Abstract
The aim of this study was to determine the chemical composition in different cultivars of Prunus persica L. fruits with special focus on polyphenols, carotenoids, sugars and organic acids content. In addition, the PCA model was applied to all data to determine the most important variables that explain the relationships between twenty selected cultivars of peaches and to identify the most attractive cultivars. The conducted study showed that the most interesting cultivars from the point of view of direct consumption are: ‘Early redhaven’, ‘Candor’, ‘Harrow beauty’ due to the large size of fruit, rich juiciness, high maturity index, as well as above-average content of polyphenols and carotenoids. In turn, fruits with medium-sized stones and fruits, a high content of dry matter and total sugars, and with a high content of carotenoids (‘Harrow beauty’, ‘Kijowska wczesna’, ‘Jersey land’), are ideal for the manufacture of healthy dried snacks. Additionally, juicy peaches with a high content of organic acids and bioactive compounds, i.e., ‘WB 258’, ‘Spring time’ and ‘Beta’, are suitable for the production of purees, smoothies, and juices. Finally, it has been shown that peach fruit is an interesting raw material with a varied chemical composition and nutritional value, strongly determined by the cultivar.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Recently, the interest in the composition of fruits has grown because of increased awareness of their possible health benefits. This results from recent studies which demonstrate, beyond any doubt, that fruits have a significant impact on reduced morbidity and mortality from chronic non-communicable diseases’ society in the 21st century.
The benefits of eating fruit are mainly connected with the richness of their chemical composition. Basic components of fruits include protein, carbohydrate (especially fructose, sorbitol and glucose), minerals (Mg, Fe, P, Cu, Ca, Na, K), vitamins (C, PP, B group, provitamin A), organic acids, pectins and a lot of bioactive secondary metabolites of plants (for example, isoprenoids and phenolic compounds) [1]. The largest range of pro-health properties is attributed to secondary metabolites (polyphenols, isoprenoids). Phenolic compounds constitute a very numerous group of natural organic substances that occur in various morphological parts of plants. They exhibit especially strong antioxidative properties that protect defense systems of the body against destructive effects of free radicals [2,3,4]. Another group of secondary metabolites of plants that exhibit health-promoting properties are isoprenoids that include triterpenes, iridoids, carotenoids and chlorophylls. They are, likewise polyphenols, classified as both preventive and intervention antioxidants, and are characterized by valuable biological properties the best documented of which is their provitamin activity [5].
The other pro-healthy benefits are also ascribed to other compounds occurring in fruits, e.g., organic acids, pectins, vitamins and minerals. The organic acids stimulate the secretion of digestive enzymes and regulate the proper chemical reactions of the body [6]. Pectin inhibits the absorption of dietary fats and their collection in the tissues of the liver. In addition, they influence the lower blood glucose levels and improves peristalsis [7]. While vitamins and minerals are responsible for the proper conduct of biochemical reactions and functioning of the human body [8].
Considering the above, it seems advisable to undertake any actions ascribing to the global trend of analyzing and promoting raw materials with a high nutritive value and health-promoting properties. Hence, the aim of this study was to determine the chemical composition of fruits of different cultivars of Prunus persica L. Batsch with special focus on polyphenols, carotenoids, sugars and organic acids content. Sugars and organic acid, as the main soluble constituents of peach fruit, have a major effect on taste and represent an index of consumer acceptability. In turn, modern consumers are increasingly interested in their personal health and expect the foods to be not only tasty and attractive but also safe and healthy, therefore polyphenols and carotenoids content were also analyzed in this study. In addition, the PCA model was applied to all data to determine the most important variables that explain the relationships between the twenty selected cultivars of peaches and to identify the most attractive cultivars.
Materials and methods
Plant material
Twenty cultivars of peach fruit were used in this study:
-
Early maturing: ‘Harbinger’, ‘Kijowska wczesna’, ‘Spring time’, ‘Beta’, ‘Maycresh’, ‘Harrow diamond’, ‘Dixired’, ‘Candor’, ‘Harnaś’, ‘Sweet haven’, ‘WB 258’,
-
Mid-early maturing: ‘Early Redhaven’, ‘SB6A–35’, ‘Jerseyland’, ‘BL6’, ‘Red Cup’, ‘Royalvee’,
-
Late maturing: ‘Flamin Fury’, ‘Harrow Beauty’, ‘Madison’ (Table 1).
All of them were appropriate for food manufacturing and were grown in Poland. The fruits were harvested at the Research Station for Cultivar Testing in Zybiszów near Wrocław (51°3′51.11″N, 16°54′43.56″E) and were collected at “ready-to-eat” ripening stage. Immediately after harvest, in fresh raw materials, the content of Vitamin C, soluble solids, pectin, ash, pH, titratable acidity, and fruit weight were measured. In turn, for the analysis of polyphenolic compounds, organic acids, sugars and carotenoids, the whole fruits were freezing with liquid nitrogen and crushing them to homogeneous powder by laboratory mill and after that freeze-drying them.
Physicochemical analysis
The soluble solids’ content was determined by a refractometer and expressed as °Brix, while the pectins’ content was analyzed according to the Morris method described by Pijanowski, Mrożewski, Horubała and Jarczyk [9] and expressed as g/100 g fruit. Total content of l-ascorbic acid, ash and dry matter as g/100 g was determined by the PN norms—PN-90/A-75101/11, PN-90/A-75101/08, PN-90/A-75101/03, respectively.
Determination of sugar content by HPLC coupled to light scattering detector
A solvent for the analysis of sugar content and determination of sugar was prepared as previously described by Nowicka, Wojdyło and Teleszko [10]. All determinations were done in triplicate and results were expressed as g/100 g dm of peach.
Determination of acids’ content by UPLC-PDA method
Obtained freeze-dried peaches (1 g) were mixed with 50 ml of redistilled water, and after that ultrasonificated for 15 min, boiled for 30 min and centrifuged for 10 min. The extracts were applied into the Sep-Pak C-18 and eluted by water to give a sample solution for the estimation of acid content. The analysis of acid content was carried out on UPLC Acquity system consisting of a sample manager, binary solvent manager, PDA detector. Empower 3 software was used for data collection and integration of chromatograms. A 10 µL sample was injected on the Supelcogel TM C-610H column (30 cm × 7.8 mm; Supelco, Bellefonte, PA, USA). The elution was carried out at 30 °C under a isocratic flow using 1 mM phosphoric acid solution at the flow rate of 0.5 mL/min. Acid components were identified by comparison with the standards. The calibration curves were prepared by plotting different concentrations ranging from 0.5 to 10 mg/mL (R2 ≤ 0.9998) of standards versus the area measurements in UPLC. Results were expressed as g/100 g dm of peach.
Analysis of polyphenol compounds
The quantitative analysis of total polyphenols by UPLC was performed as described by Wojdyło, Nowicka, Laskowski and Oszmiański [11]. The results were expressed as mg/100 g dm of peach fruits.
Analysis of carotenoids
Determination of carotenoids by UPLC was prepared as previously described by Wojdyło, Nowicka and Bąbelewski [12]. The results were expressed as mg/100 g dm of peach fruits.
Statistical analysis
Results obtained in this study were analyzed and interpreted using statistical methods, including principal component analysis (PCA) to determine correlations, with the use of Statistica ver. 12.50 software.
Result and discussion
Nutritional and chemical components in different peach cultivars
In the present study, different physicochemical parameters were evaluated in 20 cultivars of peach fruit including fruit weight and contents of soluble solids, dry matter, pectins, ash, and Vitamin C (Table 1).
The average fruit weight was 103.7 g, wherein 9 cultivars were heavier and 11 were lighter. Among the analyzed peach cultivars, the largest fruits were identified in ‘Red cup’ and ‘Jerseyland’ cv., weighing 160.2 g and 140.3 g, respectively. In contrast, the smallest fruits were observed in the early cultivars: ‘Spring time’—60.1 g and ‘Harbringer’—64.0 g. Generally, the later cultivars of peach had heavier fruits than those harvested earlier and vice versa. Other authors showed that, except for the harvest time, fruit weight might also depend on the cultivar of fruit, fruit load, and on climatic and agricultural conditions [11, 13]. Although the early cultivars ‘Harbinger’ and ‘Springtime’ were also characterized by the lowest mass of the stones, the study showed no clear relationship between harvest time and stone mass. Therefore, it can be assumed that the size of stone is a cultivar-specific trait.
Differences between peaches cultivars were also reflected in the chemical composition of fruits. Dry matter content of peach fruit ranged from 10.3% (‘SB6A-35’) to 15.7% (‘Harrow Beauty’). Compared to the other fruits, the content of total solids was similar to that in apple (13–20%), but definitely lower than in berry fruit, like chokeberry (39.3–53.4%), blackcurrant (20.4–23.5%) or highbush cranberry (15.9–22.3%) [14, 15]. According to Zatylny et al. [15], the content of dry matter depends on the cultivar, but other authors showed that the total solid content might be influenced by many factors like harvest time, degree of fruit dehydration, an increase in the insoluble solids’ content of the fruit during maturation or climatic and agricultural conditions [13, 14].
The soluble solid content was also analyzed in this study. It is a characteristic which largely determines the final content of dry matter. Our study showed a relationship between the content of soluble solids in the analyzed peach fruits and their solids’ content. In the examined peaches, the soluble solid content ranged from 9.0 °Brix in ‘SB6A-35’ to 14.4 °Brix in ‘Harrow Beauty’, with the mean value accounting for 11.3 °Brix. This is consistent with findings reported by Zhang, Peng, Zhang, Song and Ma [16] and by Cirilli, Bassi and Ciacciulli [17] who showed the average soluble solids’ content in peaches to reach 12 °Brix. The soluble solid content determined in our study in peach fruit is similar to that determined in apricots (12.9 °Brix), nectarines (14.2 °Brix) or apples (10.7–12.5 °Brix), but definitely lower compared to chokeberry (18.3 °Brix) and blackcurrant (16.8 °Brix). The soluble solids include: oligosaccharides, polysaccharides, organic acids, dyes and tannins, and other soluble compounds. Therefore, their content is usually higher in strongly colored fruits containing more sugars and acids.
In this study, we analyzed peach fruits also for the content of ash, which depends not only on the species or cultivar, but also on the growing conditions [18]. Among the analyzed fruits, the highest content of mineral compounds was found in fruits of ‘Harrow beauty’ (0.55%), ‘Madison’ (0.54%), ‘Candor’ (0.52%) and ‘Jerseyland’ (0.51%) cultivars. In turn, ‘Harbringer’, ‘Kijowska wczesna’, ‘Spring time’, and ‘Harnaś’ cultivars had almost two times lower ash content accounting for 0.28% on average. Ash content is basically determined by minerals: magnesium, iron, phosphorus, copper, calcium, potassium and sodium, which in fruits occur in the form easily absorbable for humans [1].
Benefits that stem from fruit consumption are mainly associated with the richness of their chemical composition, including the contents of pectins and vitamin C. Pectins inhibit the absorption of dietary fats and their deposition in liver tissues. In addition, they contribute to blood glucose level reduction and improve peristalsis [7]. In turn, vitamin C is responsible for the proper course of biochemical reactions and body functions and is classified as both preventive and intervention antioxidants. In addition, it is characterized by valuable biological properties, the best documented of which is its provitamin activity [8]. In the peach fruits analyzed in our study, the content of pectins ranged from 0.9% (‘Early redhaven’) to 1.6% (‘Harrow beauty’). Peach fruits are considered to be very good sources of pectin, whose content in these fruits is comparable to that in apple (0.9%), Japanese quince (1.0%), and blackcurrant (1.7%) [15]. In the case of Vitamin C content, it differed greatly in individual peach cultivars, ranging from 4.13 to 16.28 mg/100 g of raw material. The highest ascorbic acid content (above 12 mg/100 g) was detected in ‘Harrow beauty’, ‘WB 258’ and ‘Jereseyland’ cultivars, while the lowest one (under 5 mg/100 g) in ‘Candor’, ‘Harnaś’, ‘Harrow diamond’ and ‘Sweet haven’ cultivars. This is in agreement with findings reported by Gil, Tomas-Barberan, Hess-Pierce and Kader [19], who determined from 3.6 to 12.6 mg/100 g of Vitamin C in peaches. In addition, they pointed out that Vitamin C was a cultivar-specific traits, which can also be observed in our research.
Sugar and acid content in different cultivars of peach
The analyzed peach fruits were also determined for sugar and acid contents. Apart from determining the total content of these compounds, in this study we analyzed the exact profile of sugars and acids in different cultivars of peach, and the results of these analyses were presented in Table 2. Both the total contents and individual profiles of these compounds appear to be crucial in shaping the taste and degree of sweetness of raw materials. Therefore, their detailed analysis allows, at the first stage of the study, to identify the best cultivars in terms of sensory properties.
Among the seven organic acids identified in peach fruits, the major ones were: malic acid (31–52%) > quinic acid (12–25%) > citric acid (2–25%) ≥ fumaric acid (9–12%) > oxalic acid (< 1%) ≥ shikimic acid (< 1%) and isocitric acid, but it was present in trace amounts and only in three cultivars. The predominant organic acid in peach fruits was malic acid, which is also confirmed by other authors [20]. Generally, the analyzed cultivars can be divided into two main groups in terms of malic acid content. The first of these is peach fruits that contain more than 4 g of malic acid /100 g dm (these were early and very late cultivars) and the other ones were these with malic acid content lower than 3.5 g per 100 g of dm (‘Dixired’; ‘Candor’, ‘Harnaś’, ‘Sweet haven’, ‘WB 258’, ‘Early redhaven’,‘Royalvee’, ‘Harrow Beauty’). Such great differences in malic acid content were not shown by other authors who demonstrated its content to remain stable both during growth and maturation of the peach fruit [20]. The conducted study showed also a high content of citric acid which ranged from 2.87 g/100 g dm in ‘Beta’ cv. to 0.19 g/100 g dm of ‘Harbringer’. Such a great difference in its content may be due to the degree of fruit maturity. It has been shown that fully mature peach fruits have a lower citric content [21]. Although, the malic acid followed by citric acid were the major organic acids of peach fruit—representing more than 65% of the total acid content determined; quinic, shikimic, fumaric, oxalic and isocitric acids were also identified in the analyzed fruits. Especially noteworthy are the last two acids (isocitric and oxalic acids), which have been never before identified in peaches.
Generally, the total content of acids demonstrated in this study (5.43 g–13.92 g/100 g dm of peach) fits within the range of values previously described in literature, but it obviously depends on the origin, cultivar, harvest date and degree of fruit maturation [20, 21].
Sugars represent the main component of fruit edible quality by imparting sweetness being one of the attributes influencing the degree of consumer satisfaction regarding peaches. The intensity of sweetness depends on the total sugar content as well as on the sugar profile. It is due to the fact that the sweetening power of fructose, glucose and sorbitol differs from that of sucrose (1.7-; 0.8- and 0.6-fold, respectively) and therefore it is important to determine the relative content of each individual sugar [17]. Our study showed sucrose to be the predominant sugar in different cultivars of peach fruit, accounting for approximately 58–74% of the total sugars content. The other major sugars were: fructose (7–14%) > glucose (5–12%) > sorbitol (3–10%). High contents of sugars in peach were also confirmed by other authors [17, 22], who demonstrated that the sucrose content should be from 40 to 80%, that of glucose and fructose (in variable ratios) together from 10 to 25%, and that of sorbitol around 10%. In addition, it is noteworthy to us that in each analyzed cultivar the content of fructose was higher than that of glucose. According to Robertson and Meredith [23], high-quality peaches have lower contents of glucose and sorbitol and a higher content of fructose compared to the low-quality peaches. Generally, the total content of sugars determined in this study ranged from 49.54 g/100 g dm in the case of ‘Maycresh’ cv. to 73.66 g/100 g of ‘Madison’ cv. Similar values were previously reported for peaches by other authors [17, 22]. In addition, the authors agree that the total and individual sugar contents are strongly affected by seasonal variability, climate, irrigation or crop load, in contrast to the sugar profile which is relatively stable across environments and genotypes [17].
Also, maturity index (MI) of fruits was analyzed in this study (Table 1). It determines the relationship between contents of total soluble solids content and acids and is used to classify fruits as sour (MI: 5–7), sour–sweet (MI: 17–24), and sweet (MI: 31–98) [24]. In addition, the MI appears to be a key factor responsible for the flavor and taste of fruit [11]. In the analyzed cultivars of peach, MI ranged from 13.24 (‘SB6A–35’) to 26.59 (‘Royalvee’). It may thus be concluded that most of the studied fruits were semi-sweet, but the later varieties were a little bit sweeter than those harvested earlier.
Quantification of bioactive compounds in different cultivars of peaches
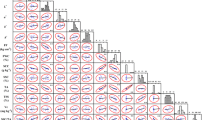
Figure 1 presents results of determinations of polyphenols and carotenoids content in peach fruit. The average content of polyphenols and carotenoids was 1732 mg and 241 mg per 100 g dm of peaches, respectively. The total content of polyphenols differed significantly and ranged from 722 mg/100 g dm in ‘Madison’ to 3116 mg/100 g dm in ‘WB 258’. In the case of carotenoids, the highest content was determined in ‘Harbringer’—390 mg/100 g dm of peach fruit, while the lowest in ‘Spring time’—40 mg/100 g dm. Positive correlations were found between the results of both bioactive compounds—PC = 0.260.
Many factors influence the content of antioxidative compounds. The most important is the cultivar, the morphological part and the technological processes used during the processing of the raw material. The appropriate agrotechnical practices and cultivation under appropriate climatic conditions are also important [25]. In addition, the phytochemical composition is largely conditioned by the process of fruit ripening—a series of physiological, biochemical and structural changes leading to obtain the full maturity fruits. Belhadj et al. [26] confirmed that the content of bioactive compounds depends on the degree of fruit maturity. Fruits during the last stage of maturity (red peaches) were characterized by three to ten times higher concentration of the tested compounds than the unripe fruits (green fruits).
The presented study showed that the total carotenoids and polyphenols content in peach fruits is significantly dependent (p ≤ 0.05) on the cultivar. Differences between concentration of carotenoids in different cultivars were also demonstrated by Belhadj et al. [26]. The research of these authors comprised four cultivars (‘Chatos’, ‘Elegant Lady’, ‘Gladys’, ‘Royal Glory’), which in full maturity were characterized by the following content of carotenoids—523.92; 504.95; 263.20; 244.22 µg βCE/g, respectively. In turn, according to Bento et al. [27], the total polyphenol content ranged from 22.4 mg/100 g dm to 134.2 mg/100 g dm. However, according to Nowicka et al. [28], peach puree contained 429 mg/100 g of product. The main reason for significant differences may be the cultivar, cultivation method, climatic conditions and the degree of fruit maturity—it was shown that green peaches are a much better source of polyphenols than partially mature ones [26]. The reduction of the total content of the tested fruit compounds during maturation is associated with an increase of polyphenol oxidase activity [26; 29].
Principal component analysis of different cultivars of peach fruits and their compounds
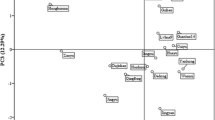
The PCA model was applied to all data to determine the most important variables that explain the relationships between the twenty selected cultivars of peaches and to identify any group patterns (Fig. 2). In addition, PCA was carried out separately for early, mid-early and late maturity cultivars of peach fruits.
PCA map showing the relationship among the physicochemical properties and analyzed peaches fruit. WFW whole fruit weight, PFW pulp fruit weight, SFW stone fruit weight, dm dry matter, SS soluble solids, MI maturity index, P pectins, Vit C Vitamin C, acids total content of organic acids, sugars total content of sugar
Two principal components explaining 52% of the overall variance (31% and 21% for PC1 and PC2, respectively) divided the analyzed cultivars into four distinct clusters. The first principal component (PC1), which explains 31% of the overall variance, is clearly identified with the MI, polyphenols, acid and ash content, while the second principal component (PC2) is related to the carotenoids, Vitamin C, pectins, sugar, dry matter content and physical properties. The factors that most contributed to PC1 (positive side) were: MI, ash and polyphenols’ content, and the organic acids to the negative side. On the other hand, the main contributors to PC2 (negative side) were sugars, vitamin C, pectins and dry matter, whilst carotenoids and fruit size contributed to the positive side.
Thus, it was shown that the common feature for the ‘Jerseyland’, ‘Candor’, ‘Harnaś’, ‘WB 258’, ‘SB6A–35’, ‘BL6’ cvs. was the high content of polyphenolic compounds and also maturity index and ash content. In addition, the PCA model showed that the early-maturing varieties as ‘Kijowska wczesna’, ‘Harbringer’, ‘Spring time’, ‘Dixired’, ‘Maycresh’, ‘Harrow diamond’, and ‘Early redhaven’ were characterized by a high content of vitamin C and organic acids. In turn, the sweetest varieties, with the highest mass of fruit and a high content of pectins were the late-maturing cultivars: ‘Madison’, ‘Harrow beauty’, and ‘Flamin fury’.
The PCA analysis carried out for the purposes of this study thus confirmed significant differences in the chemical composition of peach fruit depending on the cultivar. At the same time, it indicated some common features of selected cultivars, owing to which it is possible to divide the analyzed peaches into more sweet ones, more sour ones or those with a higher content of polyphenolic compounds.
Conclusion
The conducted study allowed for a very accurate analysis of the physicochemical properties, including the content of phytochemicals, in different cultivars of peach fruit growing in Poland. The analysis of the obtained results enabled indicating differences between particular cultivars, as well as identifying the most valuable peaches for both direct consumption and processing. And so, the following cultivars seem to be the most interesting from the point of view of direct consumption: ‘Early redhaven’, ‘Candor’, ‘Harrow beauty’ due to the large size of fruit, rich juiciness, high MI index, as well as above-average content of polyphenols and carotenoids as well as ‘WB 258’ with a slightly lower MI index but a very high content of phytochemicals—polyphenols, carotenoids and vitamin C. In turn, fruits with medium-sized stones and fruits, a high content of dry matter and total sugars, and with a high content of carotenoids—‘Harrow beauty’, ‘Kijowska wczesna’, ‘Jersey land’, are ideal for the manufacture of healthy dried snacks. Additionally, juicy peaches with a high content of organic acids and bioactive compounds, i.e., ‘WB 258’, ‘Spring time’ and ‘Beta’, are suitable for the production of purees, smoothies, and juices.
The PCA analysis carried out for the purposes of this study thus confirmed significant differences in the chemical composition of peach fruit depending on cultivar. At the same time, it indicated some common features of selected cultivars, owing to which it is possible to divide the analyzed peaches into more sweet ones, more sour ones or these with a higher content of polyphenolic compounds. Finally, it has been shown that peach fruit is an interesting raw material with a varied chemical composition and nutritional value.
References
Slavin JL, Lloyd B (2012) Health benefits of fruits and vegetables. Adv Nutr Int Rev J 3:506–516
Heim KE, Tagliaferro AR, Bobilya DJ (2002) Flavonoid antioxidants: chemistry, metabolism and structure–activity relationships. J Nutr Biochem 13:572–584
Rosenbaugh EG, Savalia KK, Manickam DS, Zimmerman MC (2013) Antioxidant-based therapies for angiotensin II-associated cardiovascular diseases. AJP 304(11):R917–R928
Kaya Y, Çebi A, Söylemez N, Demir H, Alp HH, Bakan E (2012) Correlations between oxidative DNA damage, oxidative stress and coenzyme Q10 in patients with coronary artery disease. Int J Med Sci 9(8):621–626
Sies H, Stahl W (1995) Vitamins E and C, beta-carotene, and other carotenoids as antioxidants. AJCN 62:1315–1321
Seymour EM, Singer AAM, Kirakosyan A, Urcuyo-Llanes DE, Kaufman PB, Bolling SF (2008) Altered hyperlipidemia, hepatic steatosis, and hepatic peroxisome proliferator-activated receptors in rats with intake of tart cherry. J Med Food 11:252–259
Devalaraja S, Jain S, Yadav H (2011) Exotic fruits as therapeutic complements for diabetes, obesity and metabolic syndrome. Food Res Int 44:1856–1865
Tetens I, Alinia S (2009) The role of fruit consumption in the prevention of obesity. J Hortic Sci Biotechnol 84(6):47–51
Pijanowski E, Mrożewski S, Horubała A, Jarczyk A (1973) Technologia produktów owocowych i warzywnych, 3th edn. PWRiL, Warszawa
Nowicka P, Wojdyło A, Teleszko M (2017) Effect of mixing different kinds of fruit juice with sour cherry puree on nutritional properties. JFST 54:114–129
Wojdyło A, Nowicka P, Laskowski P, Oszmiański J (2014) Evaluation of sour cherry (Prunus cerasus L.) fruits for their polyphenol content, antioxidant properties and nutritional components. J Agric Food Chem 62(51):12332–12345
Wojdyło A, Nowicka P, Bąbelewski P (2018) Phenolic and carotenoid profile of new goji cultivars and their anti-hyperglycemic, anti-aging and antioxidant properties. J Funct Foods 48:632–642
Torrecillas A, Domingo R, Galego R, Ruiz-Sánchez MC (2000) Apricot tree response to withholding irrigation at different phonological periods. Sci Hort 85(3):201–215
Kheiralipour K, Tabatabaeefar A, Mobli H, Rafiee S, Sahraroo A, Rajabipour A, Jafari A (2008) Some physical properties of apple. Pak J Nutr 7(5):667–672
Zatylny AM, Ziehl WD, St-Pierre RG (2005) Physicochemical properties of fruit of chokecherry (Prunus virginiana L.), highbush cranberry (Viburnumtrilobum Marsh.), and black currant (Ribes nigrum L.) cultivars grown in Saskatchewan. Can J Plant Sci 85(2):425–429
Zhang B, Peng B, Zhang C, Song Z, Ma R (2017) Determination of fruit maturity and its prediction model based on the pericarp index of absorbance difference (I AD) for peaches. PLoS One. https://doi.org/10.1371/journal.pone.0177511
Cirilli M, Bassi D, Ciacciulli A (2016) Sugars in peach fruit: a breeding perspective. Hortic Res 3:15067
Ercisli S, Orhan E (2007) Chemical composition of white (Morus alba), red (Morus rubra) and black (Morus nigra) mulberry fruits. Food Chem 103:1380–1384
Gil MI, Tomas-Barberan FA, Hess-Pierce B, Kader AA (2002) Antioxidant capacities, phenolic compounds, carotenoids, and vitamin C contents of nectarine, peach, and plum cultivars from California. J Agric Food Chem 50:4976–4982
Flores P, Hellin P, Fenoll J (2012) Determination of organic acids in fruits and vegetables by liquid chromatography with tandem-mass spectrometry. Food Chem 132:1049–1054
Wu BH, Quilot B, Génard M, Kervella J, Li SH (2005) Changes in sugar and organic acid concentrations during fruit maturation in peaches, P. davidiana and hybrids as analyzed by principal component analysis. Sci Hortic 103:429–439
Karav S, Eksi A (2012) Antioxidant capacity and total phenolic contents of peach and apricot cultivars harvested from different regions of Turkey. IJFNS 1(4):13–17
Robertson JA, Horvat RJ, Lyon BG, Meredith FI, Senter SD, Okie WR (1990) Comparison of quality characteristics of selected yellow- and white-fleshed peach cultivar. J Food Sci 15:323–334
Nikdel K, Seifi E, Babaie H, Sharifani M, Hemmati K (2016) Physicochemical properties and antioxidant activities of five Iranian pomegranate cultivars (Punica granatum L.) in maturation stage. Acta Agric Slov 107(2):277–286
Cao S, Liang M, Shi L, Shao J, Song C, Bian K, Chen W, Yang Z (2017) Accumulation of carotenoids and expression of carotenogenic genes in peach fruit. Food Chem 214:137–146
Belhadj F, Somrani I, Aissaoui N, Messaoud C, Boussaid M, Marzouki MN (2016) Bioactive compounds contents, antioxidant and antimicrobial activities during ripening of Prunus persica L. varieties from the North West of Tunisia. Food Chem 204:29–36
Bento C, Gonçalves AC, Silva B, Silva LR (2018) Assessing the phenolic profile, antioxidant, antidiabetic and protective effects against oxidative damage in human erythrocytes of peaches from Fundão. J Funct Foods 43:224–223
Nowicka P, Wojdyło A, Samoticha J (2016) Evaluation of phytochemicals, antioxidant capacity, and antidiabetic activity of novel smoothies from selected Prunus fruits. J Funct Foods 25:397–407
Zhong K, Wu J, Wang Z, Chen F, Liao X, Hu X, Zhang Z (2007) Inactivation kinetics and secondary structural change of PEF-treated POD and PPO. Food Chem 100:115–123
Acknowledgements
This work was supported by the Foundation for Polish Science (FNP). Publication was supported by Wroclaw Centre of Biotechnology, the programme The Leading National Research Centre (KNOW) for years 2014–2018 and purpose subsidy 2017 (MNiSW) for The Faculty of Biotechnology and Food Sciences, Wrocław University of Environmental and Life Science.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Compliance with ethics requirements
The research does not include any human subjects and animal experiments.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
OpenAccess This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Nowicka, P., Wojdyło, A. & Laskowski, P. Principal component analysis (PCA) of physicochemical compounds’ content in different cultivars of peach fruits, including qualification and quantification of sugars and organic acids by HPLC. Eur Food Res Technol 245, 929–938 (2019). https://doi.org/10.1007/s00217-019-03233-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00217-019-03233-z