Abstract
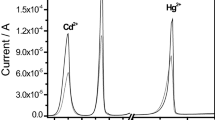
The conditions were worked out for the simultaneous stripping voltammetric determination of components of the ternary Pb(II)-Cu(II)-Cd(II) and binary Ag(I)-Cu(II) systems at a carbon-paste electrode in 2% aqueous solutions of the water soluble polymer polyethyleneimine (PEI) and its thiourea-containing derivative (TU-PEI). Water-soluble polymers are shown to reduce the mutual effects of the components of the binary and ternary system at the electrode surface. The different complex stability of Ag(I), Pb(II) or Cd(II) and Cu(II) with PEI and TU-PEI allows Ag(I), Pb(II) and Cd(II) to be determined in the presence of a large excess of Cu(II).
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 17 July 1997 / Revised: 1 December 1997 / Accepted: 3 December 1997
Rights and permissions
About this article
Cite this article
Osipova, E., Sladkov, V., Kamenev, A. et al. Voltammetric stripping analysis of metal concentrates with polymers soluble in aqueous solutions. Fresenius J Anal Chem 361, 311–313 (1998). https://doi.org/10.1007/s002160050891
Issue Date:
DOI: https://doi.org/10.1007/s002160050891