Abstract
Vicine and convicine may be removed from faba bean by hydrolysis to the corresponding aglycones, divicine and isouramil. For total elimination of their toxicity, further degradation of the aglycones should be shown. The aim of the study was to investigate hydrolysis of vicine and convicine using the enzymatic activity in faba bean in flour suspensions and selected lactic acid bacteria used as starters for faba bean fermentation. In addition, the effect of acidity on the stability of vicine and convicine was investigated. Sourdoughs were used in a baking process to obtain breads as final products. Vicine, convicine, and their aglycones were analyzed using reversed phase high pressure liquid chromatography with UV detection (RP-HPLC-UV). Incubation of the suspensions showed rather small vicine and convicine losses. Acidity itself did not cause losses under the conditions studied, apart from that of convicine at low pH. In sourdough fermentation with strains of Lactobacillus plantarum and Pediococcus pentosaceus, losses of vicine and convicine were dependent on the fermentation temperature and the β-glucosidase activity of the starter. Compared to fermentation at 20 °C, more intense acidification at 25 °C resulted in decrease of vicine up to 85% and convicine up to 47%. Levels of vicine and convicine in breads were comparable to levels in sourdoughs. Furthermore, the aglycones were not detected from breads.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Grain legumes have been minor crops among the high-yield cereals and oilseeds that have notable fertilizer requirements. Alternatives for imported soybean are needed, and grain legumes, including pea, faba bean, lupine and lentil could be grown locally in Europe [1]. Faba bean has a high protein yield and an acceptable yield stability compared to other protein-rich crops [1]. There is growing interest in plant-based proteins in human nutrition [2]. Addition of faba bean flour to cereals can increase their protein content and supplement nutritional quality by, for example, complementing the amino acid profile.
Faba bean contains certain harmful compounds that have not been reduced notably via plant breeding [3, 4]. The pyrimidine glycosides, vicine and convicine, are under particular observation, since they cause hemolytic anemia called favism for people who have genetic deficiency of the glucose-6-phosphate dehydrogenase (G6PD)-enzyme [5, 6]. Vicine and convicine are hydrolyzed to their corresponding reactive aglycones, divicine and isouramil, which have strong oxidative capacity [7]. Practical processing methods for their removal should be applied, yet they are relatively heat stable, and their complete removal is challenging [8, 9].
As one approach, vicine and convicine are hydrolyzed to aglycones using endogenous or added β-glucosidase enzyme or using fermentation with microbes that are able to synthesize this enzyme [10,11,12,13,14]. When the toxic aglycones are released, they are supposed to react and decompose rapidly due to their instability and thus lose their toxicity [15,16,17,18]. The potential of the β-glucosidase from almonds to hydrolyze vicine and convicine in food systems has been demonstrated in earlier studies [10, 12]. In suspensions, the aglycones were almost completely lost in 2 h at pH 5 and 37 °C with added β-glucosidase [12]. In addition to the β-glucosidases (β-d-glucoside glucohydrolase, EC 3.2.1.21), acidity has been suggested as inducing hydrolysis of the β-glycosidic bond of vicine and convicine, especially when heated to 100 °C [17].
Endogenous β-glucosidases participate in many reactions in plants, e.g., they hydrolyze glycosides liberating the active forms in plant defense mechanisms [19, 20], and have a role in cellulolysis [21]. Nevertheless, the role of the β-glucosidases in faba bean, as well as their activity toward vicine and convicine, is rather unclear. Arese and De Flora [22] stated in a review that hydrolysis of vicine and convicine can occur in faba bean seeds by endogenous β-glucosidase, even though the efficacy of it was not studied. Rizzello et al. [13] measured the β-glucosidase activity of faba bean doughs using the commonly used artificial p-nitrophenyl-β-d-glucopyranoside (pNPG) substrate, but not using vicine or convicine.
Many strains of lactic acid bacteria (LAB), including Lactobacillus plantarum, Lactobacillus brevis, Pediococcus pentosaceus, and Oenococcus oeni, are capable of producing β-glucosidase in fermenting cereals, legumes, milk products, and fruits [14, 23,24,25,26]. The applicability of substrates is often broad, even though the activities toward different substrate structures may vary [25, 27, 28]. Vicine and convicine were almost completely lost in faba bean suspensions inoculated with Fusarium graminearum (25 °C for 72 h) or L. plantarum (30 °C, 48 h) [11] and in faba bean sourdoughs (30 °C, 48 h) inoculated with L. plantarum B24W [13, 14], most likely because of β-glucosidase producing bacteria. In addition to its positive effects on hydrolysis of vicine and convicine, LAB fermentation can increase the nutritional quality of faba bean and improve sensory perceptions of the bean [14, 29]. Composite wheat bread containing 30% faba bean sourdough showed better nutritional quality of the proteins than breads containing 30% non-fermented faba bean or those made with wheat flour only [30].
However, hydrolysis of vicine and convicine should be carried out in fermentation conditions that are mild enough to attain acceptable technological and sensory properties for baking. In earlier studies, fermentation with L. plantarum B24W at 30 °C for 48 h effectively hydrolyzed vicine and convicine, but the suitability of the obtained sourdough for baking was not tested [13, 14]. In addition, the presence and stability of aglycones in bread are poorly understood. There are indications that the aglycones were present and unstable in faba bean paste and sourdoughs [10, 13], yet the confirmation is still needed.
The main aim of the present study was to investigate hydrolysis of vicine and convicine using the endogenous β-glucosidase of faba beans in suspensions and fermentation with selected LAB possessing β-glucosidase activity in sourdoughs. Furthermore, the stability of the vicine and convicine in an acidic medium was tested. The changes in vicine and convicine levels were investigated under various incubation conditions relevant to food processes and as part of the baking process with the addition of sourdough. Wheat composite breads containing 30% of fermented faba bean were prepared as final products. To evaluate the total elimination of vicine and convicine more specifically, their aglycone forms were also analyzed in the baking process.
Materials and methods
Hydrolysis of vicine and convicine in suspensions
Materials
Selected faba bean cultivars were used to prepare suspensions to study the effect of endogenous β-glucosidase activity (Table 1). The faba beans were grown at Viikki Research Farm at the University of Helsinki, which is in southern Finland. In addition, a commercial faba bean flour from Cereal Veneta (San Martino di Lupari, PD, Italy) was used.
The effect of the incubation conditions (pH, temperature) was tested on two flours (cultivar Kontu and Italian flour) for a 4 and 24 h incubation period (Table 1). After selecting the incubation conditions, faba beans from four cultivars (Alexia, CDC Fatima, Kontu, and CDC SSNS-1) from 2 years (2011, 2015) were studied to compare the activities of endogenous β-glucosidase among different faba bean cultivars (Table 1).
All the faba bean samples from Finland were milled to particle size of 0.5 mm using a Centrifugal Mill ZM200 (Retsch, Haan, Germany) without dehulling first. The Italian beans were dehulled and stone-milled. The flour contained 36 g of protein of dry matter (DM) [30]. The faba beans grown in Finland in 2009–2012 contained an average of 32 g of protein of DM [31]. The moisture content of the flours was measured in duplicate according to the AACC method 44-15A [32], and the moisture contents were used to calculate the results on a DM basis.
Incubation experiments
First, the effects of pH and temperature were studied in suspensions of 0.5 g of flour, with 5 ml of two 0.1 M sodium acetate buffers (pH 4.5 and pH 5), and Milli-Q water (pH 6.5) (Millipore Corporation, Bedford, MA, USA) at 30 °C (Table 1). The suspension in buffer at pH 5 was also tested at 37 °C. This short incubation was conducted for 4 h in a shaking water bath. Next, longer incubations (24 h) were conducted at pH values 4.5 and 5 and at low pH values (1 and 2) at 37 °C (Table 1). For the low pH values, hydrochloric acid was used as diluted solutions (0.1 N, 0.01 N). Finally, to enable comparison of the selected cultivars, incubations were conducted for 4 h at pH 5 and 37 °C (Table 1). All suspensions were prepared and incubated as above.
All incubation experiments were carried out twice with three separate replicates. For each sample set, a non-incubated suspension was used as a control sample. For the long-incubation experiments, suspensions of autoclaved flours were also used as controls.
Faba bean fermentation and bread making
Materials and micro-organisms
The Italian faba bean flour (see “Materials and micro-organisms”) was used to prepare the sourdoughs and in the baking experiments. The LAB strains: Lactobacillus plantarum DPPMA B24W (B24W) (also known as VTT E-133328) from the Department of Soil, Plant and Food Science (University of Bari), L. plantarum VTT E-78076 (E76) from the culture collection of the Technical Research Centre of Finland (VTT), and Pediococcus pentosaceous I02 (I02), previously isolated from faba bean sourdough [33] and belonging to the Department of Food and Nutrition (University of Helsinki), were used in this study. The strain B24W has been used in earlier studies in different conditions to remove vicine and convicine [13, 14]. β-glucosidase activities of the strains were screened prior their use for fermentation. Units (U) for the activity were defined as the release of 1 μmol of p-nitrophenol from the substrate pNPG per ml per min and were measured as described in [23]. The obtained values presented as an average with standard deviation were 0.043 U ± 0.001 U, 0.021 U ± 0.000 U, and 0.028 U ± 0.000 U for the strains B24W, E76, and I02, respectively.
Sourdough fermentation and baking
Before fermentation, the strains were routinely propagated in MRS broth at 30 °C (Oxoid Ltd., Basingstoke, Hampshire, UK) until the late exponential growth phase was reached (ca. 12 h). Cells were harvested using centrifugation (10,000g, 10 min, 4 °C), washed twice with 50 mmol/l of sterile potassium phosphate buffer (pH 7.0), and re-suspended in part of the tap water used to prepare the dough.
The faba bean flour and tap water were mixed in an optimal 40:60 ratio (w/v), as previously described in [30]. The LAB cells were inoculated at an initial cell density of approximately log 6.0 cfu/g of dough. All the doughs were fermented at 20 and 25 °C for 24 h to enable acidification and production of organic acids (lactic acid, acetic acid) by LAB. The sourdoughs were prepared three times at 25 °C, once in a larger batch and twice in a smaller batch, and twice at 20 °C, and were analyzed for LAB growth and chemical parameters at 0 h and 24 h. The presumptive lactic acid bacteria were enumerated using MRS agar medium as reported in [28]. Samples (25 g) of sourdough were taken for pH, TTA (total titratable acidity), lactic acid, vicine and convicine, and the aglycones and were stored frozen at − 20 °C (see “Determination of acidification in sourdoughs and bread” and “Extraction of vicine, convicine and the aglycones”). The samples for vicine, convicine, and the aglycones were freeze dried (Christ Alpha 2-4 LD Plus, Osterode, Germany) prior to use. The residual moisture content of the freeze-dried samples was measured in duplicate according to AACC method 44-15A.
The fermented sourdoughs were used for wheat bread baking, as described in [30]. The mixed doughs were raised for 15 min at 30 °C. Then, they were molded and again raised for 45 min at 30 °C. Baking was carried out at 200 °C for 15 min. In total, four breads were obtained, each containing a 30:70 ratio of faba bean flour to wheat flour and a 60:40 ratio of total flour to water. The control doughs (NF) contained non-fermented faba bean and were prepared under the same conditions as above. Samples (25 g) of fermented doughs and NF control doughs and slices (25 g) of their corresponding breads were used for pH, TTA and vicine, convicine, and aglycone analysis and were stored frozen at − 20 °C (see “Determination of acidification in sourdoughs and bread” and “Extraction of vicine, convicine and the aglycones”). A subset of samples (for vicine, convicine, and the aglycones) were freeze dried prior to use. All baking experiments were conducted once for sourdoughs (from larger batch) from 25 °C and twice from those prepared at 20 °C.
Determination of acidification in sourdoughs and bread
First, the frozen samples of the sourdoughs and breads that were measured for acidity were thawed for 30 min at room temperature. To measure pH and TTA, 10 g of sourdough was suspended in 90 ml of Milli-Q water by mixing for 1 min with a hand blender (Bamix M122, Mettlen, Switzerland). Correspondingly, 10 g of bread was first mixed with 5 ml of acetone and then suspended in 100 ml of Milli-Q water. The measurements were conducted in duplicate using Mettler Toledo EasyPlus Titration with pH-meter (Mettler Toledo, DGi115-SC), which gave the initial pH value, and then the samples were titrated to pH 8.5 using 1 N NaOH.
Lactic acid content was measured in duplicate in sourdough samples that had been fermented at 20 °C using an HPLC–UV method [34]. One gram of sourdough was mixed with 1 ml of 5 mM H2SO4 solution. The mixture was centrifuged for 10 min at 10,000g with Sigma 1‒14 K (Sigma Laborzentrifugen GmbH, Germany) at 4 °C, and the supernatant was taken and diluted to 1:25. The solution was filtrated and injected into the HPLC system. The column was an Agilent Hi-Plex H (7.7 × 300 mm) with a particle size of 8 μm (Agilent Technologies, Santa Clara, CA, USA). The column was used at 50 °C. The eluent was a 5 mM H2SO4 solution that had a flow rate of 0.4 ml/min. Detection was conducted at 210 nm. From the sourdoughs fermented at 25 °C, lactic acid was measured in duplicate using a lactic acid kit (Megazyme, Ireland). First, 1 g of sourdough was mixed with 1 ml of Milli-Q water and centrifuged as described above. Then, 1 ml of supernatant was deproteinized using Carrez reagents and diluted 1:50–1:100 to the final volume prior to use.
Extraction of vicine, convicine and the aglycones
The sample preparation of the suspensions after incubation for vicine and convicine analysis was begun by adding 1 ml (8000 µg/ml) of internal standard uridine (≥ 99%, Sigma-Aldrich, USA) and 15 ml of Milli-Q water and mixing for 1 min with a Vortex. Then, to deproteinize the solutions, 2.5 ml of strong perchloric acid was added. After mixing, the supernatant was separated from the flour particles by centrifuging with Hermle Z323 (Hermle LaborTechnik GmbH, Germany) for 10 min at 10,000g. The supernatant was collected and its volume set to 25 ml. The filtered solutions were injected into the HPLC (see “HPLC-UV analysis of vicine, convicine and the aglycones”). Deproteinization was not conducted for the samples that were incubated at pH 1 and 2. All analyses were carried out in three replicates.
The dried sourdough and bread samples for vicine and convicine analysis were ground (Grindomix GM 200, Retsch GmbH, Haan, Germany) at 8000 rpm for 30 s and extracted similarly as in [35]. Samples of 0.4–0.5 g were extracted three times with 5 ml of perchloric acid (7%) after addition of 1 ml of uridine (4000 µg/ml). The volume was set to 25 ml, and then the samples were filtered and injected into the HPLC (see “HPLC-UV analysis of vicine, convicine and the aglycones”). Three analytical replicates were performed.
The aglycones were extracted from selected samples (fermentations with I02 and B24W) of the freeze-dried sourdoughs, the final doughs that contained 30% faba bean flour, and the corresponding breads that had been stored at − 20 °C. The NF control doughs and breads were also analyzed. The extraction followed the same procedure as in [12]. The freeze-dried samples (0.1‒0.3 g) were weighed in a Falcon tube, and then 10 ml of Milli-Q water was added. After mixing for 2 min, the suspensions were transferred to Amicon Ultra 0.5-ml filters (Millipore, Billerica, MA) and centrifuged with Sigma 1‒14 K for 5 min at 10,000g and 8 °C. Then, the supernatants were transferred into HPLC vials and analyzed directly (see “HPLC-UV analysis of vicine, convicine and the aglycones”). In addition, to evaluate stability of the aglycones, the suspended samples were re-analyzed after they had stood in test tubes at room temperature for 0.5, 2, and 20 h. All analyses were conducted in duplicate.
HPLC–UV analysis of vicine, convicine and the aglycones
The reversed-phase HPLC–UV method used to analyze vicine, convicine, and the aglycones was based on earlier work [12, 35]. An Agilent 1200 instrument (Agilent Technologies, Santa Clara, CA, USA) was equipped with a degasser unit (G1379B), a binary pump (G1312A), a Hip-ALS autosampler (G1367B), a thermostatted column compartment (G1316A), and a diode array detector (G1315B). Separation was conducted using an Atlantis T3 column (4.6 × 100 mm, 3 µm, C18 phase) (Waters, Milford, USA) attached to an Atlantis T3 guard column (4.6 × 20 mm). The compounds were separated using Milli-Q water containing 0.1% of formic acid as an eluent at a flow rate of 0.8 ml/min. The column temperature was 30 °C, and the column was washed with 70% acetonitrile in gradient run. The injection volume was 10–20 µl. The compounds were detected at 273 nm, and the peak areas were recorded as mAu × sec using Agilent 1200 online software. The vicine, convicine, and aglycones were identified according to their retention times and the UV spectra observed earlier in [12, 35].
The vicine standard (≥ 98%, Cfm Oskar Tropitzsch GmbH, Germany) was used for quantification against internal standard uridine. The standard solutions of vicine ranged from 0.15 to 3 µg in injection and had a constant level of uridine (3 µg in injection) in 7% perchloric acid. Convicine was also quantified from the standard curve of vicine. Flour from the Kontu (2011) cultivar was used as an in-house reference sample to monitor the levels. The vicine and convicine concentrations (n = 20) were 5.64 ± 0.07 mg/g (CV % 3.6) and 3.70 ± 0.20 mg/g (CV % 6.5) per fresh weight, respectively.
The losses of vicine and convicine were evaluated by comparing the incubated suspensions to non-incubated ones, and the fermented samples to the starting point of fermentation (0 h). In baking, fermented breads were compared to control NF bread. Loss was calculated as a percentage value.
Data analyses
Statistical differences between the losses of vicine and convicine in incubated suspensions were evaluated using one-way ANOVA and Least Significant Difference (LSD) (SPSS version 22.0, IBM SPSS Statistics, Chicago, IL, USA). Differences were considered statistically significant at p < 0.05.
Results and discussion
Effect of incubation on vicine and convicine contents in suspensions
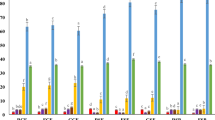
The losses of vicine and convicine were rather low during the short incubations of faba bean flours, indicating low activity of the endogenous β-glucosidase for these substrates (Fig. 1a). Overall, 87–94% of vicine and 89–95% of convicine was left in the suspensions of Kontu flour after incubation for 4 h at the pH values and temperatures tested. In suspensions of the Italian flour, 87–98% and 91–100% of vicine and convicine, respectively, were left. The losses were slightly higher at the lower pH values of 4.5 and 5 than at pH 6.5. Previously, vicine and convicine from faba bean paste were shown to be hydrolyzed by β-glucosidase from almonds more effectively at low pH values (3.5–4.5) than at pH 6 [10]. In that study, decreasing the pH using lemon juice was crucial to activating the β-glucosidase. In the present study, due to minor differences in the losses at 30 °C and 37 °C, all longer incubations and those in the cultivar comparison experiments were carried out at 37 °C.
Losses of vicine and convicine in two faba bean flours (Kontu and Italian) in a short incubation (4 h) at pH range 4.5‒6.5 and 30 °C and at pH 5 and 37 °C; b long incubation (24 h) at pH values 4.5 and 5 and at low pH (1 and 2) and for autoclaved flours at pH 5 and 37 °C; c at pH 5 and 37 °C in cultivar comparison (Alexia, CDC Fatima, Kontu, CDC SSNS-1) from 2 years (2011, 2015). The remaining percentage value (%) is calculated by comparing the vicine and convicine contents to those in the non-incubated controls. The incubation was repeated twice with three individual replicates. The results were calculated per dry weight. Bars marked by different letters (A > C, a > c) are significantly different by the LSD test (P < 0.05, n = 6)
When incubation time was prolonged to 24 h, somewhat higher losses were found (Fig. 1b). After incubation at pH values of 4.5 and 5, 74–81% of vicine and 75–79% of convicine were left in the Kontu and Italian flour suspensions, respectively. The differences in the results caused by the two pH values were not statistically significant (p < 0.05). In the suspensions made from autoclaved flours, no vicine and convicine were lost during the 24 h incubation at pH 5. The heat treatment inactivated the endogenous enzymes and microbes, both of which may have contributed to the losses observed during the long incubation periods. Vicine was also quite stable at pH 1 and 2; 90–92% of vicine remained at both these pH values. In contrast, convicine was less stable in these acidic conditions. In the suspensions of both flours, 82–84% of convicine was left at pH 2 and only 33–34% at pH 1 (Fig. 1b). This result is in line with previous findings, in which convicine was more unstable than vicine was in acidic conditions [36].
The losses of vicine and convicine did not differ significantly in the suspensions of the selected faba bean cultivars. When incubated for 4 h at pH 5 and 37 °C, 89–95% of vicine and 86–96% of convicine remained in the four cultivars studied from the 2 years (Fig. 1c). The losses of vicine and convicine were statistically similar (p < 0.05) for all cultivars and years, apart from two cultivars from 2015: Alexia and Kontu. The Alexia cultivar showed a slightly higher loss of vicine than the other cultivars and, in the Kontu cultivar, slightly more vicine was left compared to the other cultivars. This result showed that there was no notable variation in activities among cultivars that were comparably grown in the same conditions. All the cultivars used in this study contained high amounts of vicine and convicine. However, new germplasms with low vicine and convicine contents, e.g., Melodié, are developed and will become more common. These sources may not need processing to remove vicine and convicine prior consumption [37].
The role of β-glucosidases in faba bean is poorly understood and its activity has been measured in only a few studies. β-glucosidase activity was discussed in a review by Arese and De Flora [22], which reported the highest activity in mature seeds. However, no information is available on the substrate used in the activity test. β-glucosidase activity was also found in faba bean flours using the artificial pNPG-substrate [13], indicating endogenous enzyme activity.
The present incubation experiments showed that some loss occurred during conditions optimal for enzymatic activity, but in practice, the hydrolysis was insufficient to significantly reduce the amounts of vicine and convicine. Under optimal conditions, enzymes are activated in a short time and, therefore, the present study initially used relatively short incubation times for suspensions and prolonged them in later experiments. In addition to hydrolysis by endogenous enzymes, a contribution of spontaneous fermentation was possible, especially during long incubations, as discussed earlier. The conditions were initially kept natural and microbial growth was not inhibited. Some hydrolysis of vicine and convicine has occurred in spontaneous fermentations [13, 14]. However, this hydrolysis was quite small and occurred in long incubations (48 h, 30 °C).
Consequently, an external enzyme source is needed to eliminate vicine and convicine. Therefore, β-glucosidase should be either added as enzyme preparates or produced by microbes, including LAB, during controlled fermentation.
Effect of fermentation and baking on vicine and convicine contents
The present study surveyed the possibility of removing vicine and convicine by fermentation using LAB possessing β-glucosidase. Italian faba bean flour was fermented with three LAB strains at two temperatures to induce more intensive breakdown of vicine and convicine than in the suspensions but under conditions mild enough to achieve acceptable sensory properties.
During fermentation at 20 °C, the cell density increase of the presumptive LAB was of 2.5–3 log cycles and reached values of 8.95–9.00 log cfu/g of dough (Table 2). The pH value for the sourdoughs was 5.3–5.4, while the TTA and lactic acid ranges were 8.5–10.0 ml of NaOH 1 N and 0.41–0.51 g/100 g, respectively. When the strains I02 and B24W were fermented at 25 °C, the growth rate (∆log) was similar to that at 20 °C. Sourdough fermented by the strain E76 showed a slight increase (∆log 3.15) and a final cell density of 9.65 log cfu/g. In both conditions tested, the differences in growth and acidification among the strains were small, although at 25 °C, changes in cell density and pH were higher for the strain E76 than for the strains I02 and B24W. As expected, fermentation at 25 °C led to higher acidity. The pH ranged from 4.4 to 4.8, the TTA from 13.0 to 14.9 ml of NaOH 1 N, and the lactic acid from 0.91 to 1.32 g/100 g.
The fermentation conditions significantly affected the loss of vicine and convicine. At a lower fermentation temperature (20 °C), losses did not occur, apart from a slight reduction in sourdough fermented by the strain I02. At a higher temperature (25 °C), strains I02 and B24W, in particular, were able to hydrolyze both vicine and convicine. In sourdoughs fermented at 25 °C for 24 h by the strains I02, E76, and B24W, the proportions of vicine remaining were in larger batch 56%, 90% and 72% and in smaller batch 15%, 76% and 18%, respectively. Those of convicine were 78%, 87% and 80% in larger batch, and in smaller batch 53%, 86% and 66%, respectively (Fig. 2a). Vicine levels were lowered in general more than convicine levels. A variation in hydrolysis efficiency was noted, even though no biological differences were observed among the replicates. Consequently, the variation may have resulted from the sampling, for example, the handling of the large sample volumes.
Losses of vicine and convicine in a sourdoughs and b breads. Fermentation was conducted at 20 °C and 25 °C for 24 h with three strains (I02, E76, and B24W). The breads contained 30% faba bean flour from the total weight of flours. The results were calculated per dry weight. The remaining percentage value (%) is calculated by comparing faba bean sourdoughs and sourdough breads with doughs in the starting point and breads made with non-fermented faba flour. There were two replicates of the experiments conducted at 20 °C, for both fermentation and baking. At 25 °C, only replicate * was used in baking, but fermentation was conducted three times. There were three analytical replicates
The β-glucosidase activity of the selected starters toward pNPG was in line with the hydrolysis levels of vicine and convicine (Fig. 2a). Among the starters, L. plantarum B24W showed the highest β-glucosidase activity, followed by P. pentosaceus I02. Therefore, the lowest losses of vicine and convicine, found in sourdough inoculated with L. plantarum E76, were probably due to its lowest β-glucosidase activity. Nevertheless, β-glucosidase activity was likely the reason for the losses of vicine and convicine, because vicine was stable, even at pH 1, and the acidity itself did not cause losses during the incubation of the autoclaved flours.
Lactobacillus plantarum B24W, originally isolated from cheese, has shown efficiency in hydrolyzing β-glycosides, i.e., isoflavones in soy milk and vicine and convicine in longer fermentation of faba bean [14, 23]. In previous studies, this strain has adapted relatively slowly in faba bean matrix, but nevertheless has shown good growth performance [14]. P. pentosaceus I02 was recently isolated from faba bean flour sourdough [33], while L. plantarum E76 was isolated from Finnish rye sourdoughs and used to ferment wheat flour [38, 39]. Despite the different origins of the strains, all grew efficiently in faba bean.
All three strains used in the present study showed activity with the synthetic pNPG substrate and further activity in hydrolyzing vicine and convicine in real fermentation conditions. However, hydrolysis required a higher fermentation temperature (25 °C), because at a low fermentation temperature (20 °C) vicine and convicine were not reduced. The present study conducted fermentation for 24 h, which was a shorter time than that used in earlier studies [13, 14]. The results showed that when acidification is controlled, marked removal of vicine and convicine may be challenging. In previous experiments, the losses of vicine and convicine were more pronounced when more intense fermentation was used, with more than 90% of vicine and convicine disappearing in 48 h at 30 °C when fermented with the strain B24W [14]. Glucosidase activities in LAB are tightly linked to the carbohydrate metabolism, and their mechanisms depend on multiple factors [28], including the specific adaptation to the environment (growth, acidification, or activation of specific metabolic pathways of a defined strain). More information is needed on the metabolic performance of β-glucosidase active strains to direct their use for the most efficient transformation of faba bean.
In the present study, the breads contained 30% of either fermented or NF faba bean. In the NF bread, the pH was 5.9 and the TTA was 4.3 ml of NaOH 1 N (Table 2). When sourdough that had been fermented at 20 °C was added, the pH values were 5.0–5.3 and the TTA values were 6.7–7.8 ml of NaOH 1 N. The higher acidity of the sourdough fermented at 25 °C reflected on the corresponding breads, showing pH 4.7–4.9 and TTA of 7.4–8.6 ml of NaOH 1 N, respectively.
When the vicine and convicine levels in sourdough breads were compared with those in NF bread (Fig. 2b), the differences were comparable to the losses in the fermentation step (Fig. 2a). Thus, the stability of the vicine and convicine in the later steps of the baking process did not differ for sourdough bread and NF bread. To further evaluate the losses in the baking process, the amounts of vicine and convicine in faba bean doughs and breads were compared, taking into account the proportion of faba bean sourdough in the breads. This comparison showed that the losses during the entire baking process were 11–25% and 11–42% for vicine and convicine, respectively.
The hydrolysis of vicine and convicine could have occurred during the bread making process due to the β-glucosidase produced by either the LAB or the baker’s yeast, which is also known to have β-glucosidase activity [40, 41]. Increased activity of β-glucosidase due to baker’s yeast has been noted in soy-wheat dough fermented for 2 h at 22–48 °C [40]. Nevertheless, in the present study, the time for hydrolysis (dough raising 15 and 45 min at 30 °C) was relatively short and the estimated losses during baking at 200 °C for 15 min were small, especially for vicine. The baking process was not expected to cause notable losses of vicine and convicine, because they are relatively heat stable [8, 10].
Aglycones formed from hydrolysis
When using a fermentation temperature of 25 °C, hydrolysis products of vicine and convicine, divicine and isouramil were detected in freeze-dried sourdoughs that had been inoculated with strains of I02 (Fig. 3) and B24W (data for I02 in Fig. 3). The retention times and UV spectra of the aglycones were consistent with those in earlier studies [12, 15]. In contrast, these aglycones were not detected in doughs fermented at a lower temperature, 20 °C (data not shown), or in either the NF control doughs or breads.
Chromatograms from suspensions of a freeze-dried sourdough (100% faba bean, inoculated with strain I02) fermented for 24 h at 25 °C; b dough (30% faba bean); and c bread (30% faba bean) showing appearance and disappearance of the aglycone forms of vicine and convicine. The injection volume was 20 µl. The measurements were made in duplicate. Fermented suspensions are marked with black lines and controls are marked with grey dotted lines
Figure 3a shows how the amounts of vicine and convicine decreased and the aglycones appeared during the sourdough process. In dough, added wheat flour diluted the contents (Fig. 3b). The content of divicine was 18% in dough from the amount that was in sourdough, and the corresponding proportion of isouramil was 12%. Finally, the aglycones were not detected at all in the breads (Fig. 3c) in contrast to the corresponding doughs (Fig. 3b). These experiments provided preliminary data about the aglycones in bread processing. The results indicated that when heat-treatment in bread baking inactivated the enzymes and stopped the hydrolysis of vicine and convicine, the aglycones that had formed earlier were degraded and gradually disappeared.
To study stability of the aglycones in suspended freeze-dried sourdoughs, they were kept at room temperature for 0.5, 2, and 20 h (Fig. 4). At 0.5 h, a marked decrease in divicine and isouramil levels occurred. This decrease continued at 2 h, and at 20 h, the aglycones were undetectable, thus indicating that they were not stable in the suspensions. In previous studies, the aglycones disappeared in suspensions treated with added β-glucosidase under pH and temperature conditions relevant to the food system [12], and the same happened in sourdoughs and doughs, indicating that stabilizing factors do not occur in faba bean matrix.
In addition to the disappearance of the aglycones, there were indications of the formation of oxidized divicine before the total loss of the UV-absorbing compounds. Our previous study found that a peak, suggested to be oxidized divicine, was formed during the hydrolysis of vicine [12]. In the present study, the peak was also formed, and its amount increased 1.3-fold at 0.5 h in suspension, while divicine decreased (Fig. 4). The aglycone degradation products were unstable, as shown by the loss of UV absorbance in a few hours, which has also been shown in previous studies [12, 15].
These results were also in line with those of previous studies on faba bean paste [10] and sourdoughs [13], in which the aglycones seemed to degrade and disappear. To our knowledge, the present study is the first in which the aglycones of vicine and convicine were studied in real food matrices.
Conclusions
This study investigated the possibility of removing vicine and convicine from faba bean using endogenous or microbiologically produced β-glucosidase activity. The need for external sources of the enzyme was confirmed, because the activity of endogenous β-glucosidase was too low to have any practical effect on removing vicine and convicine. Likewise, acidity itself did not cause vicine losses. Instead, LAB fermentation with β-glucosidase positive strains induced the hydrolysis of vicine and convicine. However, removal efficiency was dependent on the strain and the fermentation temperature. Mild fermentation conditions did not lead to significant reduction in the amounts of vicine and convicine. Further screening of β-glucosidase active LAB strains and deeper understanding of their performance during faba bean fermentation are necessary to enable elimination of vicine and convicine while also maintaining acceptable sensory properties. Vicine and convicine were found in breads as well, thus indicating that baking under slightly acidic conditions did not degrade them. The aglycones of vicine and convicine were detectable before baking but not in the final bread product. Fermentation of faba bean or addition of food-grade β-glucosidase enzyme can be considered as a tool for removal of vicine and convicine, although optimization of processing conditions is required.
References
Watson CA, Reckling M, Preissel S, Bachinger J, Bergkvist G, Kuhlman T, Lindström K, Nemecek T, Topp CFE, Vanhatalo A, Xander P, Murphy-Bokern D, Stoddard F (2017) Adv Agron 144:235–303
Day L (2013) Trends Food Sci Technol 32:25–42
Jacob C, Carrasco B, Schwember AR (2016) Plant Cell Tissue Organ Culture 127:561–584
Ray H, Georges F (2010) GM crops 1(2):99–106
Arese P, Gallo V, Pantaleo A, Turrini F (2012) Transfus Med Hemother 39:328–334
Cappelini MD, Fiorelli G (2008) Lancet 371:64–74
Baker MA, Bosia A, Pescarmona G, Turrini F, Arese P (1984) Toxicol Pathol 12:331–336
Cardaror-Martinez A, Maya-Ocana K, Ortiz-Moreno A, Herrera-Cabrera BE, Davila-Ortiz G, Muzquiz M (2012) J Food Qual 35:419–428
Patterson CA, Curran J, Der T (2017) Cereal Chem 94:2–10
Arbid MSS, Marquardt RR (1985) J Sci Food Agric 36:839–846
McKay AM (1992) J Appl Bacteriol 72:475–478
Pulkkinen M, Zhou X, Lampi A, Piironen V (2016) Food Chem 212:10–19
Rizzello CG, Losito I, Facchini L, Katina K, Palmisano F, Gobbetti M, Coda R (2016) Sci Rep 6:32452
Coda R, Melama L, Rizzello CG, Curiel JA, Sibakov J, Holopainen U, Pulkkinen M, Sozer N (2015) Int J Food Microbiol 193:34–42
Chevion M, Navok T, Glaser G, Mager J (1982) Eur J Biochem 127:405–409
Marquardt RR, Frohlich AA (1981) J Chromatogr A 208:373–379
Marquardt RR, Frohlich AA, Arbid MSS (1989) J Agric Food Chem 37:455–462
Marquardt RR, Arbid MSS, Frohlich AA (1989) J Agric Food Chem 37:448–454
Pavlik M, Vanova M, Laudova V, Harmatha J (2002) Rostl Vyroba 48:543–547
Desroches P, Mandon N, Baehr JC, Huignard J (1997) J Insect Physiol 43:439–446
Ketudat Cairns JR, Esen A (2010) Cell Mol Life Sci 67:3389–3405
Arese P, De Flora A (1990) Semin Hematol 27:1–40
Di Cagno R, Mazzacane F, Rizello CG, Vincentini O, Silano M, Giuliano G, De Angelis M, Gobbetti M (2010) J Agric Food Chem 58:10338–10346
Grimaldi A, Bartowsky E, Jiranek V (2005) J Appl Microbiol 99:1061–1069
Michlmayr H, Schuemann C, da Silva NMBB, Kulbe KD, del Hierro AM (2010) J Appl Microbiol 108:550–559
Verni M, Wang C, Montemurro M, De Angelis M, Katina K, Rizzello CG, Coda R (2017) Front Microbiol 8:2461
Sestelo ABF, Poza M, Villa TG (2004) World J Microbiol Biotechnol 20:633–637
Michlmayr H, Kneifel W (2014) FEMS Microbiol Lett 352:1–10
Rizzello CG, Calasso M, Campanella D, De Angelis M, Gobbetti M (2014) Int J Food Microbiol 180:78–87
Coda R, Varis J, Verni M, Rizzello CG, Katina K (2017) LWT Food Sci Technol 82:296–302
Lizarazo CI, Lampi A, Liu J, Sontag-Strohm T, Piironen V, Stoddard FL (2015) J Sci Food Agric 95:2053–2064
American Association of Cereal Chemists, AACC (1994) AACC method 44-15A. Moisture—Air-oven methods. Approved Methods of the American Assn. of Cereal Chemist. AACC, St Paul, Minnesota
Coda R, Kianjam M, Pontonio E, Verni M, Di Cagno R, Katina K, Rizzello CG, Gobbetti M (2017) Int J Food Microbiol 248:10–21
Chamlagain B, Deptula P, Edelmann M, Kariluoto S, Grattepanche F, Lacroix C, Varmanen P, Piironen V (2016) LWT Food Sci Technol 72:117–124
Pulkkinen M, Gautam M, Lampi A, Ollilainen V, Stoddard F, Sontag-Strohm T, Salovaara H, Piironen V (2015) Food Res Int 76:168–177
Marquardt RR, Muduuli DS, Frohlich AA (1983) J Agric Food Chem 31:839–844
Gallo V, Skorokhod OA, Simula LF, Marrocco T, Tambini E, Schwarzer E, Marget P, Duc G, Arese P (2018) Blood 131:1621–1625
Katina K, Poutanen K, Autio K (2004) Cereal Chem 81:598–610
Katina K, Heiniö R, Autio K, Poutanen K (2006) LWT Food Sci Technol 39:1189–1202
Riedl KM, Zhang YC, Schwartz SJ, Vodovotz Y (2005) J Agric Food Chem 53:8253–8258
Inamdar AN, Kaplan JG (1966) Can J Biochem 44:1099–1108
Acknowledgements
Open access funding provided by University of Helsinki including Helsinki University Central Hospital. This work was carried out under the European Project “BIOPROT” (FP7-ERA-Net-SUSFOOD), with a grant from The Doctoral Program in Food Chain and Health of the University of Helsinki. The authors would like to thank Miikka Olin for valuable technical help and Docent Frederick Stoddard and Dr. Clara Lizarazo for providing the faba beans. We also gratefully acknowledge funding from the Finnish Cultural Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Compliance with ethics requirements
This article does not contain any studies with human or animal subjects.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Pulkkinen, M., Coda, R., Lampi, AM. et al. Possibilities of reducing amounts of vicine and convicine in faba bean suspensions and sourdoughs. Eur Food Res Technol 245, 1507–1518 (2019). https://doi.org/10.1007/s00217-019-03282-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00217-019-03282-4