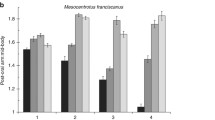
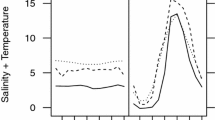
Abstract
Morphological plasticity is an adaptive response to heterogenous environments when a fitness advantage is conferred. Larval sea urchins, for example, are hypothesized to increase individual fitness in dilute feeding environments by elongating their feeding structure relative to their body size. Morphological plasticity for larval sea urchins is also coupled with significant shifts in the associated bacterial community, but whether this change to the associated microbiota occurs before, during, or following the expression of morphological plasticity is unclear. Using the sea urchin Lytechinus variegatus, we compare the temporal pattern of the microbial community and how it relates to the timing of morphological plasticity for larvae cultured in different food concentrations. From prefeeding through the expression of morphological plasticity, we observed that the microbiota associated with L. variegatus larvae exhibit a four-stage successional pattern, where changes in this community shift prior to the expression of the environment-specific morphology. The high food treatment, which results in the short-arm phenotype, deviated from the microbial trajectory of larval siblings prior to measurable morphological plasticity. These data suggest that a holobiont may exhibit shifts in the associated bacterial community corresponding with variation in the feeding environment that could occur in anticipation of or prior to morphological plasticity.




Similar content being viewed by others
References
Adams DK, Sewell MA, Angerer RC, Angerer LM (2011) Rapid adaptation to food availability by a dopamine-mediated morphogenetic response. Nat Commun 2:592
Agrawal AA (2001) Phenotypic plasticity in the interactions and evolution of species. Science 294:321–326
Amir A, McDonald D, Navas-Molina JA, Kopylova E, Morton JT, Xu ZZ, Kightley EP, Thompson LR, Hyde ER, Gonzalez A, Knight R (2017) Deblur rapidly resolves single-nucleotide community sequence patterns. mSystems 2:e00191-00116
Bates JM, Mittge E, Kuhlman J, Baden KN, Cheesman SE, Guillemin K (2006) Distinct signals from the microbiota promote different aspects of zebrafish gut differentiation. Dev Biol 297:374–386
Bittleston LS, Wolock CJ, Yahya BE, Chan XY, Chan KG, Pierce NE, Pringle A (2018) Convergence between the microcosms of Southeast Asian and North American pitcher plants. eLife 7:e36741
Boidron-Metairon IF (1988) Morphological plasticity in laboratory-reared echinoplutei of Dendraster excentricus (Eschscholtz) and Lytechinus variegatus (Lamarck) in response to food conditions. J Exp Mar Biol Ecol 119:31–41
Bolyen E, Rideout JR, Dillon MR, Bokulich NA, Abnet C, Al-Ghalith GA, Alexander H, Alm EJ, Arumugam M, Asnicar F, Bai Y, Bisanz JE, Bittinger K, Brejnrod A, Brislawn CJ, Brown CT, Callahan BJ, Caraballo-Rodríguez AM, Chase J, Cope E, Da Silva R, Dorrestein PC, Douglas GM, Durall DM, Duvallet C, Edwardson CF, Ernst M, Estaki M, Fouquier J, Gauglitz JM, Gibson DL, Gonzalez A, Gorlick K, Guo J, Hillmann B, Holmes S, Holste H, Huttenhower C, Huttley G, Janssen S, Jarmusch AK, Jiang L, Kaehler B, Kang KB, Keefe CR, Keim P, Kelley ST, Knights D, Koester I, Kosciolek T, Kreps J, Langille MG, Lee J, Ley R, Liu Y, Loftfield E, Lozupone C, Maher M, Marotz C, Martin BD, McDonald D, McIver LJ, Melnik AV, Metcalf JL, Morgan SC, Morton J, Naimey AT, Navas-Molina JA, Nothias LF, Orchanian SB, Pearson T, Peoples SL, Petras D, Preuss ML, Pruesse E, Rasmussen LB, Rivers A, Robeson MS II, Rosenthal P, Segata N, Shaffer M, Shiffer A, Sinha R, Song SJ, Spear JR, Swafford AD, Thompson LR, Torres PJ, Trinh P, Tripathi A, Turnbaugh PJ, Ul-Hasan S, van der Hooft JJ, Vargas F, Vázquez-Baeza Y, Vogtmann E, von Hippel M, Walters W, Wan Y, Wang M, Warren J, Weber KC, Williamson CH, Willis AD, Xu ZZ, Zaneveld JR, Zhang Y, Zhu Q, Knight R, Caporaso JG (2019) Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nat Biotechnol 37:852–857
Bordenstein SR, Theis KR (2015) Host biology in light of the microbiome: ten principles of holobionts and hologenomes. PLoS Biol 13:e1002226
Brooks AW, Kohl KD, Brucker RM, van Opstal EJ, Bordenstein SR (2016) Phylosymbiosis: relationships and functional effects of microbial communities across host evolutionary history. PLoS Biol 14:e2000225
Burke B, Steinberg P, Rusch D, Kjelleberg S, Thomas T (2011) Bacterial community assembly based on functional genes rather than species. Proc Natl Acad Sci USA 108:14288–14293
Byrne M, Sewell MA, Prowse TAA (2008) Nutritional ecology of sea urchin larvae: influence of endogenous and exogenous nutrition on echinopluteal growth and phenotypic plasticity in Tripneustes gratilla. Funct Ecol 22:643–648
Caporaso JG, Kuczynski J, Stombaugh J, Bittinger K, Bushman FD, Costello EK, Fierer N, Pena AG, Goodrich JK, Gordon JI, Huttley GA, Kelley ST, Knights D, Koenig JE, Ley RE, Lozupone CA, McDonald D, Muegge BD, Pirrung M, Reeder J, Sevinsky JR, Turnbaugh PJ, Walters WA, Widmann J, Yatsunenko T, Zaneveld J, Knight R (2010) QIIME allows analysis of high-throughput community sequencing data. Nat Methods 7:335–336
Carrier TJ, Reitzel AM (2017) The hologenome across environments and the implications of a host-associated microbial repertoire. Front Microbiol 8:802
Carrier TJ, Reitzel AM (2018) Convergent shifts in host-associated microbial communities across environmentally elicited phenotypes. Nat Commun 9:952
Carrier TJ, Reitzel AM (2019a) Bacterial community dynamics during embryonic and larval development of three confamilial echinoids. Mar Ecol Prog Ser 611:179–188
Carrier TJ, Reitzel AM (2019b) Shift in bacterial taxa precedes morphological plasticity in a larval echinoid. BioRxiv. https://doi.org/10.1101/640953
Carrier TJ, King BL, Coffman JA (2015) Gene expression changes associated with the developmental plasticity of sea urchin larvae in response to food availability. Biol Bull 228:171–180
Carrier TJ, Macrander J, Reitzel AM (2018a) A microbial perspective on the life-history evolution of marine invertebrate larvae: if, where, and when to feed. Mar Ecol 39:e12490
Carrier TJ, Wolfe K, Lopez K, Gall M, Janies DA, Byrne M (2018b) Diet-induced shifts in the crown-of-thorns (Acanthaster sp.) larval microbiome. Mar Biol 165:157
Carrier TJ, Dupont S, Reitzel AM (2019) Geographic location and food availability offer differing levels of influence on the bacterial communities associated with larval sea urchins. FEMS Microbiol Ecol 95:fiz103
DeWitt TJ, Sih A, Wilson DS (1998) Costs and limits of phenotypic plasticity. Trends Ecol Evol 13:77–81
Fenaux L, Strathmann MF, Strathmann RR (1994) Five tests of food-limited growth of larvae in coastal waters by comparison of rates of development and form of echinoplutei. Limnol Oceanogr 39:84–98
Fraune S, Anton-Erxleben F, Augustin R, Franzenburg S, Knop M, Schröder K, Willoweit-Ohl D, Bosch TCG (2015) Bacteria-bacteria interactions within the microbiota of the ancestral metazoan Hydra contribute to fungal resistance. ISME J 9:1543–1556
Gilbert SF, Sapp J, Tauber AI (2012) A symbiotic view of life: we have never been individuals. Q Rev Biol 87:325–341
Guillard RRL (1975) Culture of phytoplankton for feeding marine invertebrates. In: Smith WL, Chanley MH (eds) Culture of marine invertebrate animals. Plenum Press, New York
Hart MW, Strathmann RR (1994) Functional consequences of phenotypic plasticity in echinoid larvae. Biol Bull 186:291–299
Klindworth A, Pruesse E, Schweer T, Peplies J, Quast C, Horn M, Glockner FO (2013) Evaluation of general 16S ribosomal RNA gene PCR primers for classical and next-generation sequencing-based diversity studies. Nucleic Acids Res 41:e1
Kohl KD, Carey HV (2016) A place for host-microbe symbiosis in the comparative physiologist’s toolbox. J Exp Biol 219:3496–3504
Kohl KD, Amaya J, Passement CA, Dearing MD, McCue MD (2014) Unique and shared responses of the gut microbiota to prolonged fasting: a comparative study across five classes of vertebrate hosts. FEMS Microbiol Ecol 90:883–894
Levins R (1968) Evolution in changing environments. Princeton University Press, Princeton
Louca S, Jacques SMS, Pires APF, Leal JS, Srivastava DS, Parfrey LW, Farjalla VF, Doebeli M (2016) High taxonomic variability despite stable functional structure across microbial communities. Nat Ecol Evol 1:15
Lozupone C, Knight R (2005) UniFrac: a new phylogenetic method for comparing microbial communities. Appl Environ Microbiol 71:8228–8235
Manahan DT, Davis JP, Stephens GC (1993) Bacteria-free sea urchin larvae: selective uptake of neutral amino acids from seawater. Science 220:204–206
McAlister JS (2007) The long arm of the larva: evolutionary responses to resource availability. University of North Carolina, Chapel Hill
McAlister JS, Miner BG (2018) Phenotypic plasticity of feeding structures in marine invertebrate larvae. In: Carrier TJ, Reitzel AM, Heyland A (eds) Evolutionary ecology of marine invertebrate larvae. Oxford University Press, Oxford
McEdward LR, Herrera JC (1999) Body form and skeletal skeletal morphometrics during larval development of the sea urchin Lytechinus variegatus. J Exp Mar Biol Ecol 232:151–176
McFall-Ngai M, Hadfield MG, Bosch TCG, Carey HV, Domazet-Loso T, Douglas AE, Dubilier N, Eberl G, Fukami T, Gilbert SF, Hentschel U, King N, Kjelleberg S, Knoll AH, Kremer N, Mazmanian SK, Metcalf JL, Nealson K, Pierce NE, Rawls JF, Reid A, Ruby EG, Rumpho M, Sanders JG, Tautz D, Wernegreen JJ (2013) Animals in a bacterial world, a new imperative for the life sciences. Proc Natl Acad Sci USA 110:3229–3236
Mileikovsky SA (1971) Types of larval development in marine bottom invertebrates, their distribution and ecological significance: a re-evalution. Mar Biol 10:193–213
Miner BG (2004) Evolution of feeding structure plasticity in marine invertebrate larvae: a possible trade-off between arm length and stomach size. J Exp Mar Biol Ecol 315:117–125
Miner BG, Vonesh JR (2004) Effects of fine grain environmental variability on morphological plasticity. Ecol Lett 7:794–801
Miner BG, Sultan SE, Morgan SG, Padilla DK, Relyea RA (2005) Ecological consequences of phenotypic plasticity. Trends Ecol Evol 20:685–692
Moeller AH, Caro-Quintero A, Mjungu D, Georgiev AV, Lonsdorf EV, Muller MN, Pusey AE, Peeters M, Hahn BH, Ochman H (2016) Cospeciation of gut microbiota with hominids. Science 353:380–382
Murillo-Rincon AP, Klimovich A, Pemöller E, Taubenheim J, Mortzfeld B, Augustin R, Bosch TCG (2017) Spontaneous body contractions are modulated by the microbiome of Hydra. Sci Rep 7:15937
Olson RR, Olson MH (1989) Food limitation of planktotrophic marine invertebrate larvae: does it control recruitment success? Annu Rev Ecol Syst 20:225–247
Padilla DK, Adolph SC (1996) Plastic inducible morphologies are not always adaptive: the importance of time delays in a stocastic environment. Evol Ecol 10:105–117
Palumbi SR (1984) Tactics of acclimation: morphological changes of sponges in an unpredictable envrionment. Science 225:1478–1480
Parker BJ, Brisson JA (2019) A laterally transferred viral gene modifies aphid wing plasticity. Curr Biol 29:2098–2103
Pauley G, Boring L, Strathmann RR (1985) Food limited growth and development of larvae: experiments with natural sea water. J Exp Mar Biol Ecol 93:1–10
Podolsky RD, McAlister JS (2005) Developmental plasticity in Macrophiothrix brittlestars: are morphologically convergent larvae also convergently plastic? Biol Bull 209:127–138
Quast C, Pruesse E, Yilmaz P, Gerken J, Schweer T, Yarza P, Peplies J, Glockner FO (2013) The SILVA ribosomal RNA gene database project: improved data processing and web-based tools. Nucleic Acids Res 41:590–596
Rendleman A, Rodriguez J, Ohanian A, Pace D (2018) More than morphology: differences in food ration drive physiological plasticity in echinoid larvae. J Exp Mar Biol Ecol 501:1–15
Rognes T, Flouri T, Nichols B, Quince C, Mahé F (2016) VSEARCH: a versatile open source tool for metagenomics. PeerJ 4:e2584
Roth-Schulze AJ, Pintado J, Zozaya-Valdés E, Cremades J, Ruiz P, Kjelleberg S, Thomas T (2018) Functional biogeography and host specificity of bacterial communities associated with the marine green alga Ulva spp. Mol Ecol 27:1952–1965
Schneider C, Rasband W, Eliceiri K (2012) NIH Image to ImageJ: 25 years of image analysis. Nat Methods 9:671–675
Sewell MA (2005) Utilization of lipids during early development of the sea urchin Evechinus chloroticus. Mar Ecol Prog Ser 304:133–142
Sewell MA, Cameron MJ, McArdle BH (2004) Developmental plasticity in larval development in the echinometrid sea urchin Evechinus chloroticus with varying food rations. J Exp Mar Biol Ecol 309:219–237
Smith K, McCoy KD, Macpherson AJ (2007) Use of axenic animals in studying the adaptation of mammals to their commensal intestinal microbiota. Semin Immunol 19:59–69
Soars NA, Prowse TAA, Byrne M (2009) Overview of phenotypic plasticity in echinoid larvae, ‘Echinopluteus transversus’ type vs. typical echinoplutei. Mar Ecol Prog Ser 383:113–125
Sterns S (1989) The evolutionary significance of phenotypic plasticity. Bioscience 39:436–445
Strathmann MF (1987) Reproduction and development of marine invertebrates of the northern Pacific coast: data and methods for the study of eggs, embryos, and larvae. University of Washington Press, Washington
Strathmann RR, Fenaux L, Strathmann MF (1992) Heterochronic developmental plasticity in larval sea urchins and its implications for evolution of nonfeeding larvae. Evolution 46:972–986
Sun X-J, Li Q (2013) The effect of food availability on development and phenotypic plasticity in larvae of the sea cucumber (Apostichopus japonicus). Invertebr Reprod Dev 57:255–263
Wein T, Dagan T, Fraune S, Bosch TCG, Reusch TBH, Hülter NF (2018) Carrying capacity and colonization dynamics of Curvibacter in the Hydra host habitat. Front Microbiol 9:443
West-Eberhard M (2003) Developmental plasticity and evolution. Oxford University Press, Oxford
Williams EA, Carrier TJ (2018) An-omics perspective on marine invertebrate larvae. In: Carrier TJ, Reitzel AM, Heyland A (eds) Evolutionary ecology of marine invertebrate larvae. Oxford University Press, Oxford, pp 288–304
Zilber-Rosenberg I, Rosenberg E (2008) Role of microorganisms in the evolution of animals and plants: the hologenome theory of evolution. FEMS Microbiol Rev 32:723–735
Acknowledgements
We thank Daniel Rittschof (Duke Univ.) for welcoming this project into his laboratory; Beatriz Orihuela (Duke Univ.) for endless logistical assistance; Josh Osterberg (Duke Univ.) for collecting adult urchins; Karen Lopez (UNC Charlotte) for technical assistance with sequencing; Daniel Janies (UNC Charlotte) for sequencing resources; Justin McAlister (College of the Holy Cross) and Jason Hodin (Univ. Washington) for discussions on morphological plasticity in echinoid larvae; and the two reviewers for providing critical feedback to an earlier version of this manuscript.
Funding
This work was supported by an NSF Graduate Research Fellowship to TJC, a Human Frontier Science Program Award to AMR (RGY0079/2016), and a North Carolina Sea Grant award to AMR and TJC (2016-R/MG-1604).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest. We do, however, note that a preprint of this manuscript is available on bioRxiv (see, Carrier and Reitzel 2019b).
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. This article does not contain any studies with human participants performed by any of the authors.
Additional information
Responsible Editor: S. Uthicke.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Reviewed by S. Russell and an undisclosed expert.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Carrier, T.J., Reitzel, A.M. Shift in bacterial taxa precedes morphological plasticity in a larval echinoid. Mar Biol 166, 164 (2019). https://doi.org/10.1007/s00227-019-3613-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-019-3613-2