Abstract
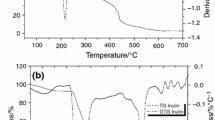
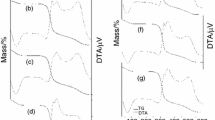
The physicochemical characteristics (thermal stability, glass transition temperature and degree of crystallinity) of inulins, obtained from the tubers of four varieties Jerusalem artichoke (Helianthus tuberosus L.) grown in Bulgaria—Energina, Verona, Topstar and Spindel, were investigated. The inulins obtained had molecular weight from 4,882 to 5,600 Da (degree of polymerization around 30 fructose units). The experimental data from differential scanning calorimetry showed that the glass transition temperature of inulins is between 51 °C and 55 °C. It was found that the first transition (due to evaporation of the unbound water) starts from 77 to 80 °C. The second transition phase started between 152 and 158 °C, and it determines the limit of inulin thermal stability. The data from differential thermal analysis confirmed the results of differential scanning calorimetry with slight differences in the starting temperatures and durations of the phase transitions. X-ray analysis showed that the degree of crystallinity of inulins is very low (except for Spindel inulin—21%).




Similar content being viewed by others
References
Ronkart SN, Blecker CS, Fourmanoir H, Fougnies C, Deroanne C, Herck JCV, Paquot M (2007) Isolation and identification of inulooligosaccharides resulting from inulin hydrolysis. Anal Chim Acta 604:81–87
Dan A, Ghosh S, Moulik SP (2009) Physicochemical studies on the biopolymer inulin: a critical evaluation of its self-aggregation, aggregate-morphology, interaction with water, and thermal stability. Biopolymers 91(9):687–699
De Gennaro S, Birch GG, Parke SA, Stancher B (2000) Studies on the physicochemical properties of inulin and inulin oligomers. Food Chem 68(2):179–183
Niness K (1999) Inulin and oligofructose: what are they? J Nutr 129:1402–1406
Hinrichs WLJ, Prinsen MG, Frijlink HW (2001) Inulin glasses for the stabilization of therapeutic proteins. Int J Pharm 215:163–174
Naskar B, Dan A, Ghosh S, Moulik SP (2010) Characteristic physicochemical features of the biopolymer inulin in solvent added and depleted states. Carbohydr Polym 81:700–706
Coudray C, Demigné C, Rayssiguier Y (2003) Effects of dietary fibers on magnesium absorption in animals and humans. J Nutr 133:1–4
Abrams S, Griffin I, Hawthorne K, Liang L, Gunn S, Darlington G, Ellis K (2005) A combination of prebiotic short- and long-chain inulin-type fructans enhances calcium absorption and bone mineralization in young adolescents. Am J Clin Nutr 82(2):471–476
Chiavaro E, Vittadini E, Corradini C (2007) Physicochemical characterization and stability of inulin gels. Eur Food Res Technol 225:85–94
Poinot P, Arvisenet G, Grua-Priol J, Fillonneau C, Le-Bail A, Prost C (2010) Influence of inulin on bread: kinetics and physico-chemical indicators of the formation of volatile compounds during baking. Food Chem 119(4):1474–1484
Blecker C, Chevalier J-P, Fougnies C, Van Herkck J-C, Deroanne C, Paquot M (2003) Characterisation of different inulin samples by DSC: influence of polymerisation degree on melting temperature. J Therm Anal Calorim 71(1):215–224
Bot A, Erle U, Vreeker R, Agterof W (2004) Influence of crystallisation conditions on the large deformation rheology of inulin gels. Food Hydrocolloids 18(4):547–556
Ronkart S, Blecker C, Fougnies C, Van Herck JC, Wouters J, Paquot M (2006) Determination of physical changes of inulin related to sorption isotherms: an X-ray diffraction, modulated differential scanning calorimetry and environmental scanning electron microscopy study. Carbohydr Polym 63:210–217
Glibowski P, Wasko A (2008) Effect of thermochemical treatment on the structure of inulin and its gelling properties. Int J Food Sci Technol 43:2075–2082
Courtin C, Swennena K, Verjansa P, Delcoura JA (2009) Heat and pH stability of prebiotic arabinoxylooligosaccharides, xylooligosaccharides and fructooligosaccharides. Food Chem 112(4):831–837
Dan A, Ghosh S, Moulik SP (2009) Physicochemistry of the interaction between inulin and alkyltrimethylammonium bromides in aqueous medium and the formed coacevates. J Phys Chem B 113(25):8505–8513
Glibowski P (2010) Effect of thermal and mechanical factors on rheological properties of high performance inulin gels and spreads. J Food Eng 99:106–113
Glibowski P, Pikus S (2011) Amorphous and crystal inulin behavior in a water environment. Carbohydr Polym 83:635–639
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31:426–428
Cakić M, Nikolić G, Ilić L (2002) FTIR spectra of iron (III) complexes with dextran, pullulan and inulin oligomers. Bull Chem Technol Macedonia 21(2):135–146
Engelsen SB, Norgaard L (1996) Comparative vibrational spectroscopy for determination of quality parameters in amidated pectins by chemometrics. Carbohydr Polym 30:9–24
Sinitsya A, Čopiková J, Prutyanov V, Skoblya S, Machovič V (2000) Amidation of highly methoxylated citrus pectin with primary amines. Carbohydr Polym 42:359–368
Zimeri JE, Kokini JL (2003) Phase transitions of inulin-waxy maize starch systems in limited moisture environments. Carbohydr Polym 51:183–190
Bartenev GM, Frenkel SI (1990) Polymer physics. ISBN 5-7245-0454-1. Chemistry, Leningrad (in Rusian)
Schaller-Povolny LA, Smith DA, Labuza TA (2000) Effect of water content and molecular weight on the moisture isotherms and glass transition properties of inulin. Int J Food Prop 3(2):173–192
Ronkart SN, Deroanne C, Paquot M, Fougnies C, Blecker CS (2010) Impact of the crystallisation pathway of inulin on its mono-hydrate to hemi-hydrate thermal transition. Food Chem 119:317–322
Ronkart SN, Paquot M, Fougnies C, Deroanne C, Blecker CS (2009) Effect of water uptake on amorphous inulin properties. Food Hydrocolloids 23:922–927
Kawai K, Fukami K, Thanatuksorn P, Viriyarattanasak C, Kajiwara K (2011) Effects of moisture content, molecular weight, and crystallinity on the glass transition temperature of inulin. Carbohydr Polym 83:934–939
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Panchev, I., Delchev, N., Kovacheva, D. et al. Physicochemical characteristics of inulins obtained from Jerusalem artichoke (Helianthus tuberosus L.). Eur Food Res Technol 233, 889–896 (2011). https://doi.org/10.1007/s00217-011-1584-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00217-011-1584-8