Abstract
Purpose
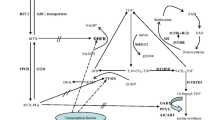
The objectives of this study were (1) to develop a population pharmacokinetic model of high-dose methotrexate (HD-MTX) in children with acute lymphoblastic leukaemia (ALL) and malignant lymphoma (ML) in order to investigate the influence of common polymorphisms in SLC19A1, MTHFR and ABCB1 on plasma levels of MTX and (2) to estimate MTX exposure in individual patients to study the association of genetic variability in the folate metabolic pathway with MTX toxicity.
Methods
The study population comprised 64 children with ALL/ML (age 1.6–16.8 years) who had received a total of 252 MTX courses (2–4 per patient). Common putative functional polymorphisms in the SLC19A1, MTHFR, MS, MTRR, TS and ABCB1 genes were analysed by PCR-based genotyping. Nonlinear mixed effects modelling was used for the pharmacokinetic analysis.
Results
The population typical value of clearance was 7.43 L/h (inter-individual variability 43.9%), central compartment volume was 16.7 L (46.6%), peripheral compartment volume was 2.6 L (63.3%) and distribution clearance was 0.0952 L/h (66.6%). MTX clearance decreased to 73.8% in patients with the MTHFR 677TT genotype. Patients homozygous for the variant MTHFR 1298A > C [odds ratio (OR) 0.14, 95% confidence interval (CI) 0.037–0.54] and SLC19A1 80A > G (OR 0.15, 95% CI 0.039-0.60) were at decreased risk for leucopenia. The TS 2R > 3R polymorphism was associated with a lower incidence of thrombocytopenia (OR 0.15, 95% CI 0.039–0.61) and mucositis (OR 0.016, 95% CI 0.0012-0.20). In contrast, the MTHFR 677TT polymorphism was associated with an increased incidence of mucositis (OR 23, 95% CI 2.1-240).
Conclusions
A population pharmacokinetic model developed in this study implies only a limited influence of genetic factors on the systemic disposition of MTX. Clearance is moderately reduced in patients with the MTHFR 677TT genotype. Genetic polymorphisms in the folate metabolic pathway and SLC19A1 were associated with HD-MTX toxicity.



Similar content being viewed by others
References
Reiter A, Schrappe M, Tiemann M, Ludwig WD, Yakisan E, Zimmermann M, Mann G, Chott A, Ebell W, Klingebiel T, Graf N, Kremens B, Muller-Weihrich S, Pluss HJ, Zintl F, Henze G, Riehm H (1999) Improved treatment results in childhood B-cell neoplasms with tailored intensification of therapy: a report of the Berlin-Frankfurt-Munster Group Trial NHL-BFM 90. Blood 94(10):3294–3306
Schrappe M, Reiter A, Ludwig WD, Harbott J, Zimmermann M, Hiddemann W, Niemeyer C, Henze G, Feldges A, Zintl F, Kornhuber B, Ritter J, Welte K, Gadner H, Riehm H (2000) Improved outcome in childhood acute lymphoblastic leukemia despite reduced use of anthracyclines and cranial radiotherapy: results of trial ALL-BFM 90. German-Austrian-Swiss ALL-BFM Study Group. Blood 95(11):3310–3322
Evans WE, Relling MV, Rodman JH, Crom WR, Boyett JM, Pui CH (1998) Conventional compared with individualized chemotherapy for childhood acute lymphoblastic leukemia. N Engl J Med 338(8):499–505
Moscow JA, Gong M, He R, Sgagias MK, Dixon KH, Anzick SL, Meltzer PS, Cowan KH (1995) Isolation of a gene encoding a human reduced folate carrier (RFC1) and analysis of its expression in transport-deficient, methotrexate-resistant human breast cancer cells. Cancer Res 55(17):3790–3794
Gorlick R, Goker E, Trippett T, Waltham M, Banerjee D, Bertino JR (1996) Intrinsic and acquired resistance to methotrexate in acute leukemia. N Engl J Med 335(14):1041–1048
Takatori R, Takahashi KA, Tokunaga D, Hojo T, Fujioka M, Asano T, Hirata T, Kawahito Y, Satomi Y, Nishino H, Tanaka T, Hirota Y, Kubo T (2006) ABCB1 C3435T polymorphism influences methotrexate sensitivity in rheumatoid arthritis patients. Clin Exp Rheumatol 24(5):546–554
Whetstine JR, Gifford AJ, Witt T, Liu XY, Flatley RM, Norris M, Haber M, Taub JW, Ravindranath Y, Matherly LH (2001) Single nucleotide polymorphisms in the human reduced folate carrier: characterization of a high-frequency G/A variant at position 80 and transport properties of the His(27) and Arg(27) carriers. Clin Cancer Res 7(11):3416–3422
Horie N, Aiba H, Oguro K, Hojo H, Takeishi K (1995) Functional analysis and DNA polymorphism of the tandemly repeated sequences in the 5′-terminal regulatory region of the human gene for thymidylate synthase. Cell Struct Funct 20(3):191–197
Frosst P, Blom HJ, Milos R, Goyette P, Sheppard CA, Matthews RG, Boers GJ, den Heijer M, Kluijtmans LA, van den Heuvel LP et al (1995) A candidate genetic risk factor for vascular disease: a common mutation in methylenetetrahydrofolate reductase. Nat Genet 10(1):111–113
Weisberg I, Tran P, Christensen B, Sibani S, Rozen R (1998) A second genetic polymorphism in methylenetetrahydrofolate reductase (MTHFR) associated with decreased enzyme activity. Mol Genet Metab 64(3):169–172
Leclerc D, Campeau E, Goyette P, Adjalla CE, Christensen B, Ross M, Eydoux P, Rosenblatt DS, Rozen R, Gravel RA (1996) Human methionine synthase: cDNA cloning and identification of mutations in patients of the cblG complementation group of folate/cobalamin disorders. Hum Mol Genet 5(12):1867–1874
Gaughan DJ, Kluijtmans LA, Barbaux S, McMaster D, Young IS, Yarnell JW, Evans A, Whitehead AS (2001) The methionine synthase reductase (MTRR) A66G polymorphism is a novel genetic determinant of plasma homocysteine concentrations. Atherosclerosis 157(2):451–456
Marzolini C, Paus E, Buclin T, Kim RB (2004) Polymorphisms in human MDR1 (P-glycoprotein): recent advances and clinical relevance. Clin Pharmacol Ther 75(1):13–33
Cheok MH, Evans WE (2006) Acute lymphoblastic leukaemia: a model for the pharmacogenomics of cancer therapy. Nat Rev Cancer 6(2):117–129
Aumente D, Buelga DS, Lukas JC, Gomez P, Torres A, Garcia MJ (2006) Population pharmacokinetics of high-dose methotrexate in children with acute lymphoblastic leukaemia. Clin Pharmacokinet 45(12):1227–1238
Piard C, Bressolle F, Fakhoury M, Zhang D, Yacouben K, Rieutord A, Jacqz-Aigrain E (2007) A limited sampling strategy to estimate individual pharmacokinetic parameters of methotrexate in children with acute lymphoblastic leukemia. Cancer Chemother Pharmacol 60(4):609–620
Rousseau A, Sabot C, Delepine N, Delepine G, Debord J, Lachatre G, Marquet P (2002) Bayesian estimation of methotrexate pharmacokinetic parameters and area under the curve in children and young adults with localised osteosarcoma. Clin Pharmacokinet 41(13):1095–1104
Moricke A, Reiter A, Zimmermann M, Gadner H, Stanulla M, Dordelmann M, Loning L, Beier R, Ludwig WD, Ratei R, Harbott J, Boos J, Mann G, Niggli F, Feldges A, Henze G, Welte K, Beck JD, Klingebiel T, Niemeyer C, Zintl F, Bode U, Urban C, Wehinger H, Niethammer D, Riehm H, Schrappe M (2008) Risk-adjusted therapy of acute lymphoblastic leukemia can decrease treatment burden and improve survival: treatment results of 2169 unselected pediatric and adolescent patients enrolled in the trial ALL-BFM 95. Blood 111(9):4477–4489
Reiter A, Schrappe M, Ludwig WD, Hiddemann W, Sauter S, Henze G, Zimmermann M, Lampert F, Havers W, Niethammer D et al (1994) Chemotherapy in 998 unselected childhood acute lymphoblastic leukemia patients. Results and conclusions of the multicenter trial ALL-BFM 86. Blood 84(9):3122–3133
ALL IC-BFM 2002–Trial Steering Committee (2002) A randomized trial of the I-BFM-SG for the management of childhood non-B acute lymphoblastic leukemia final version of therapy protocol from May 3, 2002. ALL IC-BFM 2002–Trial Steering Committee
Reiter A, Tiemann M, Ludwig WD, Wacker HH, Yakisan E, Schrappe M, Henzler D, Sykora KW, Brandt A, Odenwald E et al (1994) NHL-BFM 90 therapy study in treatment of malignant non-Hodgkin’s lymphomas in children and adolescents. Part 1: classification and allocation to strategic therapy groups. BIF study group. Klin Pädiatr 206(4):222–233
Miller AB, Hoogstraten B, Staquet M, Winkler A (1981) Reporting results of cancer—treatment. Cancer 47(1):207–214
Miller SA, Dykes DD, Polesky HF (1988) A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acids Res 16(3):1215–1215
Shaw GM, Lammer EJ, Zhu HP, Baker MW, Neri E, Finnell RH (2002) Maternal periconceptional vitamin use, genetic variation of infant reduced folate carrier (A80G), and risk of spina bifida. Am J Med Genet 108(1):1–6
Jacques PF, Bostom AG, Selhub J, Rich S, Ellison RC, Eckfeldt JH, Gravel RA, Rozen R (2003) Effects of polymorphisms of methionine synthase and methionine synthase reductase on total plasma homocysteine in the NHLBI Family Heart Study. Atherosclerosis 166(1):49–55
Matsuo K, Suzuki R, Hamajima N, Ogura M, Kagami Y, Taji H, Kondoh E, Maeda S, Asakura S, Kaba S, Nakamura S, Seto M, Morishima Y, Tajima K (2001) Association between polymorphisms of folate- and methionine-metabolizing enzymes and susceptibility to malignant lymphoma. Blood 97(10):3205–3209
Iacopetta B, Grieu F, Joseph D, Elsaleh H (2001) A polymorphism in the enhancer region of the thymidylate synthase promoter influences the survival of colorectal cancer patients treated with 5-fluorouracil. Br J Cancer 85(6):827–830
Kurzawski M, Pawlik A, Gornik W, Drodzik M (2006) Frequency of common MDR1 gene variants in a Polish population. Pharmacol Rep 58(1):35–40
Grabar PB, Logar D, Lestan B, Dolzan V (2008) Genetic determinants of methotrexate toxicity in rheumatoid arthritis patients: a study of polymorphisms affecting methotrexate transport and folate metabolism. Eur J Clin Pharmacol 64(11):1057–1068
Bergstrand M, Hooker AC, Wallin JE, Karlsson MO (2009) Prediction corrected visual predictive checks. Paper presented at the American Conference on Pharmacometrics (ACoP), Mashantucket, CT, USA. Available at: http://2009.go-acop.org/acop2009/posters. Accessed 24 Feb 2011
Bohanec Grabar P, Jazbec J, Dolzan V (2007) Gene-gene interactions in the folate metabolic pathway influence the risk for acute lymphoblastic leukemia in children. Leuk Lymphoma 48(4):786–792
Pui CH, Evans WE (2006) Treatment of acute lymphoblastic leukemia. N Engl J Med 354(2):166–178
Cunningham L, Aplenc R (2008) Role of the folate-pathway and the thymidylate synthase genes in pediatric acute lymphoblastic leukemia treatment response. In: Innocenti F (ed) Cancer drug discovery and development: genomics and pharmacogenomics in anticancer drug development and clinical response. Humana Press, Totowa, pp 299–309
Schmiegelow K (2009) Advances in individual prediction of methotrexate toxicity: a review. Br J Haematol 146(5):489–503
Gervasini G (2009) Polymorphisms in methotrexate pathways: what is clinically relevant, what is not, and what is promising. Curr Drug Metab 10(6):547–566
Kim YI (2009) Role of the MTHFR polymorphisms in cancer risk modification and treatment. Future Oncol 5(4):523–542
Relling MV, Fairclough D, Ayers D, Crom WR, Rodman JH, Pui CH, Evans WE (1994) Patient characteristics associated with high-risk methotrexate concentrations and toxicity. J Clin Oncol 12(8):1667–1672
Wall AM, Gajjar A, Link A, Mahmoud H, Pui CH, Relling MV (2000) Individualized methotrexate dosing in children with relapsed acute lymphoblastic leukemia. Leukemia 14(2):221–225
Faltaos DW, Hulot JS, Urien S, Morel V, Kaloshi G, Fernandez C, Xuan H, Leblond V, Lechat P (2006) Population pharmacokinetic study of methotrexate in patients with lymphoid malignancy. Cancer Chemother Pharmacol 58(5):626–633
Monjanel-Mouterde S, Lejeune C, Ciccolini J, Merite N, Hadjaj D, Bonnier P, Piana P, Durand A (2002) Bayesian population model of methotrexate to guide dosage adjustments for folate rescue in patients with breast cancer. J Clin Pharm Ther 27(3):189–195
Batey MA, Wright JG, Azzabi A, Newell DR, Lind MJ, Calvert AH, Boddy AV (2002) Population pharmacokinetics of adjuvant cyclophosphamide, methotrexate and 5-fluorouracil (CMF). Eur J Cancer 38(8):1081–1089
Laverdiere C, Chiasson S, Costea I, Moghrabi A, Krajinovic M (2002) Polymorphism G80A in the reduced folate carrier gene and its relationship to methotrexate plasma levels and outcome of childhood acute lymphoblastic leukemia. Blood 100(10):3832–3834
Imanishi H, Okamura N, Yagi M, Noro Y, Moriya Y, Nakamura T, Hayakawa A, Takeshima Y, Sakaeda T, Matsuo M, Okumura K (2007) Genetic polymorphisms associated with adverse events and elimination of methotrexate in childhood acute lymphoblastic leukemia and malignant lymphoma. J Hum Genet 52(2):166–171
Huang L, Tissing WJ, de Jonge R, van Zelst BD, Pieters R (2008) Polymorphisms in folate-related genes: association with side effects of high-dose methotrexate in childhood acute lymphoblastic leukemia. Leukemia 22(9):1798–1800
Kantar M, Kosova B, Cetingul N, Gumus S, Toroslu E, Zafer N, Topcuoglu N, Aksoylar S, Cinar M, Tetik A, Eroglu Z (2009) Methylenetetrahydrofolate reductase C677T and A1298C gene polymorphisms and therapy-related toxicity in children treated for acute lymphoblastic leukemia and non-Hodgkin lymphoma. Leuk Lymphoma 50(6):912–917
van Kooten Niekerk PB, Schmiegelow K, Schroeder H (2008) Influence of methylene tetrahydrofolate reductase polymorphisms and coadministration of antimetabolites on toxicity after high dose methotrexate. Eur J Haematol 81(5):391–398
Kishi S, Griener J, Cheng C, Das S, Cook EH, Pei D, Hudson M, Rubnitz J, Sandlund JT, Pui CH, Relling MV (2003) Homocysteine, pharmacogenetics, and neurotoxicity in children with leukemia. J Clin Oncol 21(16):3084–3091
Seidemann K, Book M, Zimmermann M, Meyer U, Welte K, Stanulla M, Reiter A (2006) MTHFR 677 ©– > T) polymorphism is not relevant for prognosis or therapy-associated toxicity in pediatric NHL: results from 484 patients of multicenter trial NHL-BFM 95. Ann Hematol 85(5):291–300
Shimasaki N, Mori T, Samejima H, Sato R, Shimada H, Yahagi N, Torii C, Yoshihara H, Tanigawara Y, Takahashi T, Kosaki K (2006) Effects of methylenetetrahydrofolate reductase and reduced folate carrier 1 polymorphisms on high-dose methotrexate-induced toxicities in children with acute lymphoblastic leukemia or lymphoma. J Pediatr Hematol /Oncol 28(2):64–68
Shimasaki N, Mori T, Torii C, Sato R, Shimada H, Tanigawara Y, Kosaki K, Takahashi T (2008) Influence of MTHFR and RFC1 polymorphisms on toxicities during maintenance chemotherapy for childhood acute lymphoblastic leukemia or lymphoma. J Pediatr Hematol/Oncol 30(5):347–352
Faganel Kotnik B, Dolzan V, Grabnar I, Jazbec J (2010) Relationship of the reduced folate carrier gene polymorphism G80A to methotrexate plasma concentration, toxicity, and disease outcome in childhood acute lymphoblastic leukemia. Leuk Lymphoma 51(4):724–726
Krajinovic M, Costea I, Primeau M, Dulucq S, Moghrabi A (2005) Combining several polymorphisms of thymidylate synthase gene for pharmacogenetic analysis. Pharmacogenomics J 5(6):374–380
Trevino LR, Shimasaki N, Yang W, Panetta JC, Cheng C, Pei D, Chan D, Sparreboom A, Giacomini KM, Pui CH, Evans WE, Relling MV (2009) Germline genetic variation in an organic anion transporter polypeptide associated with methotrexate pharmacokinetics and clinical effects. J Clin Oncol 27(35):5972–5978
Rau T, Erney B, Gores R, Eschenhagen T, Beck J, Langer T (2006) High-dose methotrexate in pediatric acute lymphoblastic leukemia: impact of ABCC2 polymorphisms on plasma concentrations. Clin Pharmacol Ther 80(5):468–476
Acknowledgements
This work was financially supported by the Slovenian Research Agency (ARRS Grants P1-0189, P1-0170 and P3-0343).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Barbara Faganel Kotnik and Iztok Grabnar contributed equally to the study
This study has been presented in part at the Population Approach Group Europe (PAGE) 2007 meeting in Copenhagen, Denmark (poster number: P3-11; available at: http://www.page-meeting.org/default.asp?abstract=1199).
Rights and permissions
About this article
Cite this article
Faganel Kotnik, B., Grabnar, I., Bohanec Grabar, P. et al. Association of genetic polymorphism in the folate metabolic pathway with methotrexate pharmacokinetics and toxicity in childhood acute lymphoblastic leukaemia and malignant lymphoma. Eur J Clin Pharmacol 67, 993–1006 (2011). https://doi.org/10.1007/s00228-011-1046-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-011-1046-z