Abstract
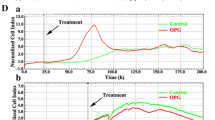
Osteoclasts originate from the hematopoietic stem cell and share a differentiation pathway with the cells of the monocyte/macrophage lineages. Development and activation of osteoclasts, and as a consequence regulation of bone resorption, depend on two growth factors: macrophage colony-stimulating factor and receptor activator of NF-κB ligand. Furthermore, cell development and activity are modulated by a microenvironment composed of cytokines and growth factors and of the extracellular matrix. Membrane transporters are a means for cells to interact with their environment. Within this study, the expression of proteins regulating cellular iron homeostasis in osteoclast-like cells grown from bone marrow-derived progenitors was compared to the expression of this set of proteins by monocyte/macrophage lineage cells. In differentiating osteoclasts, levels of transcripts encoding transferrin receptor 1 and divalent metal transporter 1 (Slc11A2) were increased, while levels of transcripts encoding ferroportin (Slc40A1) and natural resistance-associated macrophage protein 1 (Slc11A1) were decreased. Supplementation of the culture media with exogenous iron led to an increase in the proliferation of osteoclast progenitor cells and to the expression of a macrophage-like phenotype, while the development of osteoclasts was reduced. Upon transfer of mature OC onto a CaP substrate, iron depletion of the medium with the Fe3+-chelator Deferoxamine Mesylate decreased CaP dissolution by ~30 %, which could be restored by addition of exogenous iron. During the 24 h of the assay, no effects were observed on total TRAP activity. The data demonstrate transcriptional regulation of the components of cellular iron transporters during OC development and suggests that iron homeostasis may contribute to fine-tuning of the RANKL-induced OC development.





Similar content being viewed by others
References
Atanga E, Dolder S, Dauwalder T, Wetterwald A, Hofstetter W (2011) TNFalpha inhibits the development of osteoclasts through osteoblast-derived GM-CSF. Bone 49:1090–1100
Balani D, Aeberli D, Hofstetter W, Seitz M (2013) Interleukin-17A stimulates granulocyte-macrophage colony-stimulating factor release by murine osteoblasts in the presence of 1,25-dihydroxyvitamin D(3) and inhibits murine osteoclast development in vitro. Arthritis Rheumatol 65:436–446
Balga R, Wetterwald A, Portenier J, Dolder S, Mueller C, Hofstetter W (2006) Tumor necrosis factor-alpha: alternative role as an inhibitor of osteoclast formation in vitro. Bone 39:325–335
Bruzzaniti A, Baron R (2006) Molecular regulation of osteoclast activity. Rev Endocr metabolic Disord 7:123–139
Choy J, Albers CE, Siebenrock KA, Dolder S, Hofstetter W, Klenke FM (2014) Incorporation of RANKL promotes osteoclast formation and osteoclast activity on beta-TCP ceramics. Bone 69:80–88
Eriksen EF (1986) Normal and pathological remodeling of human trabecular bone: three dimensional reconstruction of the remodeling sequence in normals and in metabolic bone disease. Endocr Rev 7:379–408
Felix R, Cecchini MG, Hofstetter W, Elford PR, Stutzer A, Fleisch H (1990) Impairment of macrophage colony-stimulating factor production and lack of resident bone marrow macrophages in the osteopetrotic op/op mouse. J Bone Miner Res 5:781–789
Felix R, Hofstetter W, Cecchini MG (1996) Recent developments in the understanding of the pathophysiology of osteopetrosis. Eur J Endocrinol/Eur Fed Endocr Soc 134:143–156
Fleming MD, Trenor CC 3rd, Su MA, Foernzler D, Beier DR, Dietrich WF et al (1997) Microcytic anaemia mice have a mutation in Nramp2, a candidate iron transporter gene. Nat Genet 16:383–386
Ganz T (2004) Hepcidin in iron metabolism. Curr Opin Hematol 11:251–254
Ganz T (2005) Cellular iron: ferroportin is the only way out. Cell Metab 1:155–157
Gengenbacher M, Sebald HJ, Villiger PM, Hofstetter W, Seitz M (2008) Infliximab inhibits bone resorption by circulating osteoclast precursor cells in patients with rheumatoid arthritis and ankylosing spondylitis. Ann Rheum Dis 67:620–624
Gunshin H, Mackenzie B, Berger UV, Gunshin Y, Romero MF, Boron WF et al (1997) Cloning and characterization of a mammalian proton-coupled metal-ion transporter. Nature 388:482–488
Hall TJ, Chambers TJ (1989) Optimal bone resorption by isolated rat osteoclasts requires chloride/bicarbonate exchange. Calcif Tissue Int 45:378–380
Ishii KA, Fumoto T, Iwai K, Takeshita S, Ito M, Shimohata N et al (2009) Coordination of PGC-1beta and iron uptake in mitochondrial biogenesis and osteoclast activation. Nat Med 15:259–266
Ishii M, Egen JG, Klauschen F, Meier-Schellersheim M, Saeki Y, Vacher J et al (2009) Sphingosine-1-phosphate mobilizes osteoclast precursors and regulates bone homeostasis. Nature 458:524–528
Jia P, Xu YJ, Zhang ZL, Li K, Li B, Zhang W et al (2012) Ferric ion could facilitate osteoclast differentiation and bone resorption through the production of reactive oxygen species. J Orthop Res 30:1843–1852
Katsumata S, Katsumata-Tsuboi R, Uehara M, Suzuki K (2009) Severe iron deficiency decreases both bone formation and bone resorption in rats. J Nutr 139:238–243
Kikuta J, Kawamura S, Okiji F, Shirazaki M, Sakai S, Saito H et al (2013) Sphingosine-1-phosphate-mediated osteoclast precursor monocyte migration is a critical point of control in antibone-resorptive action of active vitamin D. Proc Natl Acad Sci USA 110:7009–7013
Kimble RB, Matayoshi AB, Vannice JL, Kung VT, Williams C, Pacifici R (1995) Simultaneous block of interleukin-1 and tumor necrosis factor is required to completely prevent bone loss in the early postovariectomy period. Endocrinology 136:3054–3061
Kong YY, Yoshida H, Sarosi I, Tan HL, Timms E, Capparelli C et al (1999) OPGL is a key regulator of osteoclastogenesis, lymphocyte development and lymph-node organogenesis. Nature 397:315–323
Kornak U, Kasper D, Bosl MR, Kaiser E, Schweizer M, Schulz A et al (2001) Loss of the ClC-7 chloride channel leads to osteopetrosis in mice and man. Cell 104:205–215
Li J, Hou Y, Zhang S, Ji H, Rong H, Qu G et al (2013) Excess iron undermined bone load-bearing capacity through tumor necrosis factor-alpha-dependent osteoclastic activation in mice. Biomed Rep 1:85–88
Meynard D, Babitt JL, Lin HY (2014) The liver: conductor of systemic iron balance. Blood 123:168–176
Mizoguchi T, Muto A, Udagawa N, Arai A, Yamashita T, Hosoya A et al (2009) Identification of cell cycle-arrested quiescent osteoclast precursors in vivo. J Cell Biol 184:541–554
Muto A, Mizoguchi T, Udagawa N, Ito S, Kawahara I, Abiko Y et al (2011) Lineage-committed osteoclast precursors circulate in blood and settle down into bone. J Bone Miner Res 26:2978–2990
Parfitt AM (1982) The coupling of bone formation to bone resorption: a critical analysis of the concept and of its relevance to the pathogenesis of osteoporosis. Metab Bone Dis Relat Res 4:1–6
Parrow NL, Fleming RE (2014) Bone morphogenetic proteins as regulators of iron metabolism. Annu Rev Nutr 34:77–94
Pietrangelo A (2010) Hereditary hemochromatosis: pathogenesis, diagnosis, and treatment. Gastroenterology 139(393–408):e1–e2
Rizzoli R, Bonjour JP, Ferrari SL (2001) Osteoporosis, genetics and hormones. J Mol Endocrinol 26:79–94
Roodman GD (2009) Osteoclasts pump iron. Cell Metab 9:405–406
Schaller S, Henriksen K, Sveigaard C, Heegaard AM, Helix N, Stahlhut M et al (2004) The chloride channel inhibitor NS3736 [corrected] prevents bone resorption in ovariectomized rats without changing bone formation. J Bone Miner Res 19:1144–1153
Svedbom A, Ivergard M, Hernlund E, Rizzoli R, Kanis JA (2014) Epidemiology and economic burden of osteoporosis in Switzerland. Arch Osteoporos. 9:187
Toxqui L, Vaquero MP (2015) Chronic iron deficiency as an emerging risk factor for osteoporosis: a hypothesis. Nutrients. 7:2324–2344
Tsay J, Yang Z, Ross FP, Cunningham-Rundles S, Lin H, Coleman R et al (2010) Bone loss caused by iron overload in a murine model: importance of oxidative stress. Blood 116:2582–2589
Vaananen HK, Karhukorpi EK, Sundquist K, Wallmark B, Roininen I, Hentunen T et al (1990) Evidence for the presence of a proton pump of the vacuolar H(+)-ATPase type in the ruffled borders of osteoclasts. J Cell Biol 111:1305–1311
Weinberg ED (2008) Role of iron in osteoporosis. Pediatr Endocrinol Rev 6(Suppl 1):81–85
Wolf M, White KE (2014) Coupling fibroblast growth factor 23 production and cleavage: iron deficiency, rickets, and kidney disease. Curr Opin Nephrol Hypertens 23:411–419
Worthen CA, Enns CA (2014) The role of hepatic transferrin receptor 2 in the regulation of iron homeostasis in the body. Front Pharmacol 5:34
Yang Q, Jian J, Abramson SB, Huang X (2011) Inhibitory effects of iron on bone morphogenetic protein 2-induced osteoblastogenesis. J Bone Miner Res 26:1188–1196
Yun S, Vincelette ND (2015) Update on iron metabolism and molecular perspective of common genetic and acquired disorder, hemochromatosis. Crit Rev Oncol/Hematol 95:12–25
Acknowledgements
The authors wish to thank Dr. Antoinette Wetterwald for helpful advice and discussions and Mark Siegrist for excellent technical support during the course of this work. The study was supported by the Swiss National Center for Competence in Research (TransCure).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Wenjie Xie, Sebastian Lorenz, Silvia Dolder and Willy Hofstetter declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
All animal experiments were performed under the permit BE23/13 issued to WH by the Veterinary Office of the State of Bern.
Rights and permissions
About this article
Cite this article
Xie, W., Lorenz, S., Dolder, S. et al. Extracellular Iron is a Modulator of the Differentiation of Osteoclast Lineage Cells. Calcif Tissue Int 98, 275–283 (2016). https://doi.org/10.1007/s00223-015-0087-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-015-0087-1