Abstract
Osteoporosis is an important global health problem resulting in fragility fractures. The vertebrae are the commonest site of fracture resulting in extreme illness burden, and having the highest associated mortality. International studies show that vertebral fractures (VF) increase in prevalence with age, similarly in men and women, but differ across different regions of the world. Ireland has one of the highest rates of hip fracture in the world but data on vertebral fractures are limited. In this study we examined the prevalence of VF and associated major risk factors, using a sample of subjects who underwent vertebral fracture assessment (VFA) performed on 2 dual-energy X-ray absorptiometry (DXA) machines. A total of 1296 subjects aged 40 years and older had a valid VFA report and DXA information available, including 254 men and 1042 women. Subjects had a mean age of 70 years, 805 (62%) had prior fractures, mean spine T-score was − 1.4 and mean total hip T-scores was − 1.2, while mean FRAX scores were 15.4% and 4.8% for major osteoporotic fracture and hip fracture, respectively. Although 95 (7%) had a known VF prior to scanning, 283 (22%) patients had at least 1 VF on their scan: 161 had 1, 61 had 2, and 61 had 3 or more. The prevalence of VF increased with age from 11.5% in those aged 40–49 years to > 33% among those aged ≥ 80 years. Both men and women with VF had significantly lower BMD at each measured site, and significantly higher FRAX scores, P < 0.01. These data suggest VF are common in high risk populations, particularly older men and women with low BMD, previous fractures, and at high risk of fracture. Urgent attention is needed to examine effective ways to identify those at risk and to reduce the burden of VF.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Osteoporosis is one of the most common diseases worldwide resulting in millions of fragility fractures each year. 50% of women and 20–25% of men over the age of 50 years will experience an osteoporosis-related fracture, which are associated with significant morbidity, healthcare costs and increased mortality [1,2,3]. Although vertebral fractures (VF) represent the most frequently affected skeletal site, their impact is not fully appreciated compared with other skeletal sites [4, 5], due to their different risk factors, presentation and diagnostic criteria, and the use of vague or unclear terminology [6,7,8,9,10,11].
Epidemiologic studies of VF are heterogeneous, varying within and across different regions [6, 11,12,13,14,15,16]. However, their prevalence is broadly similar to hip fractures rising exponentially with age [6, 10, 13], in both men and women [6, 10,11,12, 17]. Their illness burden for patients and healthcare systems is also similar, but greater than forearm fractures [2,3,4,5,6,7,8,9,10, 18, 19]. Specific populations are at increased risk including those with rheumatic diseases [20,21,22,23], taking glucocorticoid therapy [24,25,26,27,28] or with previous fractures [13, 29,30,31].
Recent publications on the prevalence, incidence and impact of VF in Ireland highlight a paucity of studies [29, 32]. Reasons include under-representation, under-diagnosis and limited data from non-hospital settings [29, 32,33,34]. Reports suggest hospital length of stay is similar to hip fractures [33], while others show VF are common when imaging studies are systematically evaluated [29, 34]. Our experience suggests the prevalence and importance of VF are under-appreciated by patients, medical staff, healthcare managers and government.
Performance of vertebral fracture assessment (VFA) scans are recommended by the International Society for Clinical Densitometry (ISCD) for patients at risk [35]. In practice, these scans assess the prevalence amongst individuals [10, 13, 17, 34, 35] or the general population [17], or those of particular interest, such as rheumatoid arthritis [34] or fracture liaison service referrals [36]. In this study, we used available VFA scans from a sample of patients in the West of Ireland to gain a better appreciation of the prevalence of VF in men and women and to explore associations with conventional major risk factors, Bone Mineral Density (BMD) and FRAX scores.
Methods
Data were extracted from 2 GE Lunar central DXA machines at a single centre as previously described [37], following approval from the institutional clinical research ethics committee (C.A. 2109). The committee granted a waiver of informed consent was granted for this study. These were saved, anonymised and subsequently available for analysis. The original cohort includes a sample of > 36,000 unique individuals with medical histories, medications and an array of DXA parameters. In this study, we include only subjects aged 40 years and older who had a VFA scan and available report from 1 of 2 GE Lunar machines at 1 centre over a 12-month period. A prior audit of > 7000 DXA referrals for our centre shows 69% are referred from primary care, 27% from hospital specialist clinics and 4% from inpatient services. Referrals are accepted per ISCD indications, or sent back to the referring clinician if more information is required or there is no appropriate indication [38]. Accepted referrals are then prioritised as ‘urgent’, ‘soon’, or ‘routine’ depending on the information provided and our impression of where the results are most likely to have the greatest clinical impact. For example, someone referred following a major fragility fracture and who is not on treatment would be ‘urgent’, while ‘screening’ for a postmenopausal woman aged 50 years with no other major risk factors would be considered routine. Waiting times for routine scans are currently 10 years, urgent scans are now < 3 months.
VFA scans are performed per ISCD indications (women aged > 70 years, men > 80 years, adults taking chronic glucocorticoid therapy, reporting height loss and reported but undocumented previous VF; the one difference being we do not apply the recommended T-score threshold) by 1 of 2 ISCD trained nurse specialists (RE and KG). All are read by a single ISCD trained and certified clinician experienced in reporting DXA and VFA scans (JJC) as per ISCD recommendations using the Genant semi-quantitative method for fracture ascertainment [35, 39, 40]. As part of our routine practice, all VFA scans are reviewed by our DXA nurse specialists at the time of scanning, and later by the reporting clinician. An outline of this process is as follows:
-
(1)
Trained DXA nurse specialists perform VFA scans and flag all patients with a known VF or an abnormal VFA scan with a suspected fracture for urgent review by the reporting clinician.
-
(2)
All VFA scans are reviewed and reported by the clinician routinely (other abnormalities are flagged for review, e.g., osteoarthritis, foreign body or other artefact).
-
(3)
Discrepancies, unusual or unclear scans are discussed at a weekly multi-disciplinary team audit with the lead clinician and 4 nurse specialists running the hospital’s DXA and Fracture Liaison Service (FLS). Agreement between our readers is currently >90% for moderate–severe fractures.
-
(4)
Additional imaging including X-rays, CT and MRI scans and reports are reviewed as necessary to verify anomalies or the presence of fractures. VFA report agreement is >70% for mild fractures.
-
(5)
Our FLS prioritises patients not on treatment with prevalent spine and hip fractures. Patients with multiple VF at the time of scanning are offered an immediate consult following their scan if a nurse specialist or clinician is available. Others are subsequently contacted as a priority, and offered an evaluation and treatment for osteoporosis or advised to follow-up urgently with their primary care doctor.
-
(6)
A copy of the DXA report highlighting the fractures and an FLS letter are sent to the patient’s doctor.
Demographic data were summarised as follows; age, gender, height, weight, BMI, primary and secondary indication for DXA scan, prior fracture, osteoporosis medication if prescribed, calcium and vitamin D supplementation, DXA T-scores, FRAX® variables, and FRAX® scores. DXA T-scores for all men and women are calculated using NHANES III reference data for white females [14]. We chose to classify all subjects as osteoporotic or not using ISCD criteria for older men and postmenopausal women, i.e., lowest T-score ≤ − 2.5 [14]. FRAX® scores were calculated by GE software FRAX® tool. VFA indications, scan technique and recommended reporting methods were performed as recommended by ISCD [35, 40]. Data for the entire cohort were summarised, compared by gender and by decade to enable comparison to publications from other countries [11, 12, 15,16,17]. The results of the VFA analysis are recorded as ‘fracture’ or ‘no fracture’, and also include the site for the first 2 fractures. If patients had > 2 VF sites, they were simply recorded as ‘multiple’. Details on fracture type and severity, or results of other imaging are not available in this dataset.
All analyses were performed using R statistical software, version 3.6.1. For categorical variables, we report subject count and percentage, and used Pearson’s Chi-squared test (if the number of subjects in each comparative group was ≥ 5) or Fisher’s exact test (if there were fewer than 5 subjects in a comparison group) to examine statistically significant differences between groups. For continuous variables, normality was assessing using Shapiro–Wilk’s test. We then calculated mean and standard deviation and used unpaired Student’s t tests to compare groups whose data were normally distributed, while for non-parametric data, we used data are expressed as medians and interquartile ranges, while Wilcoxon’s rank sum test examined for statistically significant differences between two groups, and Kruskal–Wallis test was used for comparing multiple groups. All significance tests carried out were two-sided, and P values < 0.05 were considered significant.
Results
1296 subjects had a VFA scan report and DXA information available for analysis including 1042 women and 254 men. Details of our entire cohort has previously been published [37, 41], while a summary of the characteristics of this sub-group are shown in Table 1. The majority are female, with a mean age of 70 years, and a range of 40–94 years. Women were significantly lighter and smaller than men. The majority of men and women had a prior fracture and a substantial proportion were taking corticosteroids, had rheumatoid arthritis, a family history of osteoporosis or another illness or medication predisposing to osteoporosis (Table 1). All had at least 1 skeletal site available for analysis of BMD; spine: 1063 (82.0%), total hip and femoral neck: 1234 (95.2%) and 1/3 distal radius: 3 (0.2%). 1217 (93.9%) had GE Lunar FRAX scores for major osteoporotic fracture and hip fracture. 35% of patients were taking osteoporosis medication, 93.3% of whom were taking anti-resorptive medication. 6% were taking teriparatide, alone or in combination with an anti-resorptive, while 0.7% were taking other medication combinations.
409 (31.6%) of the 1296 subjects had a DXA classification (T-score ≤ − 2.5) of osteoporosis (Table 1). The prevalence of osteoporosis increased steadily with age, particularly for women, from 16.1% in those aged 40–49 years, to 41.3% amongst those 80 years and older (Table 2). Mean BMD was significantly lower for women than men, and the proportion of women classified as osteoporotic by DXA was almost twice that of men (Table 1). Women had higher mean fracture risk scores than men (Table 1). Although the 10-year risk of fracture (GE Lunar FRAX®) increased with age for both genders, women had higher scores at each age category (Table 2).
805 patients (62.1%) had a prior fracture (Table 3). Hip fractures are more common in men, while wrist/forearm fractures are more common in women (both P < 0.001). The prevalence of spine, humerus, and other fractures is similar between both genders. Almost 1 in 3 patients have multiple (> 1) fractures. 95 (7.3%) patients had a known VF prior to scanning, 84 (88%) of whom had a visible reported fracture on their scan. An additional 199 (15.4%) patients had a VF visible on their VFA scan.
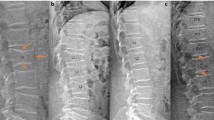
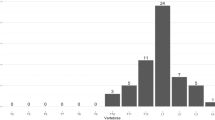
283 (21.8%) patients had at least 1 VF on their VFA scan, including 225 (21%) women and 61 (23%) of men. 152 had a single fracture, and a further 61 had 3 or more fractures. The sites for one fracture are shown in Fig. 1, while the sites and frequency of multiple fractures are shown in Fig. 2. The proportion of subjects with 1, 2 and 3 or more VF was similar across both genders (Women vs. Men: 11.2% vs. 12.8%, 4.5% vs. 4.9% and 4.5% vs. 4.9%, respectively). The prevalence of VF increased with age, a pattern which is significantly more striking in women (Table 4). Although men experienced an increase in VF with age, this trend was offset by the high proportion of men aged 40–49 years with VF.
Table 5 summarises the association between the presence of VF, classification of osteoporosis, fracture risk and major risk factors for fracture. The majority of VF (57%) occurred in individuals with a T-score > − 2.5 (Table 5). However, subjects with a DXA classification as osteoporosis (lowest T-score ≤ − 2.5) were more likely to have VF compared to subjects without: 29.8% vs. 18.8% (P < 0.001). Not surprisingly, 10-year fracture risk scores were significantly higher amongst those with VF than those without (Table 5). People with VF were also older, lighter and more likely to have a prior fracture, height loss and to be on osteoporosis treatment. Interestingly, people with VF were also less likely to be taking corticosteroids or have other causes of osteoporosis.
Discussion
In this study examining lateral DXA spine scans, we found VF are common in people with or at high risk for fracture or with established osteoporosis. The prevalence of VF increased with age, particularly in women. The prevalence of VF in this population was actually 3 times higher than their known VF prevalence prior to their VFA scan, many of whom had 2 or more VF. Although the majority of the study’s cohort did not meet the DXA threshold to be classified as osteoporotic, the presence of VF was significantly associated with lower BMD and higher fracture risk scores. These data have important implications for considering VFA scans in Irish adults undergoing DXA.
VF represents a major problem in osteoporosis care, and the commonest site of injury [2, 6, 9, 10, 13, 17]. Clinical fractures have a similar impact on quality of life, morbidity and mortality to hip fractures [4, 9, 19]. Unlike other fractures, the presence and importance of VF are greatly under-appreciated [6,7,8, 10, 12, 13, 29]. General population studies from Europe, China, Latin and North America show VF prevalence increases with age in both men and women, irrespective of which criteria are used [6, 11, 12, 15,16,17]. VF are more prevalent in our study than general population studies, because a VFA scan was performed on the basis of risk factors [14, 35]. Thus, a large proportion already have osteoporosis and fractures, are older, have rheumatoid arthritis, or are taking corticosteroid medications, all of which represent significant risk factors [1, 10, 13, 34].
Rheumatic diseases are associated with a higher risk of osteoporotic fracture, particularly rheumatoid arthritis and ankylosing spondylitis [20,21,22,23, 34, 42]. The risk of VF in ankylosing spondylitis is almost threefold higher [20, 22] which is similar to rheumatoid arthritis [21, 42]. 14% of our study population have rheumatoid (14%), which is much greater than the population prevalence. Glucocorticoids are commonly used to treat rheumatic diseases and increase the risk of osteoporotic fracture, particularly with higher doses [24,25,26,27,28, 43]. Clinical trials of glucocorticoid-induced osteoporosis show a strikingly high prevalence of osteoporotic fracture even in populations with normal or almost normal BMD [25,26,27,28, 43]. The prevalence of VF at baseline ranges from 10 to 36% in those with normal BMD [26, 28, 43] and 13% to 37% in those with low BMD [25, 28]. Patients with a prior fracture are at much greater risk of fracture [29,30,31], and although osteoporosis treatment is effective, fractures can still occur [25,26,27,28, 43]. In our study, patients with VF were more likely to have a prior fracture (85% vs. 56%), have osteoporosis by DXA criteria (43% vs. 28%), and be taking osteoporosis treatment (52% vs. 31%).
In this study, we found the prevalence of fractures was threefold higher than the reported prevalence prior to scanning, perhaps reflecting the nature of these fractures, where only around 1 in 3 present with clinical symptoms [6, 10, 13]. Unfortunately, we do not have clinical information on the presence or absence of back pain or other symptoms in this dataset. These results suggest the spine is the commonest site of fracture in this population, which is under-represented in published data for our country [29, 33]. 70% of people in this study did not have a DXA T-score below the osteoporosis threshold. This is in line with others [17], and a well described epidemiologic phenomenon [44]. Although some believe fractures in people whose BMD is not below the fracture threshold do not need treatment, a diagnosis of osteoporosis can be made and treatment offered in the presence of a major osteoporotic fracture and the absence of major trauma or other explanations [14, 32, 45]. Others have shown the risk of subsequent fracture is significantly higher in patients with low BMD and prevalent fractures than those with low BMD alone [30, 31].
Our study has important strengths and limitations. Prior studies in European populations have not included subjects from Ireland [11], and gaps remain in our understanding of the epidemiology and utility and applicability of diagnostic and risk tools for our populations [32]. Our study includes more than 1000 adults referred for VFA scanning, all of whom met ISCD indications for a scan. Many, therefore, had established osteoporosis, a prior fracture or were at high risk for fracture, so our results are not generalizable to the Irish population, such as those described by others [11, 12, 15,16,17], or our whole cohort [37].
The sample contains few men compared to some larger studies and likely reflects selection bias, particularly those 40–49 years of age. A prior study of a cohort of rheumatoid arthritis (RA) patients reported VF prevalence using this technology [34], but there are limited data on other populations and more than 85% of our population did not have RA. Clinicians and nurses were not blinded to the patients’ diagnoses and DXA results which could have resulted in ascertainment bias. Fracture type and grade were not available, nor the dose and duration of corticosteroids, which are further important limitations. We are conservative in diagnosing mild fractures as per ISCD recommendations [35, 40], and may have over or under-estimated the true prevalence of VF using this approach. Reassuringly agreement between our department VFA readings and other radiology reports is 88% for those with a known VF. Others have shown the prevalence changes substantially depending on which imaging method, analysis and criteria are employed [8, 11, 12]. We have also shown that a formal process to review available images results in a significant increase in fracture diagnosis [29, 34].
In our experience the presence and importance of VF for patients is greatly under-appreciated. Patients are often dismissed by health professionals when presenting with milder symptoms before they finally obtain imaging to confirm their diagnosis. In practice when the results could change patient management and there is doubt about the diagnosis, additional imaging should be sought or reviewed. Our results show that in persons deemed at increased risk for vertebral fracture, VFA scanning significantly increased the diagnosis of vertebral fractures, the presence of which is under-appreciated. This could have significant implications for the diagnosis and management of patients with osteoporosis at significant risk of subsequent fracture.
Summary and Conclusions
In this study, we show that VF are common in high risk Irish adults, many of whom are unaware of their presence or who do not meet a DXA threshold for osteoporosis. Careful consideration should be given to performing VFA scans in high risk populations such as this.
References
Compston JE, McClung MR, Leslie WD (2019) Osteoporosis. Lancet 393:364–376
Borgstrom F, Karlsson L, Ortsater G, Norton N, Halbout P, Cooper C et al (2020) Fragility fractures in Europe: burden, management and opportunities. Arch Osteoporos 15:59
Singer A, Exuzides A, Spangler L, O’Malley C, Colby C, Johnston K et al (2015) Burden of illness for osteoporotic fractures compared with other serious diseases among postmenopausal women in the United States. Mayo Clin Proc 90:53–62
Cauley JA, Thompson DE, Ensrud KC, Scott JC, Black D (2000) Risk of mortality following clinical fractures. Osteoporos Int 11:556–561
Lau E, Ong K, Kurtz S, Schmier J, Edidin A (2008) Mortality following the diagnosis of a vertebral compression fracture in the Medicare population. J Bone Joint Surg Am 90:1479–1486
Ballane G, Cauley JA, Luckey MM, El-Hajj Fuleihan G (2017) Worldwide prevalence and incidence of osteoporotic vertebral fractures. Osteoporos Int 28:1531–1542
Costa AG, Wyman A, Siris ES, Watts NB, Silverman S, Saag KG et al (2013) When, where and how osteoporosis-associated fractures occur: an analysis from the global longitudinal study of osteoporosis in women (GLOW). PLoS ONE 8:e83306
Lentle BC, Hg Oei E, Goltzman D, Rivadeneira F, Hammond I, Oei L et al (2018) Vertebral fractures and morphometric deformities. J Bone Miner Res 33:1544–1545
Nevitt MC, Thompson DE, Black DM, Rubin SR, Ensrud K, Yates AJ et al (2000) Effect of alendronate on limited-activity days and bed-disability days caused by back pain in postmenopausal women with existing vertebral fractures. Fracture intervention trial research group. Arch Intern Med 160:77–85
Schousboe JT (2016) Epidemiology of Vertebral Fractures. J Clin Densitom 19:8–22
O’Neill TW, Felsenberg D, Varlow J, Cooper C, Kanis JA, Silman AJ (1996) The prevalence of vertebral deformity in European men and women: the European vertebral osteoporosis study. J Bone Miner Res 11:1010–1018
Jackson SA, Tenenhouse A, Robertson L (2000) Vertebral fracture definition from population-based data: preliminary results from the Canadian multicenter osteoporosis study (CaMos). Osteoporos Int 11:680–687
Kendler DL, Bauer DC, Davison KS, Dian L, Hanley DA, Harris ST et al (2016) Vertebral fractures: clinical importance and management. Am J Med 129:221.e1-221.e10
Schousboe JT, Shepherd JA, Bilezikian JP, Baim S (2013) Executive summary of the 2013 international society for clinical densitometry position development conference on bone densitometry. J Clin Densitom 16:455–466
Clark P, Cons-Molina F, Deleze M, Ragi S, Haddock L, Zanchetta JR et al (2009) The prevalence of radiographic vertebral fractures in Latin American countries: the Latin American vertebral osteoporosis study (LAVOS). Osteoporos Int 20:275–282
Cui L, Chen L, Xia W, Jiang Y, Cui L, Huang W et al (2017) Vertebral fracture in postmenopausal Chinese women: a population-based study. Osteoporos Int 28:2583–2590
Cosman F (2017) Spine fracture prevalence in a nationally representative sample of US women and men aged >/=40 years: results from the national health and nutrition examination survey (NHANES) 2013–2014–-supplementary presentation. Osteoporos Int 28:2319–2320
Cauley JA, Lui LY, Paudel ML, Taylor BC, Cawthon PM, Hillier TA et al (2016) Impact of radiographic vertebral fractures on inpatient healthcare utilization in older women. Bone 88:165–169
Svedbom A, Borgstom F, Hernlund E, Strom O, Alekna V, Bianchi ML et al (2018) Quality of life for up to 18 months after low-energy hip, vertebral, and distal forearm fractures-results from the ICUROS. Osteoporos Int 29:557–566
Geusens P, De Winter L, Quaden D, Vanhoof J, Vosse D, van den Bergh J et al (2015) The prevalence of vertebral fractures in spondyloarthritis: relation to disease characteristics, bone mineral density, syndesmophytes and history of back pain and trauma. Arthritis Res Ther 17:294
Wright NC, Lisse JR, Walitt BT, Eaton CB, Chen Z, Women’s Health Initiative (2011) Arthritis increases the risk for fractures–results from the women’s health initiative. J Rheumatol 38:1680–1688
Cooper C, Carbone L, Michet CJ, Atkinson EJ, O’Fallon WM, Melton LJ 3rd (1994) Fracture risk in patients with ankylosing spondylitis: a population based study. J Rheumatol 21:1877–1882
Weiss RJ, Wick MC, Ackermann PW, Montgomery SM (2010) Increased fracture risk in patients with rheumatic disorders and other inflammatory diseases—a case-control study with 53,108 patients with fracture. J Rheumatol 37:2247–2250
van Staa TP, Leufkens HG, Abenhaim L, Zhang B, Cooper C (2000) Oral corticosteroids and fracture risk: relationship to daily and cumulative doses. Rheumatology 39:1383–1389
Reid DM, Hughes RA, Laan RF, Sacco-Gibson NA, Wenderoth DH, Adami S et al (2000) Efficacy and safety of daily risedronate in the treatment of corticosteroid-induced osteoporosis in men and women: a randomized trial. European corticosteroid-induced osteoporosis treatment study. J Bone Miner Res 15:1006–1013
Reid DM, Devogelaer JP, Saag K, Roux C, Lau CS, Reginster JY et al (2009) Zoledronic acid and risedronate in the prevention and treatment of glucocorticoid-induced osteoporosis (HORIZON): a multicentre, double-blind, double-dummy, randomised controlled trial. Lancet 373:1253–1263
Saag KG, Shane E, Boonen S, Marin F, Donley DW, Taylor KA et al (2007) Teriparatide or alendronate in glucocorticoid-induced osteoporosis. N Engl J Med 357:2028–2039
Saag KG, Pannacciulli N, Geusens P, Adachi JD, Messina OD, Morales-Torres J et al (2019) Denosumab versus risedronate in glucocorticoid-induced osteoporosis: final results of a twenty-four-month randomized, double-blind, double-dummy trial. Arthritis Rheumatol 71:1174–1184
Kelly MA, McCabe E, Bergin D, Kearns SR, McCabe JP, Armstrong C et al (2020) Osteoporotic vertebral fractures are common in hip fracture patients and are under-recognized. J Clin Densitom. https://doi.org/10.1016/j.jocd.2020.05.007
Lindsay R, Silverman SL, Cooper C, Hanley DA, Barton I, Broy SB et al (2001) Risk of new vertebral fracture in the year following a fracture. JAMA 285:320–323
Ross PD, Davis JW, Epstein RS, Wasnich RD (1991) Pre-existing fractures and bone mass predict vertebral fracture incidence in women. Ann Intern Med 114:919–923
McCabe E, Ibrahim A, Singh R, Kelly M, Armstrong C, Heaney F et al (2020) A systematic review of the Irish osteoporotic vertebral fracture literature. Arch Osteoporos 15:34
Kelly MA, McGowan B, McKenna MJ, Bennett K, Carey JJ, Whelan B et al (2018) Emerging trends in hospitalisation for fragility fractures in Ireland. Ir J Med Sci 187:601–608
Mohammad A, Lohan D, Bergin D, Mooney S, Newell J, O’Donnell M et al (2014) The prevalence of vertebral fracture on vertebral fracture assessment imaging in a large cohort of patients with rheumatoid arthritis. Rheumatology 53:821–827
Borges JLC, da Sousa Silva M, Ward RJ, Diemer KM, Yeap SS, Lewiecki EM (2019) Repeating vertebral fracture assessment: 2019 ISCD official position. J Clin Densitom 22:484–488
Lems WF, Paccou J, Zhang J, Fuggle NR, Chandran M, Harvey NC et al (2021) Vertebral fracture: epidemiology, impact and use of DXA vertebral fracture assessment in fracture liaison services. Osteoporos Int 32:399–411
Erjiang E, Wang T, Yang L, Dempsey M, Brennan A, Yu M et al (2020) The Irish dual-energy X-ray absorptiometry (DXA) health informatics prediction (HIP) for Osteoporosis Project. BMJ Open 10:e040488
Mohammad A, Aamir MU, Mooney S, Coughlan RJ, Carey JJ (2014) Appropriateness of referrals to a tertiary referral centre for bone mineral density testing. Ir J Med Sci 183:533–537
Genant HK, Wu CY, van Kuijk C, Nevitt MC (1993) Vertebral fracture assessment using a semiquantitative technique. J Bone Miner Res 8:1137–1148
Rosen HN, Vokes TJ, Malabanan AO, Deal CL, Alele JD, Olenginski TP et al (2013) The official positions of the international society for clinical densitometry: vertebral fracture assessment. J Clin Densitom 16:482–488
Erjiang E, Wang T, Yang L, Dempsey M, Brennan A, Yu M et al (2020) Machine learning can improve clinical detection of low BMD: the DXA-HIP study. J Clin Densitom. https://doi.org/10.1016/j.jocd.2020.10.004
van Staa TP, Geusens P, Bijlsma JW, Leufkens HG, Cooper C (2006) Clinical assessment of the long-term risk of fracture in patients with rheumatoid arthritis. Arthritis Rheum 54:3104–3112
Cohen S, Levy RM, Keller M, Boling E, Emkey RD, Greenwald M et al (1999) Risedronate therapy prevents corticosteroid-induced bone loss: a twelve-month, multicenter, randomized, double-blind, placebo-controlled, parallel-group study. Arthritis Rheum 42:2309–2318
Siris ES, Chen YT, Abbott TA, Barrett-Connor E, Miller PD, Wehren LE et al (2004) Bone mineral density thresholds for pharmacological intervention to prevent fractures. Arch Intern Med 164:1108–1112
Cosman F, de Beur SJ, LeBoff MS, Lewiecki EM, Tanner B, Randall S et al (2014) Clinician’s guide to prevention and treatment of osteoporosis. Osteoporos Int 25:2359–2381
Funding
Open Access funding provided by the IReL Consortium.
Author information
Authors and Affiliations
Contributions
JC and LY performed the analysis, interpretation of data and prepared the first draft of the paper. EE and TW contributed to the analysis and the interpretation of data. KG, RE, CA, FH and EM contributed to the acquisition of the data. All authors revised the paper critically for intellectual content and approved the final version. All authors agree to be accountable for the work and to ensure that any questions relating to the accuracy and integrity of the paper are investigated and properly resolved.
Corresponding author
Ethics declarations
Conflict of interest
John Carey, Lan Yang, Erjiang E, Tingyan Wang, Kelly Gorham, Rebecca Egan, Attracta Brennan, Mary Dempsey, Catherine Armstrong, Fiona Heaney, Eva McCabe and Ming Yu declare that they have no conflict of interest.
Ethical Approval
The collection and analysis of data was approved by our institutional Clinical Research Ethics Committee: C.A. 2109.
Research Involving Human Rights
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed Consent
The committee granted a waiver of informed consent was granted for this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Carey, J.J., Yang, L., Erjiang, E. et al. Vertebral Fractures in Ireland: A Sub-analysis of the DXA HIP Project. Calcif Tissue Int 109, 534–543 (2021). https://doi.org/10.1007/s00223-021-00868-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-021-00868-7