Abstract
Motor imagery is suggested to be functionally equivalent to physical execution as they each utilise a common neural representation. The present study examined whether motor imagery correspondingly reflects the spatial characteristics of physically executed movements, including the signal-dependent noise that typically manifests in more variable end locations (as indicated by effective target width; We). Participants executed or imagined a single, upper-limb target-directed aim in the horizontal medio-lateral direction. The start and end of the imagined movements were indexed by the lifting and lowering of the limb over the home position, respectively. Following each imagined movement, participants had to additionally estimate their imagined end location relative to the target. All the movements had to be completed at a pre-specified criterion time (400 ms, 600 ms, 800 ms). The results indicated that the We increased following a decrease in movement time for execution, but not imagery. Moreover, the total error of imagined movements was greater than the actual error of executed movements. While motor imagery may comprise a neural representation that also contributes to the execution of movements, it is unable to closely reflect the random sources of variability. This limitation of motor imagery may be attributed to the comparatively limited efferent motor signals.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
For many years, there has been great interest surrounding motor imagery, where bodily movements are mentally simulated without physically executing the movement itself. This interest has no doubt manifested from the benefits that are served by imagery including the learning (Romano-Smith et al. 2018, 2019; Vogt, 1995) and re-learning (Crosbie et al. 2004; Dijkerman et al. 2004; Braun et al. 2013) of motor skills. Perhaps the most common or accepted theoretical framework surrounding motor imagery involves the concept of functional equivalence, which states that the neural representation that is responsible for the execution of movement is the same as that for the imagery of movement (Jeannerod 1994, 1999). This concept has been strongly supported by evidence of imagery and execution correspondingly activating the fronto-parietal neural regions (Filimon et al. 2007; Hétu et al. 2013). Moreover, there is evidence that imagery can modulate the corticospinal excitability (e.g., opponense pollicis during imagined forearm extension/flexion) that is generated by transcranial magnetic stimulation at the primary motor cortex (M1) (Fadiga et al. 1999).
Numerous studies have provided supporting evidence at the behavioural level by demonstrating that the task constraints influencing executed movement times may correspondingly influence imagined movement times (Decety and Jeannerod 1995; Glover and Dixon 2013; Gueugneau et al. 2017; Papaxanthis et al. 2002; Radulescu et al. 2010; Roberts et al. 2019; Rozand et al. 2015; Sirigu et al. 1995; Sirigu et al. 1996; Slifkin 2008; for a review, see Guillot and Collet 2005). Specifically, the greater the task difficulty, then the longer it takes to execute and imagine the movements that are required to complete the task. This principle is heavily adapted from the inherent trade-off between movement speed and accuracy (Woodworth 1899), which can be more formally expressed by a logarithmic function known otherwise as Fitts’ Law (Fitts 1954; Fitts and Peterson 1964). This law of human movement is able to predict movement time using the formula: MT = a + b(ID), where a and b represent the intercept and slope coefficient, respectively. Meanwhile, the ID represents the index of difficulty, which can be defined as: log2(2A/W), where A and W represent the movement amplitude and designated target width, respectively. While the executed and imagined movement times are rarely the same due to the differences in base movement times (as indicated by the intercept), they nevertheless demonstrate a close similarity in their relation with task difficulty (as indicated by the slope coefficient) (e.g., Wong et al. 2013).
While this line of research has greatly advocated the equivalence between execution and imagery, it can be argued that it is restricted to findings within the temporal domain. That is, it pertains to situations where only the executed or imagined movement times are evaluated in the presence of particular task constraints. However, there has been comparatively limited research surrounding the equivalence between execution and imagery in terms of the spatial domain. At the same time, it is also possible to reflect the fore mentioned trade-off between movement speed and accuracy by inversely examining the end locations when movement times are held constant (Carlton 1994; Schmidt et al. 1979; Wright and Meyer 1983; Zelaznik et al. 1981). Thus, it is possible to ask whether motor imagery comprises of the same spatial characteristics as physically executed movements once constraints are placed on the movement times.
With this in mind, it is noteworthy that increasing movement velocity (synonymous with a decreased movement time at set amplitude) can increase the within-participant variability of the end location (Schmidt et al. 1979). This finding highlights how the noise or stochastic properties of human movement are signal-dependent–greater impulses manifest in more noise (see also, Faisal et al. 2008; Meyer et al. 1988). This approach to the trade-off between movement speed and accuracy can be formally expressed by a linear function that is sometimes referred to as Schmidt’s Law. This law of human movement may be calculated as: We = a + b(D/MT), where a and b once more represent the intercept and slope coefficient, respectively. Meanwhile, the D and MT represent the distance and movement time (equating to velocity), and the We represents the effective target width, which may be calculated from the within-participant standard deviation (SD) of end locations: SD × 4.133. This spatial parameter is based on the assumption that the end locations across a series of movement attempts reflect a normal or Gaussian distribution, which may be scaled to a hypothetical target boundary that subtends a select proportion of the end locations. In this regard, a multiple of 4.133 represents approximately 95% of the distribution (Welford 1968).
To this end, the aim of the present study was to examine whether motor imagery similarly reflects the signal-dependent noise that typically manifests within execution. To investigate, we had participants execute or imagine a single, horizontal aiming movement with the upper-limb at a pre-specified criterion time. The imagined movements unfolded by having the participants vertically lift and lower the limb to index the start and end of their movements, respectively. In addition, participants precisely estimated, where they imagined their movements to have ended relative to the intended target once the imagery was completed.
Consequently, there were two measures of imagined location: within-trial; when the limb was not intended to deviate from its original location, and post-trial; when the limb was recalled from its previous imagined location. These measures recognise a distinction that is often drawn by neuroscientific and behavioural models (e.g., Csibra 2007; Henke 2010; Shanks 2010; Shriffin and Schneider 1977). Indeed, the former measure is indicative of covert localisation, where the utility of a common neural representation for execution and imagery may unintentionally contaminate the end location (i.e., subtle variations in the end location relative to the start location) (for examples, see Dijkerman and Smit 2007; Kilner et al. 2003; Ray et al. 2013; Roberts et al. 2015). This measure is synonymous with implicit or procedural forms of memory, and it is highly sensitive to lower-level or bottom-up processes. Alternatively, the latter measure indicates overt localisation, where the imagined outcome of movements may be successfully recalled after the imagery has been completed. This measure more greatly captures explicit or declarative forms of memory, and pertains to higher-level or top-down processes. By featuring both of these measures, it is possible to corroborate each of their outcomes, while avoiding any failure in detecting a possible influence within imagery.
To this end, the feature of signal-dependent noise within the human sensorimotor system has been closely reflected by the findings of the We being positively related to the movement velocity (or inversely related to the movement times at set amplitude) (Schmidt et al. 1979). In other words, the spatial variability is contingent upon the magnitude of the efferent motor signals that are directly responsible for the physical execution of target-directed movements. Thus, it stands to reason that the We will be inversely related to the criterion movement times within execution. However, owing to the limited magnitude or complete absence of efferent signals during motor imagery, it is predicted that there will no such relation within imagery. Specifically, there will be an increase in the We following decreases in movement time for execution, although there will be limited differences in the We across movement times for imagery (covert and overt measures).
Methods
Participants
An apriori power analysis was initially conducted using G*Power software (version 3.1.9.4; see Faul et al. 2007) including the input parameters of: α = 0.05, 1 − β = 0.90, and f = 0.40 (large). The required effect size was directly adapted from a collection of studies that featured statistical main effects of temporal- and accuracy-constraints for measures of effective target width (e.g., Slifkin and Eder, 2017) and movement time (e.g., Heremans et al. 2011; McCormick et al. 2013), respectively. Likewise, it can be indirectly supported by the strong correlation coefficients that are evidenced between the effective target width and movement velocity (Schmidt et al. 1979), as well as the movement time and index of difficulty (Fitts 1954; Fitts and Petersen 1964) (rs > 0.90). There was a minimum requirement of 15 participants for this particular study. There were 17 participants (age range = 21–40; 14 male and 3 female; 15 right-handed and 2 left-handed (self-declared)) with normal or corrected-to-normal vision and free from any neurological condition that agreed to take part in the study. The study was designed and conducted in accordance with the Declaration of Helsinki (2013), and approved by the local research ethics committee.
Materials
Participants sat directly in front of a digitizing graphics tablet (GTCO Calcomp Drawing Board VI; temporal resolution = 125 Hz, spatial resolution = 1000 lines per inch) and LCD computer monitor (47.5 cm × 27.0 cm; temporal resolution = 75 Hz; spatial resolution = 1280 × 800 pixels) (Fig. 1a). Targets were represented as black cross-hairs (L × W = 20 × 2 mm) that were printed on white A4 paper with a 24-cm separation (centre-to-centre), which was secured to the active surface of the tablet by placing it under an attached translucent polypropylene sheet. Stimuli that were generated on the monitor were controlled by Matlab (2018b) (The Mathworks Inc., Natick, MA) running Psychtoolbox (version 3.0.11) (Pelli 1997).
Execution protocol
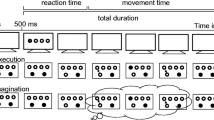
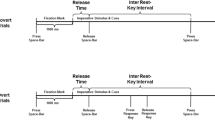
Participants were tasked with executing a single, three-dimensional aiming movement in the horizontal medio-lateral direction (right-to-left for right-handed participants, left-to-right for left-handed participants) toward one of the cross-hairs using a stylus pen on the tablet with their dominant upper-limb (Fig. 1b). A trial would commence by initially contacting the surface of the tablet with the tip of the stylus pen. After a 2-s delay, an auditory tone would sound to indicate the start of the trial. Participants were then free to commence the movement in their own time. The start and end of the movement was taken as participants initially lifting the stylus from the home position and once more establishing contact with the tablet surface near the target position, respectively.
Participants had to reach as close as possible to the centre of the target at a pre-specified criterion time. Movement time was allowed to vary ± 10% of the criterion time to qualify as a successful trial. There were three possible criterion times including 400 ms (± 40), 600 ms (± 60) and 800 ms (± 80). Movement time was calculated as the difference in time between the initial lift and subsequent contact with the tablet. Terminal feedback of the movement time was immediately displayed on the monitor following the completion of each movement. If the feedback appeared in green, then it would indicate that the participants successfully reached the criterion time and could move onto the next trial. If the feedback appeared in red, then it would indicate that the participants failed to reach the criterion time and needed to repeat the trial. Participants had to a press key on the adjacent keypad that was connected to the computer to remove the feedback and move on to the next attempt.
Imagery protocol
Participants were tasked with imagining the same aiming movement as the execution protocol (Fig. 1b). Likewise, the trial events unfolded in a similar fashion to the execution protocol, although the start and end of the movement was indexed by participants initially lifting the stylus before once more establishing contact with the tablet surface near the home position.
Participants had to start and end their imagined movements at the same pre-specified criterion times as the execution protocol. Because the physical movements during imagery accumulated minimal displacement compared to execution, we sought to corroborate the potentially subtle variations in the end location within trials. Thus, if the imagined movement successfully reached the criterion time, then the participants were prompted to additionally estimate the imagined location of their limb following the completion of imagery. Specifically, participants had to move the stylus on the tablet to direct a cursor around the area of the cross-hair that was displayed on the monitor. When the participants perceived themselves to be positioned at the same location as the end of the imagined movement relative to the target, then they simply held the position of stylus while selecting an adjacent button near the tip of the stylus. If the imagined movement failed to reach the criterion time, then participants were not prompted to estimate their imagined location and needed to repeat the trial.
Block procedures
As well as being familiarised with the aiming task, it was essential for participants to be initially familiarised with the criterion times prior to formally undertaking the execution and imagery protocols. Likewise, the equivalence between execution and imagery is often contingent upon the prior practice or physical exposure to the task dynamics, where a representation may be initially constructed in practice and later awakened in imagery (Yoxon et al. 2015, 2017). Thus, participants undertook one set of practice trials when they were first introduced to each of the criterion times. The practice trials were completed immediately prior to one of the execution or imagery protocols depending on their order. The practice trials were similar to the execution protocol including the provision of terminal feedback of the movement time. In the event that participants failed to reach a criterion time, then they had to repeat the trial. Practice continued to unfold until the participants reached the criterion time on ten trials.
The criterion times were undertaken in a blocked order of trials, which were counter-balanced between participants using a Latin-square design. Within each block of criterion times, participants completed the execution and imagery protocols in a blocked order of trials. The order of the execution and imagery protocols within each block of criterion times was also counter-balanced between participants. The execution and imagery protocols would continue to unfold until the participants reached the criterion time on 15 trials before subsequently progressing on to the next criterion time.
Data management
Position data from the tablet were stored as pixelated coordinates from the monitor. Positional errors were calculated within the horizontal (x-axis) and vertical (y-axis) directions and converted into millimetres. For the executed and imagined movements, positional error was regarded as the distance between the end location and target, which was taken as the participant mean start location plus the movement amplitude (24 cm). For the estimated imagined location, positional error was regarded as the distance between the estimated location and centre of the cross-hair location on the monitor.
While the aiming task was an unconstrained three-dimensional movement that generates measures within the horizontal and vertical directions, the theoretical underpinnings and empirical research principally relate to the central tendency and dispersion of the primary axis alone (e.g., Elliott et al. 2017; Meyer et al. 1988; Schmidt et al. 1979; Slifkin and Eder 2017; Zelaznik et al. 1981; cf. Carlton 1994). Thus, the dependent measures were derived from the primary direction of the movement (i.e., horizontal; x-axis). The signed difference in the horizontal position of the limb and target for each of the individual trials was initially calculated. Therein, the within-participant standard deviation (SD) (population n degrees-of-freedom) was calculated, and converted by a multiplicative value of 4.133 to obtain the We: SD × 4.133 (Welford 1968). Prior to these calculations and formal statistical analysis, trials were removed when participants mistakenly failed to move during the execution protocol (< 15-cm movement amplitude), definitively moved during the imagery protocol (> 15-cm movement amplitude) or failed to reach the criterion movement times (± 10%).Footnote 1
In addition to the We itself, we also calculated the individual participant slope coefficients that pertained to the relation between We and MT. While the We is linearly related to velocity (i.e., We = a + b(D/MT)), it is not possible to derive this parameter from imagined movements. Nevertheless, a linear relation between We and velocity should translate into a similar relation between We and MT when D is held constant (24 cm).
Statistical analysis
In line with the principles of open science, the individual participant data and calculations for the relevant measures have been uploaded to the Open Science Framework: https://osf.io/d3n6u. We separately analysed the actual imagined locations within the trials and estimated imagined locations after the trials to indicate both covert (synonymous with subtle unintended end locations) and overt (synonymous with consciously selected end locations) aspects of spatial localisation, respectively. Covert localisation of the limb was first assessed by analysing the We using a two-way repeated-measures ANOVA, which featured the factors of protocol (execution, imagery) and temporal window (400 ms, 600 ms, 800 ms). Depending on an assessment of normal distribution using the Shapiro–Wilk test, we compared the participant slope coefficients of execution and imagery using a paired sample t-test (parametric) or Wilcoxon signed-rank test (non-parametric). Meanwhile, the overt localisation of estimated imagined locations was assessed by analysing the We across the criterion times using a one-way repeated-measures ANOVA. Providing confirmation of the data being normally distributed, the participant slope coefficients were compared to a theoretical value of zero using a single-sample t-test (synonymous with no linear relation).
Significant interactions from the factorial ANOVA were initially decomposed by conducting simple effect analyses on each level of protocol (using the mean square error and degrees-of-freedom of error from the original factorial ANOVA). Further significant effects that featured more than two means were decomposed using the Tukey HSD post hoc procedure. Effect sizes from the ANOVAs were indicated by partial eta-squared (ηp2). All statistical effects were declared as significant at p < 0.05.
Results
Covert localisation
Analysis of the We indicated a significant main effect of protocol, F(1, 16) = 27.30, p < 0.001, ηp2 = 0.63, and temporal window, F(2, 32) = 16.44, p < 0.001, ηp2 = 0.51. These statistical effects were superseded by a significant interaction between protocol and temporal window, F(2, 32) = 26.59, p < 0.001, ηp2 = 0.62 (Figs. 2, 3a). Simple effect analyses on each level of protocol revealed a significant main effect of temporal window for execution, F(2, 32) = 55.89, p < 0.001, ηp2 = 0.78. Post hoc analysis revealed a significantly larger width for the 400-ms window compared to the 600-ms window, which was also significantly larger than the 800-ms window (Tukey HSD value = 1.56).Footnote 2 However, there was no significant main effect of temporal window for imagery, F(2, 32) = 0.14, p > 0.05, ηp2 = 0.00. Comparison of the individual participant slopes that pertain to the We-MT relations indicated a significantly more negative slope for execution (Mdn = − 14.89, IQR = 13.81) compared to imagery (Mdn = 0.17, IQR = 6.34), T = 1, z = − 3.57, p < 0.001.
Illustration of the mean horizontal and vertical locations with respect to the target (0, 0) for execution (red) and imagery (covert = green, overt = blue). Symbols indicate the different temporal windows (400 ms = squares; 600 ms = circles; 800 ms = triangles). Dotted error bars collectively span the We (equivalent to 95% of the distribution)
Overt localisation
Analysis of the We indicated that there was no significant main effect of temporal window, F(2, 32) = 0.1.14, p > 0.05, ηp2 = 0.07 (Figs. 2, 3b). Meanwhile, the individual participant slopes indicated no significant difference compared to a theoretical value of zero (M = − 8.46, SE = 6.37), t(16) = 1.33, p > 0.05.
Following observation of the mean and within-participant variability (Fig. 2), we observed a much larger degree of error from the estimates of imagined end locations compared to the covert measures. To explore this trend further, we additionally compared the total error of executed and estimated imagined locations (synonymous with the within-participant root mean square error) within the primary and secondary direction of movement. For the primary direction, there was significantly less error produced by execution (M = 2.47, SE = 0.15) compared to the estimates following imagery (M = 4.41, SE = 0.40), t(16) = 4.34, p < 0.01. Likewise, for the secondary direction, there was significantly less error for execution (Mdn = 1.91, IQR = 0.40) compared to the estimates following imagery (Mdn = 4.68, IQR = 16.27), T = 6, z = − 3.34, p < 0.01. Because the current executed and estimated imagined locations were captured by separate measures (i.e., within-trial vs. post-trial), where any differences between them could merely result from some other independent process (e.g., memory), these findings should be interpreted with at least some degree of caution.
Discussion
The present study examined whether the increasing spatial variability that is exhibited following decreases in executed movement times may also be reflected in motor imagery. That is, we asked whether the inverse relation between We and criterion movement time for executed movements may correspondingly manifest in the covert or overt localisation of imagined movements. Both these measures ensured that non-conscious lower-level and conscious higher-level components of the imagined movement locations could be evaluated, respectively. Broadly speaking, the findings showed that the We was inversely related to the criterion movement time only for execution, and not for imagery. At the same time, there was an increased error in the post-trial estimates of the imagined end locations compared to the actual executed movements.
The inverse relation between the We and movement time pertains to the inherent trade-off between movement speed and accuracy, where the noise or stochastic properties of human movement are signal-dependent (Faisal et al. 2008; Meyer et al. 1988; Schmidt et al. 1979). In other words, these random sources of spatial variability are contingent upon the projection of efferent motor signals. Thus, it stands to reason that with the comparatively limited efferent motor signals during imagery, there is limited semblance to the downstream consequences on covert spatial localisation. That said, the limited equivalence between the spatial characteristics of execution and imagery within the present study alludes to mere random sources of variability as opposed to intended or pre-planned sources of variability (van Beers 2009). Indeed, it is still possible that the neural representation underlying the equivalence between execution and imagery may extend to the spatial characteristics of movement, but only when it originates from intended sources (i.e., pre-planned direction of movement). For example, in a similar vein to the execution of bimanual movements, there is evidence that the executed straight-line movements of one limb can become contaminated and begin to coordinate with the imagined circular movements of the unused limb (Piedimonte et al. 2018; see also, Ramsey et al. 2010). In this instance, the imagined movement direction may have awakened a neural representation that is correspondingly designed for the physical execution of that same movement direction (Jeannerod 1994, 1999).
Because of the limited similarity in the trends that emerge between the movement times within executed and imagined movements, it is debatable whether the current measures of covert and overt localisation even capture the correspondence between execution and imagery. Indeed, while there were limited differences in the within-participant variability of imagined movements, it is possible to evidence a correspondence between execution and imagery by more closely observing the between-participant variability. That is, the between-participant variability that often manifests in executed movements may also unfold within the imagined movements (for a similar logic, see Welsh et al. 2009; Welsh and McDougall 2012). With this in mind, further correlations (Spearman’s rho; non-parametric) on the We indicated a positive relation between the executed and imagined movements (covert 400 ms: rs = 0.36, p > 0.05; 600 ms: rs = 0.67, p < 0.001; 800 ms: rs = 0.56, p < 0.05; overt 400 ms: rs = 0.19, p > 0.05; 600 ms: rs = 0.05, p > 0.05; 800 ms: rs = 0.38, p > 0.05). This relation may be interpreted as the characteristics of executed movements spilling-over onto imagined movements, where the more precise individuals are in execution, then the more precise they are in imagery. Presumably, the representation that is responsible for appropriately parameterizing executed movements is the same representation that is utilised for imagined movements. However, the relation between executed and imagined movements was only evident within the covert measure (as opposed to overt localisation), which would suggest that these effects pertained to a lower-level, downstream consequence of utilising a neural representation that appears outside of conscious awareness (for similar covert responses, see Kilner et al. 2003).
Despite the limited similarity between the executed and overt measure of imagined movements, the additional analysis of total error indicated a modest and more erred spatial estimate for imagery compared to the actual error for execution. This finding may refute potential concerns surrounding participant engagement within the imagery protocol as this possibility would have alternatively manifested in a smaller amount of error (i.e., near perfection). Instead, it is possible that participants were consciously aware of the spatial variability that could manifest from executed movements (constant error (CE) grand M = 0.87 mm, We grand M = 7.80 mm; hypothetical range of error (CE ± We/2) = − 3.03–4.77 mm), and consequently estimated over a larger area that could safely fit the distribution of actual end locations (CE grand M = -0.32, We grand M = 14.22 mm; hypothetical range of error (CE ± We/2) = − 7.43–6.79 mm). This trend concurs with computational models of sensorimotor control, where the nervous system converges onto a movement approach or central tendency that compensates for the distribution and associated likelihood of movement outcomes (Harris and Wolpert 1998; Wolpert and Ghahramani 2000). For example, it has been shown that individuals typically undershoot intended target locations to avoid the cost of overshooting when they initially perceive an increased likelihood of missing the target (Elliott et al. 2004; Roberts 2020).
Alternatively, it is possible that the overly erred estimate of imagined end locations following the pre-specified criterion times may resemble the previously evidenced increase in imagined compared to executed movement times under particular task constraints. That is, the base movement times (as indicated by the intercept from the Fitts’ Law equation) tend to be greater for imagined compared to executed movements when the task difficulty is altered and time is no longer constrained (e.g., Wong et al. 2013). This trend has been primarily attributed to the additional demands placed on the control of imagined movements, including the need to index the start and end of the movements (e.g., lifting the stylus before returning to a similar position) (Glover and Baran 2017; Glover and Dixon 2013; for an alternative explanation, see Yoxon et al. 2015, 2017). In the context of the present study, it is possible that the additional demands that are placed on the control of imagined movements could no longer be accommodated within the movement time, because it was constrained, and thus alternatively manifested in a larger estimate of spatial error.
At the same time, it is also important to recognise the underlying contributions of planning and control within executed movements to appreciate the overly erred estimate of imagined end locations. Indeed, the early portion of executed movements (e.g., prior to peak velocity) are usually subject to a degree of error, where the parameterization of movement prior to the movement itself is not adequate enough to reach the intended target (van Beers 2009; for measuring the contribution of planning/control, see Elliott et al. 1999; Heath et al. 2004). Consequently, it is possible to utilise sensory feedback within the movement (e.g., after peak velocity) to adjust the trajectory whenever an error ensues (Elliott et al. 2001). In the context of imagery, the utility of a representation that is correspondingly used for the initial execution of movement may solely contribute to the outcome of imagined movements because of the absence of any movement-specific sensory feedback. Thus, while it is possible for imagery to infer and perhaps compensate for the error that is associated with planning, it is unable to overcome this error in a way that can be achieved during execution (for a similar logic, see Glover and Baran 2017; Glover and Dixon 2013).
In conclusion, while it is well accepted that motor imagery is consistent with the trade-off between movement speed and accuracy when using the Fitts aiming paradigm (temporal domain), the present study shows how it is not entirely consistent with the trade-off when alternatively adopting the Schmidt aiming paradigm (spatial domain). That is, imagined movements fail to account for the signal-dependent noise that typically manifests within executed movements. This trend was demonstrated in both covert and overt measures of spatial localisation. Thus, we contend that while motor imagery encompasses a neural representation that is correspondingly used for execution, it is limited to intentional or pre-planned components of movement without accurately incorporating the noise or stochastic properties of the movement itself. Future research may similarly explore whether motor imagery encapsulates the spatial characteristics of executed movements, including the separate contribution of intended and random sources of spatial variability.
Notes
The number of attempts where participants failed to reach each of the criterion times (± 10%) were analysed using a two-way repeated-measures ANOVA, which featured factors of protocol (execution, imagery) and temporal window (400 ms, 600 ms, 800 ms). There was a significant main effect of protocol, F(1, 16) = 56.11, p < .001, ηp2 = 78, as there were significantly fewer trials with a temporal error for execution (M = 12 trials, SE = 2.00) compared to imagery (M = 23 trials, SE = 1.14). There was a significant main effect of temporal window, F(2, 32) = 104.02, p < 0.05, ηp2 = 0.22. Post hoc analysis revealed that there were significantly more errors generated for the 400- (M = 21 trials, SE = 2.44) compared to 800-ms window (M = 14 trials, SE = 1.84), while there were no significant differences for the comparisons to the 600-ms window (M = 18 trials, SE = 1.77) (Tukey HSD value = 6.10). Meanwhile, there was no significant interaction between protocol and temporal window, F(2, 32) = 0.02, p > 0.05, ηp2 = 0.01.
Trend analyses for the execution protocol indicated both a significant linear, F(1, 16) = 45.76, p < 0.001, ηp2 = 0.74, and quadratic, F(1, 16) = 7.74, p < 0.05, ηp2 = 0.33, component. While reflecting a negative linear relation between We and MT, there appeared less of a decrease between the 600- and 800-ms windows. This outcome is not without precedence as previous findings have shown a reduced linear relation for longer criterion movement times (e.g., Schmidt et al. 1979). Presumably, the more prolonged temporal windows (≥600 ms) accommodate a feedback processing time-lag, where online adjustments to the trajectory may serve to reduce the spatial variability (Zelaznik et al. 1981; see also, Elliott et al. 2001).
References
Braun S, Kleynan M, van Heel T, Kruithof N, Wade D, Beurskens A (2013) The effects of mental practice in neurological rehabilitation; a systematic review and meta-analysis. Front Hum Neurosci 7:390. https://doi.org/10.3389/fnhum.2013.00390
Carlton LG (1994) The effects of temporal-precision and time-minimization constraints on the spatial and temporal accuracy of aimed hand movements. J Mot Behav 26:43–50. https://doi.org/10.1080/00222895.1994.9941660
Crosbie JH, McDonough SM, Gilmore DH, Wiggam MI (2004) The adjunctive role of mental practice in the rehabilitation of the upper limb after hemiplegic stroke: a pilot study. Clin Rehabil 18:60–68. https://doi.org/10.1191/0269215504cr702oa
Csibra G (2007) Action mirroring and action understanding: an alternative account. In: Haggard P, Rossetti Y, Kawato M (eds) Sensorimotor foundations of higher cognition: attention and performance XXII. Oxford University Press, Oxford, pp 435–459
Decety J, Jeannerod M (1995) Mentally simulated movements in virtual reality: does Fitts's law hold in motor imagery? Behav Brain Res 72:127–134. https://doi.org/10.1016/0166-4328(96)00141-6
Dijkerman HC, Smit MC (2007) Interference of grasping observation during prehension, a behavioural study. Exp Brain Res 176:387–396. https://doi.org/10.1007/s00221-006-0627-z
Dijkerman HC, Ietswaart M, Johnston M, MacWalter RS (2004) Does motor imagery training improve hand function in chronic stroke patients? A pilot study. Clin Rehabil 18:538–549. https://doi.org/10.1191/0269215504cr769oa
Elliott D, Binstead G, Heath M (1999) The control of goal-directed limb movements: correcting errors in the trajectory. Hum Mov Sci 18:121–136. https://doi.org/10.1016/S0167-9457(99)00004-4
Elliott D, Helsen WF, Chua R (2001) A century later: Woodworth's (1899) two-component model of goal-directed aiming. Psychol Bull 127:342–357. https://doi.org/10.1037/0033-2909.127.3.342
Elliott D, Hansen S, Mendoza J, Tremblay L (2004) Learning to optimize speed, accuracy, and energy expenditure: a framework for understanding speed-accuracy relations in goal-directed aiming. J Mot Behav 36:339–351. https://doi.org/10.3200/JMBR.36.3.339-351
Elliott D, Lyons J, Hayes SJ, Burkitt JJ, Roberts JW, Grierson LEM, Hansen S, Bennett SJ (2017) The multiple process model of goal-directed reaching revisited. Neurosci Biobehav R 72:95–110. https://doi.org/10.1016/j.neubiorev.2016.11.016
Fadiga L, Buccino G, Craighero L, Fogassi L, Gallese V, Pavesi G (1999) Corticospinal excitability is specifically modulated by motor imagery: a magnetic stimulation study. Neuropsychologia 37:147–158. https://doi.org/10.1016/s0028-3932(98)00089-x
Faisal AA, Selen LP, Wolpert DM (2008) Noise in the nervous system. Nat Rev Neurosci 9:292–303. https://doi.org/10.1038/nrn2258
Faul F, Erdfelder E, Lang AG, Buchner A (2007) G*Power 3: A flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav Res Meth 39:175–191. https://doi.org/10.3758/BF03193146
Filimon F, Nelson JD, Hagler DJ, Sereno MI (2007) Human cortical representations for reaching: mirror neurons for execution, observation, and imagery. NeuroImage 37:1315–1328. https://doi.org/10.1016/j.neuroimage.2007.06.008
Fitts PM (1954) The information capacity of the human motor system in controlling the amplitude of movement. J Exp Psychol 47:381–391. https://doi.org/10.1037/h0055392
Fitts PM, Peterson JR (1964) Information capacity of discrete motor responses. J Exp Psychol 67:103–112. https://doi.org/10.1037/h0045689
Glover S, Baran M (2017) The motor-cognitive model of motor imagery: evidence from timing errors in simulated reaching and grasping. J Exp Psychol Hum Percept Perform 43:1359–1375. https://doi.org/10.1037/xhp0000389
Glover S, Dixon P (2013) Context and vision effects on real and imagined actions: support for the common representation hypothesis of motor imagery. J Exp Psychol Hum Percept Perform 39:1352–1364. https://doi.org/10.1037/a0031276
Gueugneau N, Pozzo T, Darlot C, Papaxanthis C (2017) Daily modulation of the speed-accuracy trade-off. Neuroscience 356:142–150. https://doi.org/10.1016/j.neuroscience.2017.04.043
Guillot A, Collet C (2005) Duration of mentally simulated movement: a review. J Mot Behav 37:10–20. https://doi.org/10.3200/JMBR.37.1.10-20
Harris CM, Wolpert DM (1998) Signal-dependent noise determines motor planning. Nature 394:780–784. https://doi.org/10.1038/29528
Heath M, Westwood DA, Binsted G (2004) The control of memory-guided reaching movement in peripersonal space. Mot Control 8:76–106. https://doi.org/10.1123/mcj.8.1.76
Henke K (2010) A model for memory systems based on processing modes rather than consciousness. Nat Rev Neurosci 11:523–532. https://doi.org/10.1038/nrn2850
Hétu S, Grégoire M, Saimpont A, Coll M-P, Eugène F, Michon P-E, Jackson PL (2013) The neural network of motor imagery: an ALE meta-analysis. Neurosci Biobeh R 37:930–949. https://doi.org/10.1016/j.neubiorev.2013.03.017
Heremans E, Smits-Engelsman B, Caeyenberghs K, Vercruysse S, Nieuwboer A, Feys P, Helsen WF (2011) Keeping an eye on imagery: the role of eye movements during motor imagery training. Neuroscience 195:37–44. https://doi.org/10.1016/j.neuroscience.2011.07.030
Jeannerod M (1994) The representing brain: neural correlates of motor intention and imagery. Behav Brain Sci 17:187–202. https://doi.org/10.1017/S0140525X00034026
Jeannerod M (1999) The 25th Bartlett Lecture. To act or not to act: perspectives on the representation of actions. Q J Exp Psychol A 52:1–29. https://doi.org/10.1080/713755803
Kilner JM, Paulignan Y, Blakemore SJ (2003) An interference effect of observed biological movement on action. Curr Biol 13:522–525. https://doi.org/10.1016/s0960-9822(03)00165-9
McCormick SA, Causer J, Holmes PS (2013) Active vision during action execution, observation and imagery: evidence for shared motor representations. PLoS ONE 8:e67761. https://doi.org/10.1371/journal.pone.0067761
Meyer DE, Abrams RA, Kornblum S, Wright CE, Smith JE (1988) Optimality in human motor performance: ideal control of rapid aimed movements. Psychol Rev 95:340–370
Papaxanthis C, Schieppati M, Gentili R, Pozzo T (2002) Imagined and actual arm movements have similar durations when performed under different conditions of direction and mass. Exp Brain Res 143:447–452. https://doi.org/10.1007/s00221-002-1012-1
Pelli DG (1997) The VideoToolbox software for visual psychophysics: transforming numbers into movies. Spat Vis 10:437–442. https://doi.org/10.1163/156856897X00366
Piedimonte A, Conson M, Frolli A, Bari S, Della Gatta F, Rabuffetti M, Keller R, Berti A, Garbarini F (2018) Dissociation between executed and imagined bimanual movements in autism spectrum conditions. Autism Res 11:376–384. https://doi.org/10.1002/aur.1902
Radulescu PV, Adam JJ, Fischer MH, Pratt J (2010) Fitts’ Law violation and motor imagery: are imagined movements truthful or lawful? Exp Brain Res 201:607–611. https://doi.org/10.1007/s00221-009-2072-2
Ramsey R, Cumming J, Eastough D, Edwards MG (2010) Incongruent imagery interferes with action initiation. Brain Cogn 74:249–254. https://doi.org/10.1016/j.bandc.2010.08.005
Ray M, Dewey D, Kooistra L, Welsh TN (2013) The relationship between the motor system activation during action observation and adaptation in the motor system following repeated action observation. Hum Mov Sci 32:400–411. https://doi.org/10.1016/j.humov.2012.02.003
Roberts JW (2020) Energy minimization within target-directed aiming: the mediating influence of the number of movements and target size. Exp Brain Res 238:741–749. https://doi.org/10.1007/s00221-020-05750-w
Roberts JW, Hayes SJ, Uji M, Bennett SJ (2015) Motor contagion: the contribution of trajectory and end-points. Psychol Res 79:621–629. https://doi.org/10.1007/s00426-014-0589-x
Roberts JW, Welsh TN, Wakefield CJ (2019) Examining the equivalence between imagery and execution. Do imagined and executed movements code relative environmental features? Behav Brain Res 370:111951. https://doi.org/10.1016/j.bbr.2019.111951
Romano-Smith S, Wood G, Wright DJ, Wakefield CJ (2018) Simultaneous and alternate action observation and motor imagery combinations improve aiming performance. Psychol Sport Exerc 38:100–106. https://doi.org/10.1016/j.psychsport.2018.06.003
Romano-Smith S, Wood G, Coyles G, Roberts JW, Wakefield CJ (2019) The effect of action observation and motor imagery combinations on upper limb kinematics and EMG during dart-throwing. Scand J Med Sci Sport 29:1917–1929. https://doi.org/10.1111/sms.13534
Rozand V, Lebon F, Papaxanthis C, Lepers R (2015) Effect of mental fatigue on speed-accuracy trade-off. Neuroscience 297:219–230. https://doi.org/10.1016/j.neuroscience.2015.03.066
Schmidt RA, Zelaznik H, Hawkins B, Frank JS, Quinn JT (1979) Motor-output variability: a theory for the accuracy of rapid motor acts. Psychol Rev 47:415–451. https://doi.org/10.1037/0033-295X.86.5.415
Shanks DR (2010) Learning: from association to cognition. Annu Rev Psychol 61:273–301. https://doi.org/10.1146/annurev.psych.093008.100519
Shiffrin RM, Schneider W (1977) Controlled and automatic human information processing: II. Perceptual learning, automatic attending and a general theory. Psychol Rev 84:127–190. https://doi.org/10.1037/0033-295X.84.2.127
Sirigu A, Cohen L, Duhamel JR, Pillon B, Dubois B, Agid Y, Pierrot-Deseilligny C (1995) Congruent unilateral impairments for real and imagined hand movements. NeuroReport 6:997–1001
Sirigu A, Duhamel JR, Cohen L, Pillon B, Dubois B, Agid Y (1996) The mental representation of hand movements after parietal cortex damage. Science 273:1564–1568. https://doi.org/10.1126/science.273.5281.1564
Slifkin AB (2008) High loads induce differences between actual and imagined movement duration. Exp Brain Res 185:297–307. https://doi.org/10.1007/s00221-007-1154-2
Slifkin AB, Eder JR (2017) Degree of target utilization influences the location of movement endpoint distributions. Acta Psychol 174:89–100. https://doi.org/10.1016/j.actpsy.2017.01.008
van Beers RJ (2009) Motor learning is optimally tuned to the properties of motor noise. Neuron 63:406–417. https://doi.org/10.1016/j.neuron.2009.06.025
Vogt S (1995) On relations between perceiving, imagining and performing in the learning of cyclical movement sequences. Br J Psychol 86:191–216. https://doi.org/10.1111/j.2044-8295.1995.tb02556.x
Welford AT (1968) Fundamentals of skill. Springer, New York
Welsh TN, McDougall LM (2012) Negative priming in a joint selection task. PLoS ONE 7:e42963. https://doi.org/10.1371/journal.pone.0042963
Welsh TN, McDougall LM, Weeks D (2009) The performance and observation of action shape future behaviour. Brain Cogn 71:64–71. https://doi.org/10.1016/j.bandc.2009.04.001
Wolpert DM, Ghahramani Z (2000) Computational principles of movement neuroscience. Nat Neurosci 3:1212–1217. https://doi.org/10.1038/81497
Wong L, Manson GA, Tremblay L, Welsh TN (2013) On the relationship between the execution, perception, and imagination of action. Behav Brain Res 257:242–252. https://doi.org/10.1016/j.bbr.2013.09.045
Woodworth RS (1899) The accuracy of voluntary movement. Psychol Rev 3:1–119. https://doi.org/10.1037/h0092992
Wright CE, Meyer DE (1983) Conditions for a linear speed–accuracy trade-off in aimed movements. Q J Exp Psychol A 35:279–296. https://doi.org/10.1080/14640748308402134
Yoxon E, Tremblay L, Welsh TN (2015) Effect of task-specific execution on accuracy of imagined aiming movements. Neurosci Lett 585:72–76. https://doi.org/10.1016/j.neulet.2014.11.021
Yoxon E, Pacione SM, Song JH, Welsh TN (2017) The action-specific effect of execution on imagination of reciprocal aiming movements. Hum Mov Sci 54:51–62. https://doi.org/10.1016/j.humov.2017.03.007
Zelaznik HN, Shapiro DC, McColsky D (1981) Effects of a secondary task on the accuracy of single aiming movements. J Exp Psychol Hum Percept Perform 7:1007–1018. https://doi.org/10.1037/0096-1523.7.5.1007
Acknowledgements
The authors would like to thank Scott Glover and Digby Elliott for their helpful comments regarding this paper.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Melvyn A. Goodale.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Roberts, J.W., Wood, G. & Wakefield, C.J. Examining the equivalence between imagery and execution within the spatial domain – Does motor imagery account for signal-dependent noise?. Exp Brain Res 238, 2983–2992 (2020). https://doi.org/10.1007/s00221-020-05939-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-020-05939-z