG, MDB, AZ, LS, GDV, FL, FC, APC, AV, DG, RG, AV, TS, GM, and GC. AG, MDB and GC take responsibility for the integrity of the data analysis.
Conflict of interest
All authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Consortia
Corresponding author
Additional information
Best investigators
Mazzaglia G (main investigator): Regional Agency for Healthcare Services of Tuscany, Italy
Cipriani F (Head Unit), Lapi F, Sessa E: Regional Agency for Healthcare Services of Tuscany, Italy
Caputi AP (Head Unit), Arcoraci V: Department of Medicine and Pharmacology, University of Messina, Messina, Italy
Corrao G (Head Unit), Ghirardi A, Scotti L, Parodi A, Zambon A: Department of Statistics and Quantitative Methods, Unit of Biostatistics, Epidemiology and Public Health, University of Milano-Bicocca, Milan, Italy
Montanaro N (Head Unit), Piccinni C, Suzzi C, Puccini A, Vaccheri A: Regional Centre for Drug Evaluation and Information (CREVIF), Department of Pharmacology, University of Bologna, Bologna, Italy.
Sturkenboom M (Head Unit): Departments of Epidemiology & Biostatistics and Medical Informatics, Pharmacoepidemiology Unit, Erasmus University Medical Center, The Netherlands
Geppetti P (Head Unit), Sati L, Di Bari M: Center of Pharmacoutilization, Pharmacoepidemiology, Pharmacovigilance and Pharmacoeconomics, University of Florence, Florence, Italy
Gregori D (Head Unit), Forlan F: Department of Public Health and Microbiology, University of Turin, Turin, Italy
Carle F (Head Unit), Gesuita R: Center of Epidemiology, Biostatistics, and Medical Information Technology, Polytechnic University of Marche, Ancona, Italy. Provided data from Marche
Staniscia T (Head Unit), Menna A: Regional Agency of Healthcare services of Abruzzo, L’Aquila, Italy
Vestri A (Head Unit): Department of Public Health and Infectious Diseases, University “La Sapienza”, Rome, Italy
Valenti M (Head Unit): Department of Medicine and Public Health, University of L’Aquila, L’Aquila, Italy
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 42 kb)
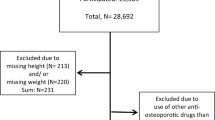
Supplemental Figure S1
Study flow diagram. AIFA-BEST project, Italy, 2003-2007 Footnote: BPs: Bisphosphonates (PDF 11 kb)
Rights and permissions
About this article
Cite this article
Ghirardi, A., Di Bari, M., Zambon, A. et al. Effectiveness of oral bisphosphonates for primary prevention of osteoporotic fractures. Eur J Clin Pharmacol 70, 1129–1137 (2014). https://doi.org/10.1007/s00228-014-1708-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-014-1708-8