Abstract
There is evidence of testosterone having deteriorating effects on cognitive and affective empathic behaviour in men and women under varying conditions. However, whether testosterone influences empathy for pain has not yet been investigated. Therefore, we tested neural responses to witnessing others in pain in a within-subject placebo-controlled testosterone administration study in healthy young women. Using functional magnetic resonance imaging, we provide affirming evidence that an empathy-inducing paradigm causes changes in the activity throughout the pain circuitry, including the bilateral insula and anterior cingulate cortex. Administration of testosterone, however, did not influence these activation patterns in the pain matrix. Testosterone has thus downregulating effects on aspects of empathic behaviour, but based on these data does not seem to influence neural responses during empathy for others’ pain. This finding gives more insight into the role of testosterone in human empathy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The steroid hormone testosterone (T) is involved in a broad repertoire of human social–emotional behaviour (Bos et al. 2012), and plays a critical role in the sexual differentiation of such behaviour (Auyeung et al. 2009). For example, T administration studies have shown T to reduce fear responses (Hermans et al. 2006a, b), reduce trust in others (Bos et al. 2010), reduce submissive gazing (Terburg Aarts and van Honk 2012), and increase aggressive responses to others (Carre et al. 2016). Overall, T seems to increase status seeking and dominance motivation (Eisenegger Haushofer and Fehr 2011), and facilitate behavioural responses dealing with threat or challenge (Bos et al. 2012). As such, several studies have shown T to reduce empathic behaviour. Empathy is a broad construct that can refer to a range of capacities such as reading the intentions and feelings from others (cognitive empathy), to vicariously experiencing the emotional state of others (affective empathy) (Decety 2011; Panksepp and Panksepp 2013; Preston and de Waal 2002). Empathy thus covers a cognitive–affective continuum (Panksepp and Panksepp 2013), wherein automatic synchronizing with others and the sharing of pain states are key evolutionarily conserved forms of empathy (Decety 2011; Keysers et al. 2010; Preston and de Waal 2002). Hermans et al. (2006a, b) showed reduced facial imitation of the emotional expressions of others, which might reflect reduced emotion sharing, a core aspect of affective empathy. T also reduces mindreading, the cognitive empathic ability whereby one infers the emotional state of the other from limited information. In women, two studies showed reduced performance on the Reading the Mind in the Eyes test after T (RMET: Olsson et al 2016; van Honk et al 2011), and a recent functional magnetic resonance imaging (fMRI) study shows this effect can depend on reduced neural connectivity of the frontal brain regions with the anterior cingulate cortex (ACC) and premotor regions (Bos et al. 2016). In men, the effect of T on cognitive empathy has recently been questioned since a large administration study failed to find an effect on RMET performance in males (Nadler et al. 2019). Thus, whereas these studies show that T reduces the cognitive empathic ability of mindreading in women, as well the motor imitation of facial expressions, a core aspect of affective sharing—empathy for pain—has not yet been studied. Empathy for pain is an evolutionarily conserved emotional response that has been recorded in rodents (Langford et al. 2006). Indeed, responses of emotional state sharing have been noted consistently in mammals, and are thought to be crucial to survival of individuals (Sivaselvachandran et al. 2016).
A meta-analysis on the topic identified a core empathy for pain network comprising the bilateral insula and medial/anterior cingulate cortex, with other regions being additionally recruited depending on the functional paradigm being utilised, for example, somatosensory regions were activated when the task used was picture based, and specifically, viewing body parts in pain recruited the inferior parietal/ventral premotor cortices (Lamm Decety and Singer 2011). Additionally, the bilateral insula and rostral ACC have activated in response to receiving a painful stimulus, and also in response to the knowledge of a loved one being in pain, while the sensorimotor regions and caudal ACC were activated only when receiving pain directly (Singer et al. 2004). The sensorimotor cortex and SMA, however, were noted to have increased activity when viewing others in pain (Decety et al. 2008) as well as the ACC and insula. Thus, empathy for pain consists of several components, reflected in distinct yet often co-occurring patterns of neural activation (Betti and Aglioti 2016). The sensory component elicits activation in primary and secondary sensory—and motor—areas (Keysers et al. 2010), whereas the affective component activates regions such as the ACC and insula. In addition, frontal and parietal regions are involved in the evaluation of higher order processing and empathy eliciting stimuli which can result in cognitive regulation of affective responses and can result in varied behavioural responses ranging from sympathy to schadenfreude (Betti and Aglioti 2016; Timmers et al. 2018; Cikara 2015).
Although activation in the pain circuitry in response to seeing others in pain has repeatedly been observed and is considered a robust phenomenon, it has also been shown sensitive to contextual and endocrine manipulations. Several studies have found physiological responses to pain of others to be modulated by in/outgroup bias. fMRI studies have found decreased activation of the ACC (Xu et al. 2009) and bilateral anterior insula (Azevedo et al. 2013) when viewing outgroup members’ pain, and a TMS study noted a decreased empathic reaction in the corticospinal system when viewing outgroup pain (Avenanti Sirigu and Aglioti 2010). In the latter study, motor-evoked potentials were measured in participants left motor cortex, as corresponding to the task’s right hand observations. Ingroup models elicited an inhibited response, which is more similar to experiencing one’s own pain.
Hormonal factors can also affect empathy for pain, as the neuropeptide oxytocin has shown to decrease neural activation of the pain circuitry upon seeing pain in others, an effect that was independent of group status of the observed other (Bos et al. 2015). In addition, studies showing an association between experienced self-pain and endogenous or exogenous T in men and women (Choi, Chung and Lee 2012; Bartley et al. 2015; White and Robinson 2015) suggest that empathy towards pain in others is also affected by T. But if T alters neural responses to pain in others, and whether this would depend on ethnicity of the outgroup are currently unknown. Therefore, in the current study, we investigate the effect of T on neural empathic responses to pain in others. Based on the effects of T on cognitive empathy and facial mimicry, T might reduce such empathic responses. However, empathy for pain is different from mindreading or mimicry, as illustrated by contrasting findings from oxytocin administration studies showing increased cognitive empathy (e.g. Domes et al. 2007), but reduced neural responses to pain in others (Bos et al. 2015). Effects of T might thus also show divergent effects on these different aspects of empathy. The possible effects of T on neural responses to pain in others can shed some light on the underlying mechanism of observed sex differences in empathic responding. Such sex differences in responses to others’ pain have been established. These differences have been associated with increased brain activity in somato-motor regions in women (Christov-Moore and Iacoboni 2019), and altered centro-parietal activity in men vs women, dependent on social stress (Gonzalez-Liencres et al. 2016). The role of neuroendocrine factors in relation to these observations have, however, not yet been determined.
Methods
Participants
Thirty healthy young females were recruited to participate in the experiment. Ethical approval for human participant recruitment was granted by the Human Research Ethics Committee of the University of Cape Town (ref 092/2011), and the study was performed in accordance with the latest declaration of Helsinki. All participants provided written informed consent and were screened for history of psychiatric conditions. Additional exclusion criteria were current or recent use of psychotropic medications, use of hormonal contraceptives, pregnancy, abnormal menstrual cycle, any endocrine disorders, any other serious medical condition, left handedness, habitual smoking, hearing problems, and colour blindness. After recruitment, data of five participants were lost due to technical problems at the scanning facility, and one participant did not comply with the task instructions, leaving a final sample of 24 participants (age range 18–37, mean age = 21.2, SD = 4.19). Thirteen of the participants were black, three were of mixed ethnicity, and eight were white. In line with the T administration literature (Bos et al. 2012), only women were considered as participants, because the parameters, including quantity and time course, for inducing neurophysiological effects after a single sublingual administration of 0.5 mg of T are known in women, whereas these parameters are not known in men (Gonzalez-Liencres et al. 2016). Each participant was paid ZAR250 (approx. €20) for their participation in this plus two other tasks unrelated to the current task.
Drug administration
Based on the existing literature on T administration, we closely followed the procedure reported by Tuiten et al (2000). The procedure involves the sublingual administration of 0.5 mg of T with a hydroxypropyl-β-cyclodextrin carrier (manufactured by Laboswiss AG, Davos, Switzerland) to healthy young females. This is a well-established single-dose T administration procedure that has been widely used and has been reliably shown to generate behavioural effects in young women (for a review, see (Bos et al. 2012)). For the dosage chosen, no side effects have been reported in this or other studies to date.
Procedure
Participants were tested on two separate days. The first and second sessions were separated by at least 48 h. The average time between sessions was 54.9 h (SD = 16.7). Both testing days fell within the follicular phase of the participants’ menstrual cycles to ensure low and stable basal levels of sex hormones (e.g. progesterone, luteinizing hormone, and follicle stimulating hormone). On both occasions, they arrived at the lab to receive the drug or placebo administration 4 h before their scheduled MRI scan. Participants were instructed not to participate in any activity that may cause excessive fluctuations in hormone production, such as sports games, exams, or sexual activity before returning to the lab to undergo a MRI scan. A flatbed scanner was used to measure participants’ finger lengths. The ratio of the length of the second digit to the fourth digit (2D:4D) is a fixed marker of fetal testosterone exposure (van Honk et al 2011). This experiment was part of a larger study and all participants performed two additional unrelated tasks during the scan session. A task on threat and escape anticipation has been published (Heany et al 2018), and a task on responses to erotic stimuli could not be analysed due to a problem with the experimental script. The present empathy for pain task was completed first, followed by the erotic task, followed finally by the threat task.
Empathy for pain task
To generate pain and no-pain conditions, short video clips of hands were shown to the participant. One hand at a time was viewed on screen in clips of 2.5 s duration. The viewed hand was either being injected by a hypodermic needle (test stimulus) or being gently prodded with an ear bud (control stimulus), as shown in Fig. 1a. The hypodermic needle was fake and no hands were genuinely injected in the making of the task. The tip of the needle or ear bud made contact with the hand 1 s into each video clip. In between each video clip, the participant saw a black fixation cross on a white background for between 3 and 8 s. Hands of two different colours were used; black hands and white hands. This resulted in four stimuli conditions (2 X 2; pain X colour) during the task. The stimuli have been used by our laboratory previously (Bos et al. 2015) and are based on Avenanti et al. (2010). The videos were recorded using a JVC handycam recorder and edited using Adobe Premiere Elements software. The task was presented to participants using e-prime software (version 2; https://pstnet.com). Participants were instructed to pay attention to the stimuli but no further instruction were given.
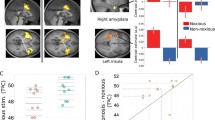
a Pictures from the empathy for pain task. From left to right: the black hand in the pain condition, and the white hand in the no-pain condition. b Coronal brain slices of the T map for the contrast of pain versus no-pain in the combined placebo and testosterone conditions, overlaid onto a T1-weighted canonical image. Accompanying MNI coordinates on the Y-axis are presented below. The T map is thresholded at P < 0.001 (uncorrected) for illustration purposes only. Image created in Adobe Illustrator
fMRI scanning parameters
All scans were obtained using a 3 Tesla Magnetom Allegra Siemens dedicated head MRI scanner (Siemens Medical Systems GmBH, Erlangen, Germany) with a four-channel phased array head coil, at the Cape Universities Brain Imaging Centre. Whole brain T2*-weighted 2D echo planar imaging (EPI) functional volumes were acquired with 36 ascending axial slices. The following parameters were used: EPI factor = 64, TR/TE:2 s/27 ms, FA 70°, FOV (anterior–posterior, inferior-superior, left–right): 64*64*36 slices, voxel size: 3.5 × 3.5 × 4 mm. Five volumes from start of the task were discarded to allow MR signal to stabilize, and 295 usable functional volumes were acquired. A T1-weighted high-resolution structural scan (magnetization-prepared rapid gradient echo; MPRAGE) was obtained once for each participant using the following parameters: TR/TE: 2.53/6.6 ms, flip angle 7°, FOV 256*256*128 mm, voxel size: 1 × 1 × 1.33 mm, volume acquisition time: 8 min 33 s.
fMRI data analysis
MR scans were analysed using SPM8 (Wellcome Department of Imaging Neuroscience, London, UK). Pre-processing included slice-time correction, motion correction of the six motion parameters (Caballero-Gaudes and Reynolds 2017) and the sum of squared difference minimization, volume realignment to the middle volume and AC–PC realignment to improve co-registration. Functional and structural volumes were co-registered and subsequently normalized to standard (MNI152) space using an indirect normalization procedure (Ashburner and Friston 1997) and resampled into 4 mm isotropic voxels using 4th degree B-spline interpolation. To confirm minimal head movement, a frame-wise displacement Matlab script was used separately to assess average percentage scan-to-scan motion to ensure displacement of < 1.1 mm (Power et al. 2012). Finally, all images were smoothed using an 8 mm FWHM Gaussian kernel, which addresses residual between subjects variance.
Statistical analysis of fMRI data at the individual subject level was performed within the general linear model framework. Neural responses to the presentation of the stimuli were modelled using 2.5 s boxcar function convolved with the hemodynamic response function, and these were included as regressors for the four task conditions. They were white/pain; black/pain; white/no-pain; black/no-pain. Six rigid body transformation parameters obtained during realignment were also included as regressors. High-pass filter cut-off was set at 1/128 Hz. For each participant, contrast maps were generated for the main effect of the separate conditions versus rest across sessions which were then entered in the second level model.
Second-level random effects modelling tested the null hypothesis of zero difference across participants between the drug and placebo conditions. Whole-brain and ROI analyses were run using FWE corrected voxel level significance, thresholded at p < 0.05 throughout. A three factor full factorial 2 × 2 × 2 ANOVA (drug/placebo, pain/no-pain, black/white) was modelled using event-related onset times of the task stimuli and tested for whole-brain and ROI effects. In addition, to control for the effect of the 2D:4D, this factor was entered as a covariate in the full factorial ANOVA. Main effects of drug were tested, as well as main effects of pain, stimulus colour, and the interactions between the three factors. Since the chosen statistical threshold (FWE corrected at an alpha of 0.05) can be considered conservative with regard to often subtle effects of endocrine manipulation, we also ran exploratory analysis at an altered threshold of a FWE corrected alpha of 0.1. For visualisation of results, statistical parametric maps have been superimposed onto a high-resolution canonical T1 scan with thresholds set at p < 0.001.
ROI masks
ROI analyses were performed based on existing knowledge of neural regions associated with empathic responses to pain, as well as those regions typically experiencing dissociating effects of T administration. The predefined anatomical ROIs used were the following: anterior insula cortex (AIC), as used in Montoya et al. (2015); supramarginal gyrus, created using the automated anatomical labeling template (AAL, Tzourio-Mazoyer et al. 2002); amygdala, based on the probabilistic atlas from Amunts et al. (2005) as implemented in SPM’s anatomy toolbox (28). Additionally, peak coordinates for the medial ACC (ACC; x = − 2, y = 23, z = 40) and pre-supplementary motor area (pSMA; x = 6, y = 18, z = 48) were taken from a meta-analysis on pain perception (Lamm et al. 2011), and 6 mm circular ROIs were created in Marsbar (Eickhoff et al. 2005). The peak voxel threshold FWE p < 0.05 was used for all ROIs. Additionally, signal beta estimates were extracted for five bilateral ROIs using Marsbar. The beta estimates for the AIC, amygdala, ACC, preSMA, and supramarginal gyri were entered in a repeated-measures ANOVA to test for additional analysis on the main effects of drug and task conditions. Of the 24 participants, 10 received the drug in their first session, and 14 received the placebo in their first session; this asymmetry called for session order to be considered. Session order and participant race were entered as between-subject factor in two separate repeated-measures ANOVAs to control for these factors.
Results
Main effects of task
Overall, the main effect of the pain stimuli compared to the control stimuli resulted in robust activation of the regions incorporated in the ‘pain matrix’. In the whole-brain analysis, we observed increased activation in regions associated with visual processing and detection of body parts, including the bilateral supramarginal gyrus, bilateral middle temporal gyrus, bilateral inferior frontal gyrus as well as bilateral insula (see Table 1 and Fig. 1b). ROI analyses for the same contrast detected additional activation in the medial ACC, and the preSMA. For the opposite contrast, of control stimuli versus pain, regions that activated were the bilateral middle frontal gyrus, left posterior cingulate cortex nearing the left precuneus, and right cuneus, all at the whole brain level. For the main effect of hand colour, we first tested activations in response to viewing black hands versus white hands and observed activated clusters in the right fusiform gyrus, right middle occipital gyrus, right lingual gyrus at the whole brain level, as well as a small cluster in the right amygdala. A few voxels within the supramarginal gyrus showed stronger activation for the opposite contrast of white versus the black (Table 1). Critical to the aim of the current study was the contrast comparing T with the placebo condition or the interactions of drug with the other factors. However, no main effect of drug, or interaction with the other factors reached significance. Contrast T maps are available to be viewed at the following URLs: https://neurovault.org/collections/EPEKJSAG/images/312992/ and https://neurovault.org/collections/FUHSXBXA/images/312994/.
Including participants’ skin colour, 2D:4D, or session order as a covariate in the full factorial did not alter the results. Our more exploratory analyses with an alpha set at 0.1 (FWE corrected) neither showed effect of the factor drug administration nor interactions of drug with another factor.
Additional analyses on extraction of the ROI
The analysis of the extracted beta values of the ROIs confirmed the whole brain analyses. The AIC (p = 0.003, f = 10.704, df = 1,23), preSMA (p = 0.012, f = 7.410, df = 1,23), and supramarginal (p < 0.001, f = 22.114, df = 1,23) responded significantly in the pain condition. However, no other conditions or interactions were significant. Finally, as a control analysis, we ran an additional ANCOVA with 2D:4D ratio as a covariate to test for possible interactions of drug administration with the 2D:4D ratio, and with participant skin colour as a between-subject factor to investigate the effect on differential responses to pain depending on stimulus skin colour. However, the added factors did not significantly interact with the effects of interest.
The above-described ANOVAs give information on whether or not to reject the null hypotheses that T did not affect empathy for pain-related neural activation. However, it gives no information on the likelihood that the null hypothesis is correct. Therefore, we used JASP (version 0.9.0.1., https://jasp-stats.org) to run Bayesian paired-sample t tests comparing the T and placebo condition for both the white and black hands in the three significantly activated ROIs. Bayes Factor (BF) in favour of the null hypotheses compared to the hypothesis that conditions (T and placebo) differed all fell between BF 3.6 and BF 4.6 indicating moderate support for the null hypothesis over the alternative hypothesis.
Discussion
In the current study, we investigated the effect of exogenous T on neural empathic responses to pain in others. As a main finding of the experiment, the current paradigm showed robust activation in young women of regions associated with pain processing when witnessing others in pain such as the insula, ACC, and pre-SMA. These results are in line with previous studies using stimuli that elicit neural empathic responses to pain in others (Azevedo et al. 2013; Gonzalez-Liencres et al. 2016; Lamm Batson and Decety 2007). Most importantly, however, our study investigated the effect of T administration in women on the observed neural responses towards pain in others. In our current within-subject sample of 24 participants, no such effects were found, whereas Bayesian analyses on the extracted data provided moderate evidence that T does not affect neural responses to pain observed in others.
Based on previous studies that have demonstrated downregulating effects of T in similar populations on empathic behaviour (Hermans Putman and van Honk 2006; Olsson et al. 2016; van Honk et al. 2011), it was hypothesized that T would attenuate neural responses to pain in others. However, no effects of T on neural responses to pain in others were observed. An important notion with regard to these findings is that the previous behavioural studies measured cognitive empathic behaviour (‘mindreading’; Olsson et al. 2016; Bos et al. 2016) and automatic facial imitation (Hermans et al. 2006a, b), but not empathy for pain in others. Thus, it is currently unknown whether T alters behavioural responses to pain in others, and the exact relation between neural responses to others’ pain in the pain circuitry and behavioural responses is also unclear. As described above, oxytocin can under some conditions increase empathic subjective responses to pain in others (Abu-Akel et al. 2015), yet this neuropeptide strongly decreases neural responses in the pain circuitry when seeing pain in others (Bos et al. 2015). It could be that the observed neural responses towards pain in others reflect aversiveness and personal distress (Abu-Akel et al. 2015; Decety and Lamm 2011), which if reduced by oxytocin can result in more appropriate helping behaviour (Hayes and Nothoff 2012). Relating to our current findings, it might thus be that T does not affect aversive responding. Although with regards to personal distress, some research has noted that T is associated with reduced pain perception in men and women (Choi, Chung and Lee 2012; Bartley et al. 2015; White and Robinson 2015). This interpretation highlights the importance of publishing null findings, especially in the light of the current criticism on the possible publication bias in oxytocin administration studies (Lane et al. 2016). Future studies combining T administration with sensitive measures of behavioural responding to pain in others should, however, test the validity of our interpretation. A study of connectivity within the pain circuitry could potentially also add insight into the prefrontal–subcortical relationship that has previously been modulated in T administration studies. This approach would require ROI-based analyses of connectivity, including a focus on the amygdala as is common in T studies, but which was not noted in our whole-brain analyses.
Given that previous studies have observed robust changes (indexed by medium-to-large effect sizes) in neural activation after the same T administration as currently used in within-subject designs with smaller samples (Bos et al. 2013; Hermans 2010), we can be confident that our current study setup has the power to detect effects of similar magnitude. That being said, our current sample size is less sensitive to detect small, or small-to-medium effect sizes, and thus replication of this null finding with larger samples is needed to exclude more subtle effects of testosterone on the neural correlates of pain observed in others. Also, recent work on oxytocin has shown differences in the effects of drug administration depending on whether the study followed a within- or between-subject design, and showed more robust effects of drug in the between-subject design (Chen et al. 2017). Since the current study follows a within-subject design, this could also be taken into account in future studies.
Irrespective of the effect of drug, we did encounter effect of hand colour of the stimuli on our data. Relative to stimuli of white hands, black hand stimuli activated the lingual gyrus and fusiform gyrus, two regions that are well established to be related to visual processing of human forms (Kozlovskiy et al. 2014; McCarthy et al. 1997). Fusiform gyrus activation noted in this contrast has also been noted in work that has found increased activation when viewing visuals of ingroup members (Golby et al. 2001; Lieberman et al. 2005). In addition, we observed increased activation of the amygdala for the black versus white hands. Although amygdala activation has been related to ingroup bias (Van Bavel et al. 2008), this is unlikely the case in our current study. First, the effects of skin colour of the stimuli did not interact with the pain condition, and were thus similar for the pain and no-pain condition, more likely reflecting differences in visual processing. Furthermore, the effects of colour of the stimuli were unrelated to participants skin colour. Our sample was purposefully selected to consist of participants of different ethnicity to increase the generalizability of the findings (black: n = 13; white: n = 8; mixed ethnicity: n = 3), and this also allowed us to test whether participants’ skin colour predicted the effect neural responses to images of black or white hands in pain. However, extracted data from the ROIs showed no interaction between participant colour with pain that depended on the skin colour of the stimuli, indicating that the neural responses of students of different ethnicity to the black and white stimuli was similar. Thus, our current data do not replicate previous studies that found differential effects towards pain in others depending on race (Avenanti et al. 2010; Azevedo et al. 2013; Xu et al. 2009). Cultural differences between European and Asian students and those born into a post-apartheid South Africa might account for these differences. It must, however, be noted that the current study was not set up to investigate ethnicity biases in empathic responding to pain in others but focused on the effect of T, as the groups were not balanced for participant ethnicity, and the current sample size limits the power to detect subtle effects of ethnicity on the data.
In conclusion, corroborating previous findings, robust task effects were observed in women in brain regions involved in the processing of pain when observing pain in others. This activation pattern was, however, unaffected by our placebo-controlled T administration. Since neural responses towards pain in others might particularly signal aversiveness and experienced personal distress (Decety and Lamm 2011; Lamm et al. 2007), it could be that T does not affect this particular aspect of empathic responding, in contrast to the downregulating effects T has on cognitive empathic abilities (Olsson et al. 2016; van Honk et al. 2011).
References
Abu-Akel A, Palgi S, Klein E, Decety J, Shamay-Tsoory S (2015) Oxytocin increases empathy to pain when adopting the other-but not the self-perspective. Soc Neurosci 10(1):7–15
Amunts K, Kedo O, Kindler M, Pieperhoff P, Mohlberg H, Shah NJ, Zilles K (2005) Cytoarchitectonic mapping of the human amygdala, hippocampal region and entorhinal cortex: intersubject variability and probability maps. Anat Embryol 210(5–6):343–352. https://doi.org/10.1007/s00429-005-0025-5
Ashburner J, Friston K (1997) Multimodal image coregistration and partitioning–a unified framework. NeuroImage 6(3):209–217. https://doi.org/10.1006/nimg.1997.0290
Auyeung B, Baron-Cohen S, Ashwin E, Knickmeyer R, Taylor K, Hackett G, Hines M (2009) Fetal testosterone predicts sexually differentiated childhood behavior in girls and in boys. Psychol Sci 20(2):144–148. https://doi.org/10.1111/j.1467-9280.2009.02279
Avenanti A, Sirigu A, Aglioti SM (2010) Racial bias reduces empathic sensorimotor resonance with other-race pain. Curr Biol 20(11):1018–1022. https://doi.org/10.1016/j.cub.2010.03.071
Azevedo RT, Macaluso E, Avenanti A, Santangelo V, Cazzato V, Aglioti SM (2013) Their pain is not our pain: Brain and autonomic correlates of empathic resonance with the pain of same and different race individuals. Hum Brain Mapp 34(12):3168–3181. https://doi.org/10.1002/hbm.22133
Bartley EJ, Palit S, Kuhn BL, Kerr KL, Terry EL, DelVentura JL, Rhudy JL (2015) Natural variation in testosterone is associated with hypoalgesia in healthy women. Clin J pain 31(8):730–739
Betti V, Aglioti SM (2016) Dynamic construction of the neural networks underpinning empathy for pain. Neurosci Biobehav Rev 63:191–206
Bos PA, Terburg D, van Honk J (2010) Testosterone decreases trust in socially naive humans. Proc Natl Acad Sci USA 107(22):9991–9995. https://doi.org/10.1073/pnas.0911700107
Bos PA, Panksepp J, Bluthe R-M, van Honk J (2012) Acute effects of steroid hormones and neuropeptides on human social-emotional behavior: a review of single administration studies. Front Neuroendocrinol 33(1):17–35. https://doi.org/10.1016/j.yfrne.2011.01.002
Bos PA, van Honk J, Ramsey NF, Stein DJ, Hermans EJ (2013) Testosterone administration in women increases amygdala responses to fearful and happy faces. Psychoneuroendocrinology 38(6):808–817. https://doi.org/10.1016/j.psyneuen.2012.09.005
Bos PA, Montoya ER, Hermans EJ, Keysers C, van Honk J (2015) Oxytocin reduces neural activity in the pain circuitry when seeing pain in others. NeuroImage 113:217–224. https://doi.org/10.1016/j.neuroimage.2015.03.049
Bos PA, Hofman D, Hermans EJ, Montoya ER, Baron-Cohen S, van Honk J (2016) Testosterone reduces functional connectivity during the “Reading the Mind in the Eyes” Test. Psychoneuroendocrinology 68:194–201. https://doi.org/10.1016/j.psyneuen.2016.03.006
Caballero-Gaudes C, Reynolds RC (2017) Methods for cleaning the BOLD fMRI signal. Neuroimage 154:128–149
Carre JM, Geniole SN, Ortiz TL, Bird BM, Videto A, Bonin PL (2016) Exogenous testosterone rapidly increases aggressive behavior in dominant and impulsive men. Biol Psychiat. https://doi.org/10.1016/j.biopsych.2016.06.009
Chen X, Gautam P, Haroon E, Rilling JK (2017) Within vs. between-subject effects of intranasal oxytocin on the neural response to cooperative and non-cooperative social interactions. Psychoneuroendocrinology 78:22–30
Choi JC, Chung MI, Lee YD (2012) Modulation of pain sensation by stress-related testosterone and cortisol. Anaesthesia 67(10):1146–1151
Christov-Moore L, Iacoboni M (2019) Sex differences in somatomotor representations of others’ pain: a permutation-based analysis. Brain Str Funct 224(2):937–947
Cikara M (2015) Intergroup schadenfreude: Motivating participation in collective violence. Curr Opin Behav Sci 3:12–17
Decety J (2011) Dissecting the neural mechanisms mediating empathy. Emot Rev 3(1):92–108
Decety JE, Lamm C (2009) Empathy versus personal distress: recent evidence from social neuroscience. In: Decety JE, Ickes WE (eds) The social neuroscience of empathy. MIT Press, Cambridge, Massachusetts, pp 199–213
Decety J, Michalska KJ, Akitsuki Y (2008) Who caused the pain An fMRI investigation of empathy and intentionality in children. Neuropsychologia 46(11):2607–2614. https://doi.org/10.1016/j.neuropsychologia.2008.05.026
Domes G, Heinrichs M, Michel A, Berger C, Herpertz SC (2007) Oxytocin improves “mind-reading” in humans. Biol Psychiat 61(6):731–733. https://doi.org/10.1016/j.biopsych.2006.07.015
Eickhoff SB, Stephan KE, Mohlberg H, Grefkes C, Fink GR, Amunts K, Zilles K (2005) A new SPM toolbox for combining probabilistic cytoarchitectonic maps and functional imaging data. NeuroImage 25(4):1325–1335. https://doi.org/10.1016/j.neuroimage.2004.12.034
Eisenegger C, Haushofer J, Fehr E (2011) The role of testosterone in social interaction. Trends Cogn Sci 15(6):263–271. https://doi.org/10.1016/j.tics.2011.04.008
Golby AJ, Gabrieli JDE, Chiao JY, Eberhardt JL (2001) Differential responses in the fusiform region to same-race and other-race faces. Nat Neurosci 4(8):845–850
Gonzalez-Liencres C, Breidenstein A, Wolf OT, Brüne M (2016) Sex-dependent effects of stress on brain correlates to empathy for pain. Int J Psychophysiol 105:47–56
Hayes DJ, Northoff G (2012) Common brain activations for painful and non-painful aversive stimuli. BMC Neurosci 13(1):60
Heany SJ, Bethlehem RAI, van Honk J, Bos PA, Stein DJ, Terburg D (2018) Effects of testosterone administration on threat and escape anticipation in the orbitofrontal cortex. Psychoneuroendocrinology 96:42–51
Hermans EJ, Putman P, Baas JM, Koppeschaar HP, van Honk J (2006a) A single administration of testosterone reduces fear-potentiated startle in humans. Biol Psychiat 59(9):872–874. https://doi.org/10.1016/j.biopsych.2005.11.015
Hermans EJ, Putman P, van Honk J (2006b) Testosterone administration reduces empathetic behavior: a facial mimicry study. Psychoneuroendocrinology 31(7):859–866. https://doi.org/10.1016/j.psyneuen.2006.04.002
Hermans EJ, Bos PA, Ossewaarde L, Ramsey NF, Fernández G, van Honk J (2010) Effects of exogenous testosterone on the ventral striatal BOLD response during reward anticipation in healthy women. NeuroImage 52(1):277–283. https://doi.org/10.1016/j.neuroimage.2010.04.019
Keysers C, Kaas JH, Gazzola V (2010) Somatosensation in social perception. Nat Rev Neurosci 11(6):417
Kozlovskiy SA, Pyasik MM, Korotkova AV, Vartanov AV, Glozman JM, Kiselnikov AA (2014) Activation of left lingual gyrus related to working memory for schematic faces. Int J Psychophysiol 94(2):241. https://doi.org/10.1016/j.ijpsycho.2014.08.928
Lamm C, Batson CD, Decety J (2007) The neural substrate of human empathy: effects of perspective-taking and cognitive appraisal. J Cogn Neurosci 19(1):42–58. https://doi.org/10.1162/jocn.2007.19.1.42
Lamm C, Decety J, Singer T (2011) Meta-analytic evidence for common and distinct neural networks associated with directly experienced pain and empathy for pain. NeuroImage 54(3):2492–2502. https://doi.org/10.1016/j.neuroimage.2010.10.014
Lane A, Luminet O, Nave G, Mikolajczak M (2016) Is there a publication bias in behavioural intranasal oxytocin research on humans? Opening the file drawer of one laboratory. J Neuroendocrinol. https://doi.org/10.1111/jne.12384
Langford DJ, Crager SE, Shehzad Z, Smith SB, Sotocinal SG, Levenstadt JS, Mogil JS (2006) Social modulation of pain as evidence for empathy in mice. Science 312(5782):1967–1970. https://doi.org/10.1126/science.1128322
Lieberman MD, Hariri A, Jarcho JM, Eisenberger NI, Bookheimer SY (2005) An fMRI investigation of race-related amygdala activity in African-American and Caucasian-American individuals. Nat Neurosci 8(6):720–722
McCarthy G, Puce A, Gore JC, Allison T (1997) Face-specific processing in the human fusiform gyrus. J Cogn Neurosci 9(5):605–610
Montoya ER, van Honk J, Bos PA, Terburg D (2015) Dissociated neural effects of cortisol depending on threat escapability. Hum Brain Mapp 36(11):4304–4316. https://doi.org/10.1002/hbm.22918
Nadler A, Camerer CF, Zava DT, Ortiz TL, Watson NV, Carré JM, Nave G (2019) Does testosterone impair men's cognitive empathy?Evidence from two large-scale randomized controlled trials. Proc R Soc B 286(1910):20191062
Olsson A, Kopsida E, Sorjonen K, Savic I (2016) Testosterone and estrogen impact social evaluations and vicarious emotions: A double-blind placebo-controlled study. Emotion 16(4):515–523. https://doi.org/10.1037/a0039765
Panksepp J, Panksepp JB (2013) Toward a cross-species understanding of empathy. Trends Neurosci 36:489–496. https://doi.org/10.1016/j.tins.2013.04.009
Power JD, Barnes KA, Snyder AZ, Schlaggar BL, Petersen SE (2012) Spurious but systematic correlations in functional connectivity MRI networks arise from subject motion. Neuroimage 59(3):2142–2154
Preston SD, de Waal FB (2002) Empathy: Its ultimate and proximate bases. Behav Brain Sci 25:1–71
Singer T, Seymour B, ODoherty J, Kaube H, Dolan RJ, Frith CD (2004) Empathy for pain involves the affective but not sensory components of pain. Science 303(5661):1157–1162
Sivaselvachandran S, Acland EL, Abdallah S, Martin LJ (2016) Behavioral and mechanistic insight into rodent empathy. Neurosci Biobehav Rev. https://doi.org/10.1016/j.neubiorev.2016.06.007
Terburg D, Aarts H, van Honk J (2012) Testosterone affects gaze aversion from angry faces outside of conscious awareness. Psychol Sci 23(5):459–463. https://doi.org/10.1177/0956797611433336
Timmers I, Park AL, Fischer MD, Kronman CA, Heathcote LC, Hernandez JM, Simons LE (2018) Is empathy for pain unique in its neural correlates? A meta-analysis of neuroimaging studies of empathy. Front Behav Neurosci 12:289
Tuiten A, van Honk J, Koppeschaar H, Bernaards C, Thijssen J, Verbaten R (2000) Time course of effects of testosterone administration on sexual arousal in women. Arch Gen Psychiatry 57(2):146–149
Tzourio-Mazoyer N, Landeau B, Papathanassiou D, Crivello F, Etard O, Delcroix N, Joliot M (2002) Automated anatomical labeling of activations in SPM using a macroscopic anatomical parcellation of the MNI MRI single-subject brain. NeuroImage 15(1):273–289. https://doi.org/10.1006/nimg.2001.0978
Van Bavel JJ, Packer DJ, Cunningham WA (2008) The neural substrates of in-group bias: A functional magnetic resonance imaging investigation. Psychol Sci 19(11):1131–1139
van Honk J, Schutter DJ, Bos PA, Kruijt A-W, Lentjes EG, Baron-Cohen S (2011) Testosterone administration impairs cognitive empathy in women depending on second-to-fourth digit ratio. Proc Natl Acad Sci USA 108(8):3448–3452. https://doi.org/10.1073/pnas.1011891108
White HD, Robinson TD (2015) A novel use for testosterone to treat central sensitization of chronic pain in fibromyalgia patients. Int Immunopharmacol 27(2):244–248
Xu X, Zuo X, Wang X, Han S (2009) Do you feel my pain? Racial group membership modulates empathic neural responses. J Neurosci 29(26):8525–8529. https://doi.org/10.1523/JNEUROSCI.2418-09.2009
Acknowledgements
The work in this paper was supported by a grant from the Netherlands Society of Scientific Research to PAB (451-14-015), to DT (451-13-004) and to JvH (056-24-010). DJS is supported by the Medical Research Council of South Africa has received research grants and/or consultancy honoraria from Abbott, Astrazeneca, Eli-Lilly, GlaxoSmithKline, Jazz Pharmaceuticals, Johnson & Johnson, Lundbeck, Orion, Pfizer, Pharmacia, Roche, Servier, Solvay, Sumitomo, Takeda, Tikvah, and Wyeth. SH is supported by Postgraduate Funding Office of UCT, and the Oppenheimer Memorial Trust, and the National Research Foundation of South Africa (NRF). The funding sources had no further role in study design; in the collection, analysis and interpretation of the data; in the writing of the report; or the decision to submit the paper for publication.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by John C. Rothwell.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Heany, S.J., Terburg, D., Stein, D.J. et al. Neural responses in the pain matrix when observing pain of others are unaffected by testosterone administration in women. Exp Brain Res 238, 751–759 (2020). https://doi.org/10.1007/s00221-020-05749-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-020-05749-3