Abstract
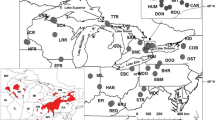
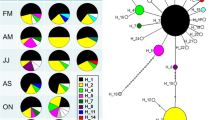
We evaluated stock structure and demographic (population) history of red snapper (Lutjanus campechanus) in the northern Gulf of Mexico (Gulf) via analysis of mitochondrial (mt)DNA sequences from 360 individuals sampled from four cohorts (year classes) at three localities across the northern Gulf. Exact tests of genetic homogeneity and analysis of molecular variance both among cohorts within localities and among localities were non-significant. Nested clade analysis provided evidence of different temporal episodes of both range expansion and restricted gene flow due to isolation by distance. A mismatch distribution of pairwise differences among mtDNA haplotypes and a maximum-likelihood coalescence analysis indicated a population expansion phase that dated to the Pleistocene and probably represents (re)colonization of the continental shelf following glacial retreat. The spatial distribution of red snapper in the northern Gulf appears to have a complex history that likely reflects glacial advance/retreat, habitat availability and suitability, and hydrology. Habitat availability/suitability and hydrology may partially restrict gene flow among present-day red snapper in the northern Gulf and give rise to a metapopulation structure with variable demographic connectivity. This type of population structure may be difficult to detect with commonly used, selectively neutral genetic markers.




Similar content being viewed by others
References
Allen GR (1985) Snappers of the world. FAO Species Catalogue, vol. 6. Food and Agriculture Organization of the United Nations, Rome
Bermingham E, McCafferty SS, Martin AP (1997) Fish biogeography and molecular clocks: perspectives from the Panamanian Isthmus. In: Kocher TD, Stepien CA (eds) Molecular systematics of fishes. Academic, San Diego, CA, pp 113–128
Bielawski J P, Gold JR (2002) Mutation patterns of mitochondrial H- and L-strand DNA in closely related cyprinid fishes. Genetics 161:1589–1597
Bortone SA, Chapman RW (1995) Identification of stock structure and recruitment patterns for the red snapper, Lutjanus campechanus, in the Gulf of Mexico. Final Report for Marfin Program Grant no. NA17FF0379-03. SE Regional Office, National Marine Fisheries Service, St. Petersburg, FL
Camper JD, Barber RC, Richardson LR, Gold JR (1993) Mitochondrial DNA variation among red snapper (Lutjanus campechanus) from the Gulf of Mexico. Mol Mar Biol Biotechnol 3:154–161
Carvalho GR, Hauser L (1995) Molecular genetics and the stock concept in fisheries. In: Carvalho, GR, Pitcher TJ (eds) Molecular genetics in fisheries. Chapman and Hall, London, pp 55–80
Christman MC (1997) Peer review of red snapper (Lutjanus campechanus) research and management in the Gulf of Mexico: statistics review. Office of Science and Technology, NOAA, Silver Spring, MD
Clement M, Posada D, Crandall KA (2000) TCS: a computer program to estimate gene genealogies. Mol Ecol 9:1657–1660
Cowen RK, Lwiza KMM, Sponaugle S, Paris CB, Olson DB (2000). Connectivity of marine populations: open or closed? Science 287:857–859
Fable WA (1980) Tagging studies of red snapper (Lutjanus campechanus) and vermilion snapper (Rhomboplites aurorubens) off the south Texas coast. Contrib Mar Sci 23:115–121
Ferber D (2001) Keeping the Stygian waters at bay. Science 291:968–973
Fischer AJ, Baker, MS Jr, Wilson CA (2004) Red snapper, Lutjanus campechanus, demographic structure in the northern Gulf of Mexico based on spatial patterns in growth rates and morphometrics. Fish Bull 102:593–603
Fu Y-X (1997) Statistical tests of neutrality of mutations against population growth, hitchhiking, and background selection. Genetics 147:915–925
Fu Y-X, Li W-H (1993) Statistical tests of neutral mutations. Genetics 133:693–709
Garber, AF, Tringali MD, Stuck KC (2004) Population structure and variation in red snapper (Lutjanus campechanus) from the Gulf of Mexico and Atlantic Coast of Florida as determined from mitochondrial DNA control region sequence. Mar Biotechnol 6:175–185
Gold JR, Richardson LR (1998) Genetic homogeneity among geographic samples of snappers and groupers: evidence of continuous gene flow? Proc Gulf Caribbean Res Inst 50:709–726
Gold JR, Sun F, Richardson LR (1997) Population structure of red snapper from the Gulf of Mexico as inferred from analysis of mitochondrial DNA. Trans Am Fish Soc 126:386–396
Gold JR, Pak E, Richardson LR (2001) Microsatellite variation among red snapper (Lutjanus campechanus) from the Gulf of Mexico. Mar Biotechnol 3:293–304
Goodyear CP (1995) Red snapper stocks in U.S. waters of the Gulf of Mexico. National Marine Fisheries Service, SE Fisheries Center, Miami Laboratory, CRD 95/96–05, Miami, FL
Goodyear CP, Phares P (1990) Status of red snapper stocks of the Gulf of Mexico: report for 1990. National Marine Fisheries Service, SE Fisheries Center, Miami Laboratory, CRD 89/90–05, Miami, FL.
Hauser L, Ward RD (1998) Population identification in pelagic fish: the limits of molecular markers. In: Carvalho GR (ed) Advances in molecular ecology. IOS, Amsterdam, pp 2191–2242
Hoese HD, Moore RH (1977) Fishes of the Gulf of Mexico. Texas A&M University Press, College Station, TX
Kritzer JP, Sale PF (2004) Metapopulation ecology in the sea: from Levin’s model to marine ecology and fisheries science. Fish Fisheries 5:131–140
Kuhner MK, Yamato J, Felsenstein J (1998) Maximum likelihood estimation of population growth rates based on the coalescent. Genetics 149:429–434
Leis JM (1987) Review of the early life history of tropical groupers (Serranidae) and snappers (Lutjanidae). In: Polovina JJ, Ralston S (eds) Tropical groupers and snappers: biology and fisheries management. Westview, Boulder, CO, pp 189–237
Levins R (1969) Some demographic and genetic consequences of environmental heterogeneity for biological control. Bull Entomol Soc Am 15:237–240
Levins R (1970) Extinction. In: Desternhaber M (ed) Some mathematical problems in biology. Am Math Soc, Providence, RI, pp 77–107
Li Y, Nowlin WD Jr, Reid RO (1997) Mean hydrographic fields and their interannual variability over the Texas–Louisiana continental shelf in spring, summer, and fall. J Geophys Res 102:1027–1049
Maddison DR, Maddison WP (2000) macclade 4: analysis of phylogeny and character evolution, ver 4.0. Sinauer, Sunderland, Mass.
Morey SL, Schroeder WL, O’Brien JJ, Zavala-Hidalgo. J (2003) The annual cycle of riverine influence in the eastern Gulf of Mexico. Geophys Res Lett 30: OCE 5.1–5.4
Nei M (1987) Molecular evolutionary genetics. Columbia University Press, New York
Palumbi SR (1994) Reproductive isolation, genetic divergence, and speciation in the sea. Annu Rev Ecol Syst 25:547–572
Palumbi SR (1996a) Nucleic acids II: the polymerase chain reaction. In: Hillis DM, Moritz C, Mable BK (eds) Molecular systematics. Sinauer, Sunderland, MA, pp 205–247
Palumbi SR (1996b) Macrospatial genetic structure and speciation in marine taxa with high dispersal abilities. In: Ferraris F, Palumbi SR (eds) Molecular zoology: advances, strategies and protocols. Wiley, New York, pp 101–117
Parker RP, Morrison JM, Nowlin WD Jr (1979) Surface drift data from the Caribbean Sea and Gulf of Mexico, 1975–1978. ONR no. N00014-75-C-0537, Department of Oceanography, Technical Report no. 79-8-T, Texas A&M University, College Station, TX
Patterson WF, Watterson JC, Shipp RL, Cowan JH Jr (2001) Movement of tagged red snapper in the northern Gulf of Mexico. Trans Am Fish Soc 130:533–545
Posada D, Crandall KA (1998) modeltest: testing the model of DNA substitution. Bioinformatics 14:817–818
Posada D, Crandall KA, Templeton AR (2000) geodis: a program for the cladistic nested analysis of the geographical distribution of genetic haplotypes. Mol Ecol 9:487–488
Rabalais NN, Turner RE, Justic D, Dortch Q, Wiseman WJ (1999) Characterization of hypoxia: topic I report for the integrated assessment on hypoxia in the Gulf of Mexico. NOAA Coastal Ocean Program Decision Analysis Series No. 15. NOAA Coastal Ocean Program, Silver Spring, MD
Rezak R, Bright TJ, McGrail DW (1985) Reefs and banks of the northwestern Gulf of Mexico. Wiley, New York
Rogers AR (1995) Genetic evidence for a Pleistocene population expansion. Evolution 49:608–615
Rogers AR, Harpending H (1992) Population growth makes waves in the distribution of pair-wise genetic differences. Mol Biol Evol 9:552–569
Rowe GT, Chapman P (2002) Continental shelf hypoxia: some nagging questions. Gulf Mexico Sci 2002:153–160
Rozas J, Sánchez-Delbarrio JC, Messeguer X, Rozas R (2003) DnaSP, DNA polymorphism analyses by the coalescent and other methods. Bioinformatics 19:2496–2497
Saillant E, Gold JR (2002) Genetic studies of red snapper (Lutjanus campechanus) in the northern Gulf of Mexico. Proc Gulf Caribbean Res Inst 55:811–820
Sambrook JE, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY
Schneider S, Roessli D, Excoffier L (2000) arlequin ver. 2000: a software for population genetic data analysis. Genetics and Biometry Laboratory, University of Geneva, Geneva
Shackleton NJ, Opdyke ND (1973) Oxygen isotope and paleomagnetic stratigraphy of equatorial Pacific core V28-V238: oxygen isotope temperatures and ice volumes on a 105 and 103 year scale. Quat Res 3:39–55
Shulman MJ, Bermingham E (1995) Early life histories, ocean currents, and the population genetics of Caribbean reef fishes. Evolution 49:897–910
Swofford DL (2001) PAUP* 4.0b: phylogenetic analysis using parsimony. Sinauer, Sunderland, MA
Szedlmayer ST (1997) Ultrasonic telemetry of red snapper, Lutjanus campechanus, at artificial reef sites in the northeast Gulf of Mexico. Copeia 1997:846–850
Szedlmayer ST, Shipp RL (1994) Movement and growth of red snapper, Lutjanus campechanus, from an artificial reef area in the northeastern Gulf of Mexico. Bull Mar Sci 55:887–896
Templeton AR (1998) Nested clade analysis of phylogeographic data: testing hypotheses about gene flow and population history. Mol Ecol 7:381–397
Templeton AR, Sing CF (1993) A cladistic analysis of phenotypic associations with haplotypes inferred from restriction endonuclease mapping. IV. Nested analyses with cladogram uncertainty and recombination. Genetics 134:659–669
Templeton AR, Boerwinkle E, Sing CF (1987) A cladistic analysis of phenotypic associations with haplotypes inferred from restriction endonuclease mapping. I. Basic theory and an analysis of alcohol dehydrogenase activity in Drosophila. Genetics 117:343–351
Templeton AR, Routman E, Phillips CA (1995) Separating population structure from population history: a cladistic analysis of the geographical distribution of mitochondrial DNA haplotypes in the tiger salamander, Ambystoma tigrinum. Genetics 140:767–782
Watterson G (1975) On the number of segregating sites in genetical models without recombination. Theoret Pop Biol 7:256–276
Williams M, Dunkerley D, De Deckker P, Kershaw P, Chappell J (1998) Quaternary environments, 2nd edn. Arnold, London
Wilson CA, Nieland DL (2001) Age and growth of red snapper, Lutjanus campechanus, from the northern Gulf of Mexico off Louisiana. Fish Bull 99:653–664
Woods MK, Fischer AJ, Cowan JH Jr, Nieland DL (2003) Size and age at maturity of female red snapper Lutjanus campechanus in the northern Gulf of Mexico. Proc Gulf Caribbean Res Inst 54:526–537
Acknowledgements
Work was supported in part by an award from the Gulf and South Atlantic Fisheries Development Foundation (Project 70-04-20000/11824), the National Marine Fisheries Service (Marfin Project 87-FF-0426), and the Texas Agricultural Experiment Station Project H-6703). Views expressed in the paper are those of the authors and do not necessarily reflect views of the sponsors. This paper is no. 40 in the series ‘Genetic Studies in Marine Fishes’ and contribution no. 130 of the Center for Biosystematics and Biodiversity at Texas A&M University.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by P.W. Sammarco, Chauvin
Rights and permissions
About this article
Cite this article
Pruett, C.L., Saillant, E. & Gold, J.R. Historical population demography of red snapper (Lutjanus campechanus) from the northern Gulf of Mexico based on analysis of sequences of mitochondrial DNA. Marine Biology 147, 593–602 (2005). https://doi.org/10.1007/s00227-005-1615-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00227-005-1615-8