Abstract
Objectives
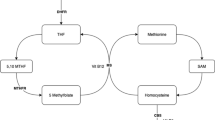
Several prospective studies reported that fibrates might increase blood total homocysteine (tHcy) concentrations. Because of this adverse effect, elevated tHcy could potentially compromise the cardiovascular benefit resulting from lipid-lowering by fibrates. In our study we aimed to find out whether the folate co-administration would modify the fibrate-induced elevation of tHcy.
Methods
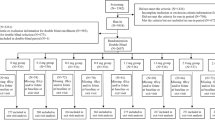
Twenty-four volunteers (m17, f7; mean age 54.9 years) with total cholesterol ≥6 mmol/l and triglycerides less than 5 mmol/l, with normal blood pressure, normal blood glucose and without any pharmacotherapy and/or clinical vascular or metabolic disease, were included in an open, randomised, prospective, crossover study. We measured lipids, tHcy, folate, vitamin B12 and renal function markers after diet, after a 6-month administration of 200 mg of fenofibrate (3 months in monotherapy followed by 3 months in combination with 10 mg of folate) and further on after an identical period of fluvastatin administration (3 months of 40 mg followed by 3 months of 80 mg).
Results
Fenofibrate in monotherapy, beside the expected lipid-lowering effect, increased tHcy from 10.0 to 14.2 μmol/l (p<0.001). Co-administration of folate decreased tHcy to 10.6 μmol/l. In contrast, fluvastatin did not significantly influence the tHcy concentrations.
Conclusion
Co-administration of folate to fenofibrate therapy has the potential to reverse the fibrate-induced elevation of tHcy.

Similar content being viewed by others
References
Lorgeril M, Salen P, Paillard F et al (1999) Lipid lowering drugs and homocysteine. Lancet 353:209–10
Dierkes J, Westphal S, Luley C (1999) Serum homocysteine increases after therapy with fenofibrate or bezafibrate. Lancet 354:219–20
Jonkers IJAM, de Man FHAF, Onkenhout W et al (1999) Implication of fibrate therapy for homocysteine. Lancet 354:1208
Goffin E, Jamar F, Desager JP (1999) Implication of fibrate therapy for homocysteine. Lancet 354:1209
Westphal S, Dierkes J, Luley C (2001) Effect of fenofibrate and gemfibrozil on plasma homocysteine. Lancet 358:39–40
Giral P, Bruckert E, Jacob N et al (2001) Homocysteine and lipid lowering agent. A comparison between atorvastatin and fenofibrate in patients with mixed hyperlipidemia. Atherosclerosis 154:421–3
Graham IM, Daly EL, Refsum HM et al (1997) Plasma homocysteine as a risk factor for vascular disease. JAMA 277:1775–81
Mayer jr O, Šimon J, Rosolová H, Racek J, Trefil L (1999) The effect of homocysteine on the coronary risk. Cor et Vasa 41:16–9
Nygard O, Nordrehaug JE, Refsum H, et al (1997) Plasma homocysteine and mortality in patients with coronary heart disease. N Engl J Med 337:230–6
Stubbs PJ, Al-Obaidi MK, Conroy RM et al (2002) Effect of Plasma Homocysteine Concentration on Early and Late Events in Patients With Acute Coronary Syndromes. Circulation 102:605–610
Shimakawa T, Nieto FJ, Malinow MR et al (1997) Vitamin intake: a possible determinant on plasma homocysteine among middle aged adults. Ann Epidemiol 4:285–293
Homocysteine lowering trialists collaboration (1998) Lowering blood homocysteine with folic acid based supplements meta-analysis of randomised trials. BMJ 316:894–8
Mayer jr O, Šimon J, Rosolová H et al (2002) The effects of folate supplementation on some coagulation parameters and oxidative status surrogates. Eur J Clin Pharm 58:1-5
Cífková R. Škodová Z, Lánská V et al (2000) Longitudinální trendy celkového a HDL-cholesterolu u reprezentativnţho vzorku populace České republiky (Longitudinal trends of total and HDL-cholesterol in a representative population sample of the Czech Republic). Vnitr Lek 46:501–505
Wood D, De Backer G, Faergeman D, Graham I, Mancia G, Pyörälä K (1998) Prevention of coronary heart disease in clinical practice. Recommendations of the Second Joint Task Force of European and other Societies on coronary prevention. Eur Heart J 19:1434–503
WHO (1978) A cooperative trial in the primary prevention of ischemic heart disease using clofibrate: report from the committee of principal investigators. Br Heart J 40:1069–118
The BIP study group (2000) Secondary prevention by raising HDL cholesterol and reducing triglycerides in patients with coronary artery. Circulation 102:21–7
Meade T, Zuhrie R, Cook C et al (2002) Bezafibrate in men with lower extremity arterial disease: randomised controlled trial. BMJ 325:1139–41
Rubins HB, Robins SJ, Collins D et al (1999) for Veterans Affairs High-Density Lipoprotein Cholesterol Intervention Trial Study Group. Gemfibrozil for the secondary prevention of coronary heart disease in men with low levels of high-density lipoprotein cholesterol. N Engl J Med 341:410–8
Frick MH, Elo O, Haapa K et al (1987) Helsinki Heart study: primary prevention trial with gemfibrozil in middle aged men with dyslipidemia. NEJM 317:1237–45
Frick MH, Sivänne M, Nieminen MS et al (1997) Prevention of angiographic progression of coronary and vein-graft atherosclerosis by gemfibrozil after coronary bypass surgery in men with low levels of HDL-cholesterol. Circulation 96:2137–43
Ericson CG, Nilson J, Grip L, Svane B et al (1997) Effect of bezafibrate treatment over five years on plaque causing 20 to 30% diameter narrowing (The BECAIT study). Am J Cardiol 80:1125–9
Hottelart C, El Esper N, Achard JM et al (1999) Le fenofibrate augmente la creatininemie mais n´alere pas le debit de filtration glomerulaire chez les patients presentant une unsuffisance renale moderee. Nephrologie 20:41–4
Acknowledgement
The study was supported by the Internal Grant Agency of the Czech Ministry of Health; grant No. 6817–3. Estimation of homocysteine was supported by the Diagnostic Division of Abbott Laboratories, Ltd., who provided the kits free-of-charge. The excellent technical assistance of Mrs. Jitka Nováková is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mayer, O., Šimon, J., Holubec, L. et al. Fenofibrate-induced hyperhomocysteinemia may be prevented by folate co-administration. Eur J Clin Pharmacol 59, 367–371 (2003). https://doi.org/10.1007/s00228-003-0616-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-003-0616-0