Abstract
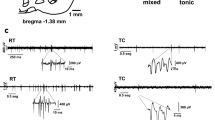
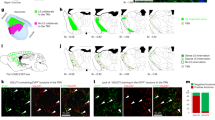
The thalamic reticular nucleus (TRN) modulates activity in the thalamus and controls excitatory input from corticothalamic and thalamocortical glutamatergic projections. It is made up of GABAergic neurons which project topographically to the thalamus. The TRN also receives inhibitory projections from the globus pallidus and the substantia nigra pars reticulata. Photostimulation of the TRN results in local inhibition in rat slice preparations but the effects of local stimulation in vivo are not known. This study aimed to characterize stimulation-evoked responses in the TRN of non-human primates (NHPs). Microelectrodes were inserted into the TRN and neurons were stimulated at 5, 10, 15, and 20 µA using 0.5 s trains at 100 Hz and the subsequent response was recorded from the same electrode. Stimulation in surrounding nuclei and the internal capsule was used for mapping the anatomical borders of the TRN. Stimulation as low as 10 µA resulted in predominantly inhibition, recorded in both single units and background unit activity (BUA). The duration of inhibition (~ 1–3 s) increased with increasing stimulation amplitude and was significantly increased in BUA when single units were present. At 20 µA of current, 93% of the single units (41/44) and 92% of BUA sites (67/73) were inhibited. Therefore, microstimulation of the NHP TRN with low currents results in current-dependent inhibition of single units and BUA. This finding may be useful to further aid in localization of deep thalamic and subthalamic nuclei during brain surgery.



Similar content being viewed by others
Abbreviations
- BUA:
-
Background unit activity
- NHP:
-
Non-human primate
- STN:
-
Subthalamic nucleus
- TRN:
-
Thalamic reticular nucleus
References
Anthofer J, Steib K, Fellner C, Lange M, Brawanski A, Schlaier J (2014) The variability of atlas-based targets in relation to surrounding major fibre tracts in thalamic deep brain stimulation. Acta Neurochir (Wien) 156(8):1497–1504. https://doi.org/10.1007/s00701-014-2103-z
Asanuma C (1994) GABAergic and pallidal terminals in the thalamic reticular nucleus of squirrel monkeys. Exp Brain Res 101:439–451. https://doi.org/10.1007/BF00227337
Azimi K, Prescott IA, Marino RA, Winterborn A, Levy R (2016) Low profile head fixation in non-human primates. J Neurosci Methods 268:23–30. https://doi.org/10.1016/j.jneumeth.2016.04.018
Bazhenov M, Timofeev I, Steriade M, Sejnowski TJ (1999) Self-sustained rhythmic activity in the thalamic reticular nucleus mediated by depolarizing GABAA receptor potentials. Nat Neurosci 2:168–174. https://doi.org/10.1038/5729
Benabid AL, Pollak P, Gao D et al (1996) Chronic electrical stimulation of the ventralis intermedius nucleus of the thalamus as a treatment of movement disorders. J Neurosurg 84:203–214
Benabid AL, Chabardes S, Mitrofanis J, Pollak P (2009) Deep brain stimulation of the subthalamic nucleus for the treatment of Parkinson’s disease. Lancet Neurol 8:67–81. https://doi.org/10.1016/S1474-4422(08)70291-6
Blomstedt P, Sandvik U, Tisch S (2010) Deep brain stimulation in the posterior subthalamic area in the treatment of essential tremor. Mov Disord 25:1350–1356
Brunenberg EJ, Platel B, Hofman PA, Ter Haar Romeny BM, Visser-Vandewalle V (2011) Magnetic resonance imaging techniques for visualization of the subthalamic nucleus. J Neurosurg 115(5):971–984. https://doi.org/10.3171/2011.6.JNS101571
Buford JA, Inase M, Anderson ME (1996) Contrasting locations of pallidal-receiving neurons and microexcitable zones in primate thalamus. J Neurophysiol 75:1105–1116
Crist CF, Yamasaki DS, Komatsu H, Wurtz RH (1988) A grid system and a microsyringe for single cell recording. J Neurosci Methods 26:117–122
Dostrovsky JO, Levy R, Wu JP, Hutchison WD, Tasker RR, Lozano AM (2000) Microstimulation-induced inhibition of neuronal firing in human globus pallidus. J Neurophysiol 84:570–574
Gentet LJ, Ulrich D (2003) Strong, reliable and precise synaptic connections between thalamic relay cells and neurones of the nucleus reticularis in juvenile rats. J Physiol 546:801–811. https://doi.org/10.1113/jphysiol.2002.032730
Gentet LJ, Ulrich D (2004) Electrophysiological characterization of synaptic connections between layer VI cortical cells and neurons of the nucleus reticularis thalami in juvenile rats. Eur J Neurosci 19:625–633. https://doi.org/10.1111/j.1460-9568.2004.03168.x
Gonzalo-Ruiz A, Lieberman AR (1995) GABAergic projections from the thalamic reticular nucleus to the anteroventral and anterodorsal thalamic nuclei of the rat. J Chem Neuroanat 9:165–174. https://doi.org/10.1016/0891-0618(95)00078-X
Halassa MM, Chen Z, Wimmer RD, Brunetti PM, Zhao S, Zikopoulos B, Wang F, Brown EN, Wilson MA (2014) State-dependent architecture of thalamic reticular subnetworks. Cell 158(4):808–821. https://doi.org/10.1016/j.cell.2014.06.025
Hazrati LN, Parent A (1991) Projection from the external pallidum to the reticular thalamic nucleus in the squirrel monkey. Brain Res 550:142–146. https://doi.org/10.1016/0006-8993(91)90418-U
Hou G, Smith AG, Zhang ZW (2016) Lack of intrinsic GABAergic connections in the thalamic reticular nucleus of the mouse. J Neurosci 36(27):7246–7252. https://doi.org/10.1523/jneurosci.0607-16.2016
Jones EG (1975) Some aspects of the organization of the thalamic reticular complex. J Comp Neurol 162:285–308. https://doi.org/10.1002/cne.901620302
Kultas-Ilinsky K, Yi H, Ilinsky IA (1995) Nucleus reticularis thalami input to the anterior thalamic nuclei in the monkey: a light and electron microscopic study. Neurosci Lett 186:25–28. https://doi.org/10.1016/0304-3940(95)11273-Y
Lafreniere-Roula M, Hutchison WD, Lozano AM, Hodaie M, Dostrovsky JO (2009) Microstimulation-induced inhibition as a tool to aid targeting the ventral border of the subthalamic nucleus. J Neurosurg 111:724–728. https://doi.org/10.3171/2009.3.JNS09111
Lam YW, Nelson CS, Sherman SM (2006) Mapping of the functional interconnections between thalamic reticular neurons using photostimulation. J Neurophysiol 96:2593–2600. https://doi.org/10.1152/jn.00555.2006
Landisman CE, Long MA, Beierlein M, Deans MR, Paul DL, Connors BW (2002) Electrical synapses in the thalamic reticular nucleus. J Neurosci 22(3):1002–1009
Lee KH, McCormick DA (1995) Acetylcholine excites GABAergic neurons of the ferret perigeniculate nucleus through nicotinic receptors. J Neurophysiol 73:2123–2128. https://doi.org/10.1152/jn.1995.73.5.2123
Legendy CR, Salcman M (1985) Bursts and recurrences of bursts in the spike trains of spontaneously active striate cortex neurons. J Neurophysiol 53:926–939
Lozano A, Hutchison W, Kiss Z, Tasker R, Davis K, Dostrovsky J (1996) Methods for microelectrode-guided posteroventral pallidotomy. J Neurosurg 84:194–202. https://doi.org/10.3171/jns.1996.84.2.0194
Marino RA, Rodgers CK, Levy R, Munoz DP (2008) Spatial relationships of visuomotor transformations in the superior colliculus map. J Neurophysiol 100:2564–2576. https://doi.org/10.1152/jn.90688.2008
McAlonan K, Cavanaugh J, Wurtz RH (2006) Attentional modulation of thalamic reticular neurons. J Neurosci 26(16):4444–4450
Moran A, Bar-Gad I (2010) Revealing neuronal functional organization through the relation between multi-scale oscillatory extracellular signals. J Neurosci Methods 186:116–129. https://doi.org/10.1016/j.jneumeth.2009.10.024
Novak P, Daniluk S, Ellias SA, Nazzaro JM (2007) Detection of the subthalamic nucleus in microelectrographic recordings in Parkinson disease using the high-frequency (> 500 Hz) neuronal background. Technical note. J Neurosurg 106:175–179
Pare D, Hazrati LN, Parent A, Steriade M (1990) Substantia nigra pars reticulata projects to the reticular thalamic nucleus of the cat: a morphological and electrophysiological study. Brain Res 535:139–146. https://doi.org/10.1016/0006-8993(90)91832-2
Paxinos G, Huang X, Toga AW (1999) The rhesus monkey brain in stereotaxic coordinates. Academic Press, San Diego
Pazo JH, Barceló AC, Bellantonio E, Pazo VC, Almarares N (2013) Electrophysiologic study of globus pallidus projections to the thalamic reticular nucleus. Brain Res Bull 94:82–89. https://doi.org/10.1016/j.brainresbull.2013.02.009
Pinault D (2004) The thalamic reticular nucleus: structure, function and concept. Brain Res Brain Res Rev 46:1–31. https://doi.org/10.1016/j.brainresrev.2004.04.008
Pollin B, Rokyta R (1982) Somatotopic organization of nucleus reticularis thalami in chronic awake cats and monkeys. Brain Res 250(2):211–221 (PMID: 7171987)
Prescott IA, Marino RA, Levy R (2017) Field evoked potentials in the globus pallidus of non-human primates. Neurosci Res 120:18–27. https://doi.org/10.1016/j.neures.2017.01.007
Raeva S, Lukashev A (1993) Unit activity in human thalamic reticularis neurons. II. Activity evoked by significant and non-significant verbal or sensory stimuli. Electroencephalogr Clin Neurophysiol 86(2):110–122 (PMID:7681378)
Raeva S, Lukashev A, Lashin A (1991) Unit activity in human thalamic reticularis nucleus. I. Spontaneous activity. Electroencephalogr Clin Neurophysiol 79(2):133–140 (PMID: 713827)
Ranck JB (1975) Which elements are excited in electrical stimulation of mammalian central nervous system: a review. Brain Res 98:417–440. https://doi.org/10.1056/NEJMra1112830
Sanchez-Vives MV, Bal T, McCormick DA (1997) Inhibitory interactions between perigeniculate GABAergic neurons. J Neurosci 17:8894–8908
Scheibel ME, Scheibel AB (1972) Specialized organizational patterns within the nucleus reticularis thalami of the cat. Exp Neurol 34:316–322
Shu Y, McCormick DA (2002) Inhibitory interactions between ferret thalamic reticular neurons. J Neurophysiol 87:2571–2576. https://doi.org/10.1152/jn.00850.2001
Steriade M, Parent A, Hada J (1984) Thalamic projections of nucleus reticularis thalami of cat: a study using retrograde transport of horseradish peroxidase and fluorescent tracers. J Comp Neurol 229:531–547. https://doi.org/10.1002/cne.902290407
Ulrich D, Huguenard JR (1995) Purinergic inhibition of GABA and glutamate release in the thalamus: implications for thalamic network activity. Neuron 15:909–918. https://doi.org/10.1016/0896-6273(95)90181-7
Villalobos N, Oviedo-Chávez A, Alatorre A, Ríos A, Barrientos R, Delgado A, Querejeta E (2016) Striatum and globus pallidus control the electrical activity of reticular thalamic nuclei. Brain Res 1644:258–266. https://doi.org/10.1016/j.brainres.2016.05.032
Zhang L, Jones EG (2004) Corticothalamic inhibition in the thalamic reticular nucleus. J Neurophysiol 91:759–766. https://doi.org/10.1152/jn.00624.2003
Zhang SJ, Huguenard JR, Prince DA (1997) GABAA receptor-mediated Cl-currents in rat thalamic reticular and relay neurons. J Neurophysiol 78:2280–2286. https://doi.org/10.1073/pnas.0712231105
Acknowledgements
Thank you to Dr. Rob Marino and Kathryn McIntosh for comments, Sean Hickman and Jennifer Green for technical support.
Funding
The presented research was funded by a Canada Foundation for Innovation Grant (#31804).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors report no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lymer, J., Prescott, I.A. & Levy, R. Microstimulation-induced inhibition of thalamic reticular nucleus in non-human primates. Exp Brain Res 237, 1511–1520 (2019). https://doi.org/10.1007/s00221-019-05526-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-019-05526-x