Abstract
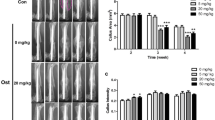
Fracture healing is retarded in the presence of cyclooxygenase-2 (COX-2) inhibitors, demonstrating an important role of COX-2 in trauma-induced woven bone adaptation. The aim of this experiment was to determine the influence of COX-2 inhibition on the remodeling and consolidation of nontraumatic woven bone produced by mechanical loading. A periosteal woven bone callus was initiated in the right tibia of female Wistar rats following a single bout of four-point bending, applied as a haversine wave for 300 cycles at a frequency of 2 Hz and a magnitude of 65 N. Daily injections of vehicle (VEH, polyethylene glycol) or the COX-2 inhibitor 5,5-dimethyl-3-3(3 fluorophenyl)-4-(4-methylsulfonal)phenyl-2(5H)-furanone (DFU, 2.0 mg · kg−1 and 0.02 mg · kg−1 i.p.), commenced 7 days postloading, and tibiae were examined 2, 3, 4, and 5 weeks postloading. Tibiae were dissected, embedded in polymethylmethacrylate, and sectioned for histomorphometric analysis of periosteal woven bone. No significant difference in peak woven bone area was observed between DFU-treated and VEH rats. However, treatment with DFU resulted in a temporal defect in woven bone formation, where the achievement of peak woven bone area was delayed by 1 week. Woven bone remodeling was observed in DFU-treated rats at 21 days postloading, demonstrating that remodeling of the periosteal callus is not prevented in the presence of a COX-2 inhibitor in the rat. We conclude that COX-2 inhibition does not significantly disrupt the mechanism of woven bone remodeling but alters its timing.




Similar content being viewed by others
References
Joldersma M, Burger EH, Semeins CM, Klein-Nulend J (2000) Mechanical stress induces COX-2 mRNA expression in bone cells from elderly women. J Biomech 33:53–61
Pavalko FM, Chen NX, Turner CH, Burr DB, Atkinson S, Hsieh YF, Qiu J, Duncan RL (1998) Fluid shear-induced mechanical signaling in MC3T3-E1 osteoblasts requires cytoskeleton-integrin interactions. Am J Physiol Cell Physiol 275:C1591–1601
Reich KM, Frangos JA (1991) Effect of flow on prostaglandin E2 and inositol trisphosphate levels in osteoblasts. Am J Physiol Cell Physiol 261:C428–C432
Okada Y, Lorenzo JA, Freeman AM, Tomita M, Morham SG, Raisz LG, Pilbeam CC (2000) Prostaglandin G/H synthase-2 is required for maximal formation of osteoclast-like cells in culture. J Clin Invest 105:823–832
Simon A, Manigrasso M, O’Connor J (2002) Cyclo-oxygenase 2 function is essential for bone fracture healing. J Bone Miner Res 17:963–976
Zhang X, Schwarz EM, Young DA, Puzas JE, Rosier RN, O’Keefe RJ (2002) Cyclooxygenase-2 regulates mesenchymal cell differentiation into the osteoblast lineage and is critically involved in bone repair. J Clin Invest 109:1405–1415
Forwood MR, Owan I, Takano Y, Turner CH (1996) Increased bone formation in rat tibiae after a single short period of dynamic loading in vivo. Am J Physiol Endocrinol Metab 270:E419–E423
Li J, Burr DB, Turner CH (2002) Suppression of prostaglandin synthesis with NS-398 has different effects on endocortical and periosteal bone formation induced by mechanical loading. Calcif Tissue Int 70:320–329
Allen HL, Wase A, Bear WT (1980) Indomethacin and aspirin: effect of nonsteroidal anti-inflammatory agents on the rate of fracture repair in the rat. Acta Orthop Scand 51:595–600
Bo J, Sudmann E, Marton PF (1976) Effect of indomethacin on fracture healing in rats. Acta Orthop Scand 47:588–599
Dimar JR 2nd, Ante WA, Zhang YP, Glassman SD (1996) The effects of nonsteroidal anti-inflammatory drugs on posterior spinal fusions in the rat. Spine 21:1870–1876
Gerstenfeld LC, Thiede M, Seibert K, Mielke C, Phippard D, Svagr B, Cullinane D, Einhorn TA (2003) Differential inhibition of fracture healing by non-selective and cyclooxygenase-2 selective non-steroidal anti-inflammatory drugs. J Orthop Res 21:670–675
Long J, Lewis S, Kuklo T, Zhu Y, Riew KD (2002) The effect of cyclooxygenase-2 inhibitors on spinal fusion. J Bone Joint Surg Am 84:1763–1768
Sudmann E, Dregelid E, Bessesen A, Morland J (1979) Inhibition of fracture healing by indomethacin in rats. Eur J Clin Invest 9:333–339
Sudmann E, Hagen T (1976) Indomethacin-induced delayed fracture healing. Arch Orthop Unfallchir 85:151–154
Reuben SS, Ekman EF (2005) The effect of cyclooxygenase-2 inhibition on analgesia and spinal fusion. J Bone Joint Surg Am 87:536–542
Dahners LE, Mullis BH (2004) Effects of nonsteroidal anti-inflammatory drugs on bone formation and soft-tissue healing. J Am Acad Orthop Surg 12:139–143
Einhorn TA (2002) Do inhibitors of cyclooxygenase-2 impair bone healing? J Bone Miner Res 17:977–978
Gajraj NM (2003) The effect of cyclooxygenase-2 inhibitors on bone healing. Reg Anesth Pain Med 28:456–465
Goodman SB, Ma T, Genovese M, Lane Smith R (2003) COX-2 selective inhibitors and bone. Int J Immunopathol Pharmacol 16:201–205
Sudmann E, Bang G (1979) Indomethacin-induced inhibition of haversian remodelling in rabbits. Acta Orthop Scand 50:621–627
Altman RD, Latta LL, Keer R, Renfree K, Hornicek FJ, Banovac K (1995) Effect of nonsteroidal antiinflammatory drugs on fracture healing: a laboratory study in rats. J Orthop Trauma 9:392–400
Goodman S, Ma T, Trindade M, Ikenoue T, Matsuura I, Wong N, Fox N, Genovese M, Regula D, Smith RL (2002) COX-2 selective NSAID decreases bone ingrowth in vivo. J Orthop Res 20:1164–1169
Turner CH, Forwood M, Rho J, Yoshikawa T (1994) Mechanical loading thresholds for lamellar and woven bone formation. J Bone Miner Res 9:87–97
Forwood MR, Bennett MB, Blowers AR, Nadorfi RL (1998) Modification of the in vivo four-point loading model for studying mechanically induced bone adaptation. Bone 23:307–310
Turner CH, Akhter MP, Raab DM, Kimmel DB, Recker RR (1991) A noninvasive, in vivo model for studying strain adaptive bone modeling. Bone 12:73–79
Parfitt AM, Drezner MK, Glorieux FH, Kanis JA, Malluche H, Meunier PJ, Ott SM, Recker RR (1987) Bone histomorphometry: standardization of nomenclature, symbols, and units. Report of the ASBMR Histomorphometry Nomenclature Committee. J Bone Miner Res 2:595–610
Li XJ, Jee WS, Chow SY, Woodbury DM (1990) Adaptation of cancellous bone to aging and immobilization in the rat: a single photon absorptiometry and histomorphometry study. Anat Rec 227:12–24
Currey JD (1984) The Mechanical Adaptations of Bone. Princeton University Press, Princeton, NJ
Su X, Sun K, Cui FZ, Landis WJ (2003) Organization of apatite crystals in human woven bone. Bone 32:150–162
Kusuzaki K, Kageyama N, Shinjo H, Takeshita H, Murata H, Hashiguchi S, Ashihara T, Hirasawa Y (2000) Development of bone canaliculi during bone repair. Bone 27:655–659
Boppart MD, Kimmel DB, Yee JA, Cullen DM (1998) Time course of osteoblast appearance after in vivo mechanical loading. Bone 23:409–415
Goodship AE, Lanyon LE, McFie H (1979) Functional adaptation of bone to increased stress. An experimental study. J Bone Joint Surg Am 61:539–546
Turner CH, Woltman TA, Belongia DA (1992) Structural changes in rat bone subjected to long-term, in vivo mechanical loading. Bone 13:417–422
Sudmann E (1975) Effect of indomethacin on bone remodelling in rabbit ear chambers. Acta Orthop Scand Suppl 160:91–115
Forwood MR (1996) Inducible cyclo-oxygenase (COX-2) mediates the induction of bone formation by mechanical loading in vivo. J Bone Miner Res 11:1688–1693
Mackie EJ, Trechsel U (1990) Stimulation of bone formation in vivo by transforming growth factor-beta: remodeling of woven bone and lack of inhibition by indomethacin. Bone 11:295–300
Evans CB (2004) The influence on human osteoblasts in vitro of non-steroidal anti-inflammatory drugs which act on different cyclooxygenase enzymes. J Bone Joint Surg Am 86:444–449
Bakker AD, Klein-Nulend J, Burger EH (2003) Mechanotransduction in bone cells proceeds via activation of COX-2, but not COX-1. Biochem Biophys Res Commun 305:677–683
Keller J, Klamer A, Bak B, Suder P (1993) Effect of local prostaglandin E2 on fracture callus in rabbits. Acta Orthop Scand 64:59–63
Dekel S, Lenthall G, Francis MJ (1981) Release of prostaglandins from bone and muscle after tibial fracture. An experimental study in rabbits. J Bone Joint Surg Br 63:185–189
Iqbal J, Zaidi M (2005) Molecular regulation of mechanotransduction. Biochem Biophys Res Commun 328:751–755
Rubin CT, Gross TS, McLeod KJ, Bain SD (1995) Morphologic stages in lamellar bone formation stimulated by a potent mechanical stimulus. J Bone Miner Res 10:488–495
Enlow D (1963) Principles of Bone Remodelling. Charles C. Thomas, Springfield, IL
Acknowledgment
We thank Ms. Wendy Kelly for expert histotechnical assistance. This project was supported in part by an Australian Postgraduate Research Award and National Health and Medical Research Council Project grants (ID 142958 and 301143). Merck and Co kindly provided the DFU.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gregory, L., Forwood, M. Cyclooxygenase-2 Inhibition Delays the Attainment of Peak Woven Bone Formation following Four-Point Bending in the Rat. Calcif Tissue Int 80, 176–183 (2007). https://doi.org/10.1007/s00223-006-0170-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-006-0170-8