Abstract
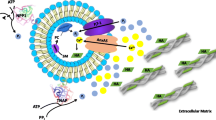
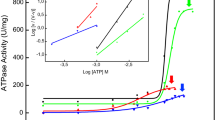
During endochondral bone formation, chondrocytes and osteoblasts synthesize and mineralize the extracellular matrix through a process that initiates within matrix vesicles (MVs) and ends with bone mineral propagation onto the collagenous scaffold. pH gradients have been identified in the growth plate of long bones, but how pH changes affect the initiation of skeletal mineralization is not known. Tissue-nonspecific alkaline phosphatase (TNAP) degrades extracellular inorganic pyrophosphate (PPi), a mineralization inhibitor produced by ectonucleotide pyrophosphatase/phosphodiesterase-1 (NPP1), while contributing Pi from ATP to initiate mineralization. TNAP and NPP1, alone or combined, were reconstituted in dipalmitoylphosphatidylcholine liposomes to mimic the microenvironment of MVs. The hydrolysis of ATP, ADP, AMP, and PPi was studied at pH 8 and 9 and compared to the data determined at pH 7.4. While catalytic efficiencies in general were higher at alkaline pH, PPi hydrolysis was maximal at pH 8 and indicated a preferential utilization of PPi over ATP at pH 8 versus 9. In addition, all proteoliposomes induced mineral formation when incubated in a synthetic cartilage lymph containing 1 mM ATP as substrate and amorphous calcium phosphate or calcium–phosphate–phosphatidylserine complexes as nucleators. Propagation of mineralization was significantly more efficient at pH 7.5 and 8 than at pH 9. Since a slight pH elevation from 7.4 to 8 promotes considerably more hydrolysis of ATP, ADP, and AMP primarily by TNAP, this small pH change facilitates mineralization, especially via upregulated PPi hydrolysis by both NPP1 and TNAP, further elevating the Pi/PPi ratio, thus enhancing bone mineralization.






Similar content being viewed by others
References
Boskey AL (2006) Mineralization, structure and function of bone. In: Seibel MJ, Robins SP, Biezikian JP (eds) Dynamics of bone and cartilage metabolism. Academic, San Diego, pp 201–212
Millán JL (2013) The role of phosphatases in the initiation of skeletal mineralization. Calcif Tissue Int. doi:10.1007/s00223-012-9672-8
Ali SY, Sajdera SW, Anderson HC (1970) Isolation and characterization of calcifying matrix vesicles from epiphyseal cartilage. Proc Natl Acad Sci USA 67:1513–1520
Robison R (1923) The possible significance of hexosephosphoric esters in ossification. Biochem J 17:286–293
Meyer JL (1984) Can biological calcification occur in the presence of pyrophosphate? Arch Biochem Biophys 231:1–8
Rezende AA, Pizauro JM, Ciancaglini P, Leone FA (1994) Phosphodiesterase activity is a novel property of alkaline phosphatase from osseous plate. Biochem J 301:517–522
Hessle L, Johnsson KA, Anderson HC et al (2002) Tissue-nonspecific alkaline phosphatase and plasma cell membrane glycoprotein-1 are central antagonistic regulators of bone mineralization. Proc Natl Acad Sci USA 99:9445–9449
Harmey D, Hessle L, Narisawa S et al (2004) Concerted regulation of inorganic pyrophosphate and osteopontin by Akp2, Enpp 1, and Ank. Am J Pathol 164:1199–1209
Terkeltaub RA (2001) Inorganic pyrophosphate generation and disposition in pathophysiology. Am J Physiol Cell Physiol 281:C1–C11
Huang R, Rosenbach M, Vaughn R et al (1994) Expression of the murine plasma cell nucleotide pyrophosphohydrolase PC-1 is shared by human liver, bone, and cartilage cells. Regulation of PC-1 expression in osteosarcoma cells by transforming growth factor-beta. J Clin Invest 94:560–567
Johnson K, Moffa A, Chen Y et al (1999) Matrix vesicle plasma cell membrane glycoprotein-1 regulates mineralization by murine osteoblastic MC3T3 cells. J Bone Miner Res 14:883–892
Gijsbers R, Ceulemans H, Stalmans W, Bollen M (2001) Structural and catalytic similarities between nucleotide pyrophosphatases/phosphodiesterases and alkaline phosphatases. J Biol Chem 276:1361–1368
Ciancaglini P, Yadav MC, Simão AMS et al (2010) Kinetic analysis of substrate utilization by native and TNAP-, NPP1-, or PHOSPHO1-deficient matrix vesicles. J Bone Miner Res 25:716–723
Simão AMS, Yadav MC, Narisawa S et al (2010) Proteoliposomes harboring alkaline phosphatase and nucleotide pyrophosphatase as matrix vesicle biomimetics. J Biol Chem 285:7598–7609
Ciancaglini P, Simão AMS, Camolezi FL et al (2006) Contribution of matrix vesicles and alkaline phosphatase to ectopic bone formation. Braz J Med Biol Res 39:603–610
Ierardi DF, Pizauro JM, Ciancaglini P (2002) Erythrocyte ghost cell-alkaline phosphatase: construction and characterization of a vesicular system for use in biomineralization studies. Biochim Biophys Acta 1567:183–192
Ciancaglini P, Simão AMS, Bolean M et al (2012) Proteoliposomes in nanobiotechnology. Biophys Rev 4:67–81
Chakkalakal DA, Mashoof AA, Novak J et al (1994) Mineralization and pH relationships in healing skeletal defects grafted with demineralized bone matrix. J Biomed Mater Res 28:1439–1443
Wu LNY, Genge BR, Dunkelberger DG et al (1997) Physicochemical characterization of the nucleational core of matrix vesicles. J Biol Chem 272:4404–4411
Arnett TR (2010) Acidosis, hypoxia and bone. Arch Biochem Biophys 503:103–109
Bollen M, Gijsbers R, Ceulemans H et al (2000) Nucleotide pyrophosphatase/phophodiesterases on the move. Crit Rev Biochem Mol Biol 35:393–432
Millán JL (2006) Mammalian alkaline phosphatases. From biology to applications in medicine and biotechnology. Wiley-VCH Verlag, Weinheim
Wu LN, Yoshimori T, Genge BR et al (1993) Characterization of the nucleational core complex responsible for mineral induction by growth plate cartilage matrix vesicles. J Biol Chem 268:25084–25094
Wu LN, Genge BR, Dunkelberger DG et al (1997) Physicochemical characterization of the nucleational core of matrix vesicles. J Biol Chem 272:4404–4411
Simão AMS, Beloti MM, Cezarino RM et al (2007) Membrane-bound alkaline phosphatase from ectopic mineralization and rat bone marrow cell culture. Comp Biochem Physiol A 146:679–687
Gijsbers R, Ceulemans H, Bollen M (2003) Functional characterization of the non-catalytic ectodomains of the nucleotide pyrophosphatase/phosphodiesterase NPP1. Biochem J 371:321–330
Camolezi FL, Daghastanli KPR, Magalhães PP et al (2002) Construction of an alkaline phosphatase-liposome system: a tool for biomineralization study. Int J Biochem Cell Biol 1282:1–11
Hartree EF (1972) Determination of protein: a modification of the lowry method that gives a linear photometric response. Anal Biochem 48:422–427
Pizauro JM, Ciancaglini P, Leone FA (1995) Characterization of the phosphatidylinositol-specific phospholipase C—released form of rat osseous plate alkaline phosphatase and its possible significance on endochondral ossification. Mol Cell Biochem 152:121–129
Heinonen JK, Lahti RJ (1981) A new and convenient colorimetric determination of inorganic orthophosphate and its application to the assay of inorganic pyrophosphatase. Anal Biochem 113:313–317
Leone FA, Baranauskas JA, Furriel RPM et al (2005) SigrafW: an easy-to-use program for fitting enzyme kinetic data. Biochem Mol Educ 33:399–403
Genge BR, Wu LN, Wuthier RE (2007) Kinetic analysis of mineral formation during in vitro modeling of matrix vesicle mineralization: effect of annexin A5, phosphatidylserine, and type II collagen. Anal Biochem 367:159–166
Wuthier RE, Rice GS, Wallace JE et al (1985) In vitro precipitation of calcium phosphate under intracellular conditions: formation of brushite from an amorphous precursor in the absence of ATP. Calcif Tissue Int 37:401–410
Wu LNY, Wuthier MG, Genge BR et al (1997) In situ levels of intracellular Ca2+ and pH in avian growth plate cartilage. Clin Orthop Relat Res 335:310–324
Wuthier RE (1977) Electrolytes of isolated epiphyseal chondrocytes, matrix vesicles, and extracellular fluid. Calcif Tissue Res 23:125–133
Genge BR, Wu LN, Wuthier RE (2007) In vitro modeling of matrix vesicle nucleation: synergistic stimulation of mineral formation by annexin A5 and phosphatidylserine. J Biol Chem 282:26035–26045
Boskey AL, Goldberg MR, Posner AS (1978) Calcium-phospholipid-phosphate complexes in mineralizing tissue. Proc Soc Exp Biol Med 157:590–593
Wu LN, Genge BR, Sauer GR, Wuthier RE (1996) Characterization and reconstitution of the nucleational complex responsible for mineral formation by growth plate cartilage matrix vesicles. Connect Tissue Res 35:309–315
Termine JD, Peckauskas RA, Posner AS (1970) Calcium phosphate formation in vitro: II. Effects of environment on amorphous crystalline transformation. Arch Biochem Biophys 140:318–325
Eanes ED (1970) Thermochemical studies on amorphous calcium phosphate. Calcif Tissue Res 5:133–145
Wuthier RE, Lipscomb GF (2011) Matrix vesicles: structure, composition, formation and function in calcification. Front Biosci 16:2812–2902
Wuthier RE (1968) Lipids of mineralizing epiphyseal tissues in the bovine fetus. J Lipid Res 9:68–78
Boskey AL, Posner AS (1976) Extraction of a calcium-phospholipid-phosphate complex from bone. Calcif Tissue Res 19:273–283
Wuthier RE, Gore ST (1977) Partition of inorganic ions and phospholipids in isolated cell, membrane and matrix vesicle fractions: evidence for Ca–Pi–acidic phospholipid complexes. Calcif Tissue Res 24:163–171
Valhmu WB, Wu LN, Wuthier RE (1990) Effects of Ca/Pi ratio, Ca2+ × Pi ion product, and pH of incubation fluid on accumulation of 45Ca2+ by matrix vesicles in vitro. Bone Miner 8:195–209
Wu LN, Genge BR, Wuthier RE (2008) Analysis and molecular modeling of the formation, structure, and activity of the phosphatidylserine-calcium-phosphate complex associated with biomineralization. J Biol Chem 283:3827–3838
Bennett RM, Lehr JR, McCarty DJ (1975) Factors affecting the solubility of calcium pyrophosphate dihydrate crystals. J Clin Invest 56:1571–1579
Prasad R (1996) Manual of membrane lipids. Springer-Verlag, Berlin
Simão AMS, Yadav MC, Ciancaglini P, Millán JL (2010) Proteoliposomes as matrix vesicles’ biomimetics to study the initiation of skeletal mineralization. Braz J Med Biol Res 43:234–241
Roberts S, Narisawa S, Harmey D et al (2007) Functional involvement of PHOSPHO1 in matrix vesicle—mediated skeletal mineralization. J Bone Miner Res 22:617–627
Xiao Z, Camalier CE, Nagashima K et al (2007) Analysis of the extracellular matrix vesicle proteome in mineralizing osteoblasts. J Cell Physiol 210:325–335
Eanes ED (1989) Biophysical aspects of lipid interaction with mineral: liposome model studies. Anat Rec 224:220–225
Carruthers A, Melchior DL (1986) How bilayer lipids affect membrane protein activity. Trends Biotechnol 11:331–335
Anderson HC, Hsu HH, Morris DC et al (1997) Matrix vesicles in osteomalacic hypophosphatasia bone contain apatite-like mineral crystals. Am J Pathol 151:1555–1561
Wu LN, Genge BR, Kang MW et al (2002) Changes in phospholipid extractability and composition accompany mineralization of chicken growth plate cartilage matrix vesicles. J Biol Chem 277:5126–5133
Lehto MT, Sharom FJ (1998) Release of the glycosylphosphatidylinositol-anchored enzyme ecto-5′-nucleotidase by phospholipase C: catalytic activation and modulation by the lipid bilayer. Biochem J 332:101–109
Lehto MT, Sharom FJ (2002) Proximity of the protein moiety of a GPI-anchored protein to the membrane surface: a FRET study. Biochemistry 41:8368–8376
Lazic S (1995) Microcrystalline hydroxyapatite formation from alkaline solutions. J Crystal Growth 147:147–154
Guicheux J, Palmer G, Shukunami C et al (2000) A novel in vitro culture system for analysis of functional role of phosphate transport in endochondral ossification. Bone 27:69–74
Wu LN, Sauer GR, Genge BR et al (2003) Effects of analogues of inorganic phosphate and sodium ion on mineralization of matrix vesicles isolated from growth plate cartilage of normal rapidly growing chickens. J Inorg Biochem 94:221–235
Balcerzak M, Malinowska A, Thouverey C et al (2008) Proteome analysis of matrix vesicles isolated from femurs of chicken embryo. Proteomics 8:192–205
Hsu HH, Camacho NP, Anderson HC (1999) Further characterization of ATP-initiated calcification by matrix vesicles isolated from rachitic rat cartilage. Membrane perturbation by detergents and deposition of calcium pyrophosphate by rachitic matrix vesicles. Biochim Biophys Acta 1416:320–332
Acknowledgments
We thank Professor Mathieu Bollen (Leuven, Belgium) for providing the NPP2/NPP1 expression construct. This work was supported in part by Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Conselho Nacional de Desenvolvimento Científico e Tecnológico, and Grants DE12889, AR53102, and AR47908 from the National Institutes of Health. A. M. S. S. was the recipient of studentships from CAPES and FAPESP.
Author information
Authors and Affiliations
Corresponding author
Additional information
The authors have stated that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Simão, A.M.S., Bolean, M., Hoylaerts, M.F. et al. Effects of pH on the Production of Phosphate and Pyrophosphate by Matrix Vesicles’ Biomimetics. Calcif Tissue Int 93, 222–232 (2013). https://doi.org/10.1007/s00223-013-9745-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-013-9745-3