Abstract
Human exhaled breath is becoming an attractive clinical source as it is foreseen to enable noninvasive diagnosis of many diseases. Because mask devices can be used for efficiently filtering exhaled substances, mask-wearing has been required in the past few years in daily life since the unprecedented COVID-19 pandemic. In recent years, there is a new development of mask devices as new wearable breath samplers for collecting exhaled substances for disease diagnosis and biomarker discovery. This paper attempts to identify new trends in mask samplers for breath analysis. The couplings of mask samplers with different (bio)analytical approaches, including mass spectrometry (MS), polymerase chain reaction (PCR), sensor, and others for breath analysis, are summarized. The developments and applications of mask samplers in disease diagnosis and human health are reviewed. The limitations and future trends of mask samplers are also discussed.
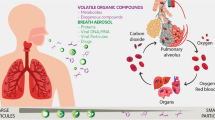
Graphical abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Human exhaled breath is a typical bioaerosol that contains a vast variety of metabolites and bioparticles such as proteins, cytokines, bacteria, and viruses. Much evidence has proven that mask devices can be efficiently used for filtering exhaled infectious viruses and bacteria from patients, and thus the spread of respiratory diseases can be effectively prevented by wearing masks [1,2,3]. In the past few years, mask devices have been widely used in daily life and unprecedented research efforts have been focused on the developments and applications of mask devices since the outbreak of the coronavirus disease 2019 (COVID-19) [2,3,4]. Many studies have been focused on the development history, manufacture, utilization, and impact on the environment of mask devices [1, 5,6,7]. New masks are also increasingly developed for better performances and new functions [7]. In general, the mask is a safe, noninvasive, and cost-effective wearable device to protect wearers.
There are commercially available various types of mask devices, which offer different levels for filtering exhaled and inhaled substances for different purposes, ranging from sneezing droplets to gaseous molecules [7, 8]. Therefore, mask devices can serve as wearable samplers for breath sampling. After sampling, mask samplers can be coupled with various (bio)analytical methods for different applications. By wearing a mask sampler, various exhaled substances such as metabolites, proteins, and microorganisms are allowed to filter onto the mask. In general, the sampling of mask samplers is mainly dependent on permeability and filtration efficiency of mask devices [6, 7]. Compared to traditional breath sampling devices such as gas bag, gas canister, breath condenser, and adsorbent tube [9,10,11,12,13], the mask sampler has many advantages including safe, convenient, simple, wearable, and low-cost, and enables new possibilities to gain insights into human biology [13, 14]. For example, the mask device is a safe breath sampler under highly infectious environments such as COVID-19 and tuberculosis.
In recent years, there is a new trend to develop mask devices as new versatile wearable breath samplers for breath analysis [8, 15]. This paper aims at describing the current developments of mask devices as breath samplers for wearable sampling of exhaled substances, emphasizing the mask types and their versatile couplings with different (bio)analytical methods, and trying to indicate the trends, prospects, and challenges on further developments and applications of mask samplers.
Mask samplers for breath sampling
As shown in Fig. 1, exhaled bioparticles such as microdroplets, bacteria, proteins, and viruses can be directly filtered onto a common mask [16]. Although small volatile molecules such as breath metabolites can pass through the pore of mask, many exhaled volatile metabolites can be selectively collected onto modified masks using different sorbents [17, 18].
Original masks
Direct mask sampling of exhaled microorganisms including bacteria and viruses has attracted intensive attention, because exhaled microorganisms usually relate to infectious diseases. For example, various masks including surgical masks [19], N95 masks [20], three-layer disposable masks [21], woven masks [22], medical disposable anesthetic masks [23], IIR surgical masks [24], and CPAP-type masks [25] have been demonstrated for collecting exhaled severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) from COVID-19 patients. Various exhaled bacteria, e.g., Bacillus, Staphylococcus, and Acinetobacter spp. from patients, were also successfully achieved by mask sampling [26,27,28]. These studies show the convenience of wearable breath sampling and the unique feature that could prevent airborne transmission of exhaled virus/bacteria. However, the low exhaled virus/bacteria load on mask could cause false negative results in clinical practices.
Moreover, it should be noted that the outer layer of masks is exposed to ambient air, and many environmental exposures such as volatile chemicals, (bio)aerosols, and (bio)particles can also be adsorbed onto the mask [29,30,31,32]. In addition, mask devices are mainly manufactured by petrochemical materials, which might cause many residues of mask contaminants (chemical residues and plastic particles) [8, 33,34,35]. Therefore, mask contaminants and air exposures could be confused with exhaled metabolites and bioparticles. It is an important task to identify the exhaled substances rather than the contaminants.
Modified masks
Modifying masks is an attractive strategy for enhanced sampling of target analytes from exhaled breath. The molecular sizes of breath metabolites (~1 nm) are much smaller than the pore size of mask fibers (>100 nm) and thus exhaled breath VOCs can pass through the mask [8]. Therefore, breath metabolites are usually collected by modified masks. Figure 2 shows various typical modified mask devices by fixing sensors or sorbent materials onto the inner surface of the mask surface. For example, solid-phase microextraction (SPME) fibers (Fig. 2a) [36], fabric-phase sorptive membranes (FPSM) (Fig. 2b) [37], porous paper strips (Fig. 2c) [32], adsorbent trap [38, 39], thermal desorption (TD) tubes [40,41,42], carbon-filled sorbent tubes [43], and other devices [44,45,46] can be fixed in masks to extract targeted breath metabolites.
Typical modified mask samplers for breath sampling: (a) SPME-in-mask, reproduced from ref [36] with permission; (b) FPSM-in-mask, reproduced from ref [37] with CC-BY 4.0 license; (c) strip-in-mask, reproduced from ref [32] with permission; (d) sniffer sensor-in-mask, reproduced from ref [77] with permission; (e) porphyrin sensor-in-mask, reproduced from ref [78] with permission; (f) CRISPR sensor-in-mask, reproduced from ref [80] with permission
Furthermore, adsorption materials have been developed to modify masks for enhanced sampling of exhaled microorganisms. For example, gelatin filters [47], cellulose acetate membranes [48], and 3D-PVA matrix [49] were fixed into masks for enhanced adsorption of exhaled bacteria. Huynh et al. [50] developed a breath sampling method for placing electret filter material into homemade PVC masks to collect exhaled viruses (i.e., influenza virus, parainfluenza 3 virus, and human rhinovirus). Particularly, much effort has been devoted for developing modified masks for enhanced sampling of exhaled SARS-CoV-2 from patients with COVID-19., e.g., electrostatic filters [25, 51], PVA strips [52,53,54], gelatin membrane filters [55], gelatin membrane [56], cold trap [57], Steri-Strips™ [58], and copper-coated materials [59].
A modified mask sampler is a simple yet efficient method for enhanced breath sampling of exhaled substances including metabolites and bioparticles. The unique feature of the modified mask is that sorbents in the mask can be designed to extract target analytes. The main limitation of modified masks is that not only exhaled substrates but also contaminates from the mask and ambient air can be collected, especially for small molecules. Designing new materials to selectively and efficiently extract exhaled biomarkers is a new trend in breath sampling.
Mask sampling coupled with (bio)analytical approaches
Analyzing exhaled substances could provide insights into biomarker research and disease diagnosis. Mask samplers can couple with various (bio)analytical techniques for versatile analysis, including mass spectrometry (MS) approaches, polymerase chain reaction (PCR) approaches, sensors, and others. A typical analytical procedure [60], including mask preparation, sample collection, and identification, was proposed (Fig. 3).
Workflow of (bio)analytical procedures of human breath with mask sampling. Solid lines show the conventional processes, while dotted lines show optional processes, reproduced from ref [8] with permission
MS approaches
Mass spectrometry is a powerful analytical technique used to analyze different complex samples with unique advantages in specificity, sensitivity, and speed, and has been widely used for breath analysis [61]. The LC-MS approach is usually used to analyze nonvolatile metabolites [37], while the GC–MS approach is mainly used for analyzing semi-volatile and volatile metabolites [38,39,40,41,42, 62,63,64,65,66,67]. Particularly, mask samplers (Fig. 4a) coupled with portable GC-MS can be used for onsite analysis of breath VOCs (Fig. 4b) [15, 66]. GC-MS and LC-MS are usually combined with multivariate data analysis methods for breath analysis [15, 68, 69]. MALDI-MS approaches are mainly used for analyzing biomacromolecules (e.g., exhaled proteins collected onto mask [70]) and microorganisms (e.g., exhaled bacterial load on mask [28]). Using such microbial MALDI-MS, the microbial cultivation and sample preparation of exhaled microorganisms are necessary processes. Without chromatographic separation and sample pretreatment, some modified masks can be coupled with ambient MS approaches for rapid analysis of breath metabolites via ambient ionization techniques such as direct analysis in real-time (DART) [36] and paper spray ionization (PSI) [32].
Mask sampling coupled with portable GC-MS for onsite investigation: a) werable mask sampler, b) portable GC-MS analysis, reproduced from ref [15] with CC-BY 4.0 license
Mask samplers coupled with MS-based approaches can be viewed as a powerful analytical technique to analyze different exhaled substances, as summarized in Table 1. Breath MS analysis can be divided into two methods: targeted analysis and nontargeted analysis. Targeted analysis mainly focuses on the identification and quantification of specific substances, while the goal of the nontargeted analysis is mostly used to classify samples and to identify or tentatively identify as many substances as possible in breath metabolites using data analysis methods. Targeted and nontargeted MS methods are complementary approaches for evaluating the composition of exhaled substances. The major problem of breath MS analysis is that conventional MS approaches (e.g., LC-MS, GC-MS, MALDI-MS) require large-volume sample consumption and a tedious analytical procedure involving sample storage, transportation, preparation, and separation, while ambient MS approaches usually suffer low sensitivity and matrix effect [71]. Other drawbacks of MS-based approaches in high-cost equipment and low on-site for breath analysis [71].
PCR approaches
PCR is a very sensitive molecular biology technique that allows amplification of a specific segment of DNA and RNA sequences, and has been widely used to identify bacteria and viruses [26, 51]. It is true that there are a variety of viruses and bacteria in exhaled breath bioaerosol, which are larger than most of pore sizes of mask fibers and thus can be directly collected by wearing a mask [8]. For example, Williams and coworkers for the first time developed a mask sampler to offer a highly efficient breath method for sampling exhaled Mycobacterium tuberculosis (Mtb), and then DNA of Mtb was extracted and detected by the PCR approach (Fig. 5) [72,73,74]. These findings showed the presence of active infection both with greater consistency and at an earlier disease stage than with sputum samples. Recent advances in mask samplers coupled with PCR approaches have been demonstrated for a useful bioanalytical tool for identifying various exhaled bacteria [26] and viruses such as SARS-CoV-2, influenza virus, parainfluenza 3 virus, and human rhinovirus [50, 57], as listed in Table 2.
Schematic of mask processing and biological analysis of exhaled Mtb, reproduced from ref [72] with permission
The high resolution of whole-genome sequencing allows precision microbial identification and characterization for accurate microbial investigations, facilitating new insight into respiratory disease and a better understanding of infectious pathogens. Rapid detection and identification of exhaled microorganisms from mask samplers are important tasks for onsite investigation and large-scale medical examination. Although PCR is a sensitive method, the ultra-trace load of exhaled microorganisms is usually far reach to the load limit of PCR detection, which poses a serious risk for false-negative diagnosis [20, 58]. A long time wearing could collect more exhaled microorganisms. Another drawback of PCR test is the labor-intensive sample preparation and time-consuming analytical procedures [75].
Sensor approaches
The chemical and biological sensor is a type of green analytical device that detects and responds to target analytes from complex matrices such as exhaled substances [76]. Two of the most important features of the sensor are selectivity and sensitivity. Modified mask by inserting small sensors into the mask is a novel wearable device for monitoring breath changes (Table 3). Breath metabolites, biomacromolecules, and microorganisms can be detected by sensor-in-mask. Bordbar et al. [77] demonstrated that a strip of colorimetric sensor array was taped in the inner surface of the mask to display the changes of exhaled breath metabolites from patients, healthy and cured participants. Using such sensor array, special breath metabolites could show color changes by interacting with volatile metabolites through colorimetric responses (Fig. 2d). Zazzo et al. [78] embedded porphyrin-based chemical sensor array onto the inner surface of FFP masks to identify breath VOCs (Fig. 2e), showing the monitoring of various special breath VOCs, e.g., ethanol, hexane, toluene, acetone, triethylamine, and acetic acid, which are mainly generated due to the ingestion of beverages (e.g., coffee and wine) and solid food (banana and mint-flavored candies). Jin et al. [79] placed sensing strips into masks to detect α-amylase from exhaled aerosols and its distribution in masks. Nguyen et al. [80] invented a modified mask device that puts a lyophilized CRISPR sensor into an N95 mask to detect SARS-CoV-2 (Fig. 2f).
The unique feature of the sensor-in-mask technique is that sampling and detection are integrated into a mask sampler, giving a noninvasive, in vivo, wearable, and real-time monitoring of exhaled substances. The relatively low cost is another advantage of sensor technology. The major limitation of the sensor-in-mask technique is that sensors can be only used for detecting some special analytes [81], which need to be pre-identified by other (bio)analytical methods such as MS, PCR, or others. Other obvious drawbacks of the sensor-in-mask technique include low accuracy, low precision, as well as low sensitivity.
Other approaches
Coupling mask sampling with other (bio)analytical methods further promotes and prospers the use of mask samplers in breath analysis (Table 3). For example, a flame photometer (FP) was used to determine the concentration of certain sodium ions that were collected by a miniature and low-weight respirable sampler designed for FFP masks [82]. A mask sampler coupled with immunochemistry analysis (ICA) was applied to analyze the exhaled cytokines [18]. Schorer et al. [16] investigated the microdroplets, proteins, and virus-like particles sprayed onto surgical masks via infrared (IR) spectroscopy, showing that the IR optical technique is a promising tool for direct virus detection at the surface of masks. Davies et al. [27] applied Henderson apparatus (HA) to test the effectiveness of homemade cotton T-shirt fabric surgical masks using Bacillus atrophaeus, bacteriophage MS2 produced during coughing. A flame photometer (FP) was used to determine the concentration of certain sodium ions that were collected by a modified mask [82].
The key performance indicators of (bio)analytical methods include sensitivity, specificity, and speed of signal response, which are mainly determined by the sample pretreatment and analytical mechanism. These (bio)analytical methods including FP, ICA, IR, and HA show their main advantages of relatively low-cost, easy operation, and versatile couplings with mask samplers, and show their potential abilities for new applications. However, there are several drawbacks of mentioned techniques consisting in limited detectable analytes and relatively low analytical performances.
Outlook
A mask sampler is a new sampling technique that fulfils the criteria of simple, low-cost, in vivo, noninvasive, easy-to-operate, wearable, and higher acceptability among patients by comparing with other clinical samples such as urine, blood, and sputum. Because exhaled metabolites, proteins and microorganisms can be collected by mask devices, mask sampling coupled with multi-omics approaches (e.g., metabolomics, proteomics, and microbiomics) is a new trend which allow a better elucidation of the relation between the breath compositions and human biology studies such as diseases, behaviors, and exposures. Furthermore, coupling mask–based breath analyses with new data analysis methods such as big data, artificial intelligence, and machine learning are also highly expected to be a trend for elucidating exhaled substances. Onsite screening rather than a laboratory examination will be more beneficial for patients, and the coupling of mask samplers with portable analytical instruments for on-site investigation is a new trend in breath analysis. Therefore, it can be expected that mask samplers will become a new clinical tool in the future.
Mask device as a new type of breath samplers for breath analysis, many limitations and potential pitfalls must be considered. Current main limitations of mask sampler techniques are the relatively low efficiency of breath sampling and the need of sample extraction for further analysis. It is expected to design new mask samplers for enhanced collection of exhaled substances, and to couple with more sensitive and specific detection methods. Another problem is chemical and biological contamination, which could come from mask materials and ambient air. It is expected that the design and manufacture of mask samplers can be significantly improved. A potential pitfall is that quantitative analysis of continuous exhaled breath using mask samplers is unclear; it is of paramount importance to elucidate the quantitative evaluation of human health state and disease development, and the quantitative relationship between breath and other clinical specimens in the future.
Abbreviations
- AMR:
-
Antimicrobial resistance
- CA:
-
Cellulose acetate
- COVID-19:
-
Coronavirus disease 2019
- CPAP:
-
Continuous positive airway pressure
- DART:
-
Direct analysis in real time
- DMS:
-
Differential mobility spectrometer
- DNA:
-
Deoxyribonucleic acid
- EBA:
-
Exhaled breath aerosols
- FFP:
-
Filtering facepiece
- FP:
-
Flame photometer
- FPSM:
-
Fabric-phase sorptive membrane
- GC:
-
Gas chromatography
- HA:
-
Henderson apparatus
- ICA:
-
Immunochemistry analysis
- IM:
-
Ion mobility
- LC:
-
Liquid chromatography
- MALDI:
-
Matrix-assisted laser desorption/ionization
- MS:
-
Mass spectrometry
- Mtb:
-
Mycobacterium tuberculosis
- PCR:
-
Polymerase chain reaction
- PSI:
-
Paper spray ionization
- PVA:
-
Polyvinyl alcohol
- RNA:
-
Ribonucleic acid
- RT:
-
Real-time
- SARS-CoV-2:
-
Severe acute respiratory syndrome coronavirus 2
- SFP:
-
Sodium flame photometry
- SPME:
-
Solid-phase microextraction
- TB:
-
Tuberculosis
- TD:
-
Thermal desorption
- TEA:
-
Triethylamine
- TOF:
-
Time of flight
- VOCs:
-
Volatile organic compounds
References
Tuñón-Molina A, Takayama K, Redwan EM, Uversky VN, Andrés J, Serrano-Aroca Á. Protective face masks: current status and future trends. ACS Appl Mater Interfaces. 2021;13(48):56725–51.
Leung NHL, Chu DKW, Shiu EYC, Chan K-H, McDevitt JJ, Hau BJP, Yen H-L, Li Y, Ip DKM, Peiris JSM, Seto W-H, Leung GM, Milton DK, Cowling BJ. Respiratory virus shedding in exhaled breath and efficacy of face masks. Nat Med. 2020;26(5):676–80.
Cheng Y, Ma N, Witt C, Rapp S, Wild PS, Andreae MO, Pöschl U, Su H. Face masks effectively limit the probability of SARS-CoV-2 transmission. Science. 2021; 372(6549): 1439–43.
Rab S, Javaid M, Haleem A, Vaishya R. Face masks are new normal after COVID-19 pandemic. Diabetes Metab Syndr. 2020;14(6):1617–9.
Matuschek C, Moll F, Fangerau H, Fischer JC, Zänker K, van Griensven M, Schneider M, Kindgen-Milles D, Knoefel WT, Lichtenberg A, Tamaskovics B, Djiepmo-Njanang FJ, Budach W, Corradini S, Häussinger D, Feldt T, Jensen B, Pelka R, Orth K, Peiper M, Grebe O, Maas K, Bölke E, Haussmann J. The history and value of face masks. Eur J Med Res. 2020;25(1):23.
Karmacharya M, Kumar S, Gulenko O, Cho Y-K. Advances in facemasks during the COVID-19 pandemic era. ACS Appl Bio Mater. 2021;4(5):3891–908.
Chua MH, Cheng W, Goh SS, Kong J, Li B, Lim JYC, Mao L, Wang S, Xue K, Yang L, Ye E, Zhang K, Cheong WCD, Tan BH, Li Z, Tan BH, Loh XJ. Face masks in the new COVID-19 normal: materials, testing, and perspectives. Research. 2020; 2020:7286735.
Hu B. Recent advances in facemask devices for in vivo sampling of human exhaled breath aerosols and inhalable environmental exposures. Trends Anal Chem. 2022; 151:116600.
Xu F, Zhou J, Yang H, Chen L, Zhong J, Peng Y, Wu K, Wang Y, Fan H, Yang X, Zhao Y. Recent advances in exhaled breath sample preparation technologies for drug of abuse detection. Trends Anal Chem. 2022; 157:116828.
Lawal O, Ahmed WM, Nijsen TME, Goodacre R, Fowler SJ. Exhaled breath analysis: a review of ‘breath-taking’ methods for off-line analysis. Metabolomics. 2017;13(10):110.
Grob NM, Aytekin M, Dweik RA. Biomarkers in exhaled breath condensate: a review of collection, processing and analysis. J Breath Res. 2008;2(3): 037004.
Ghosh C, Singh V, Grandy J, Pawliszyn J. Recent advances in breath analysis to track human health by new enrichment technologies. J Sep Sci. 2020;43(1):226–40.
Hu B, Ouyang G. In situ solid phase microextraction sampling of analytes from living human objects for mass spectrometry analysis. Trends Anal Chem. 2021; 143:116368.
Szunerits S, Dӧrfler H, Pagneux Q, Daniel J, Wadekar S, Woitrain E, Ladage D, Montaigne D, Boukherroub R. Exhaled breath condensate as bioanalyte: from collection considerations to biomarker sensing. Anal Bioanal Chem. 2023;415(1):27–34.
Yuan ZC, Hu B. Mass spectrometry-based human breath analysis: towards COVID-19 diagnosis and research. J Anal Test. 2021;5(4):287–97.
Schorer V, Haas J, Stach R, Kokoric V, Gross R, Muench J, Hummel T, Sobek H, Mennig J, Mizaikoff B. Towards the direct detection of viral materials at the surface of protective face masks via infrared spectroscopy. Sci Rep. 2022;12(1):2309.
Phukkaphan N, Eamsa-Ard T, Aunsa-Ard W, Khunarak C, Nitivanichsakul T, Roongpuvapaht B, Kerdcharoen T. Detection of COVID-19 infection based on electronic nose technique: preliminary study. The 2022 International Electrical Engineering Congress (iEECON2022), IEEE, 2022, pp. 1–4.
Geer Wallace MA, Pleil JD, Madden MC. Identifying organic compounds in exhaled breath aerosol: non-invasive sampling from respirator surfaces and disposable hospital masks. J Aerosol Sci. 2019; 137:105444.
Nagle S, Tandjaoui-Lambiotte Y, Boubaya M, Athenais G, Alloui C, Bloch-Queyrat C, Carbonnelle E, Brichler S, Cohen Y, Zahar JR, Delagreverie H. Environmental SARS-CoV-2 contamination in hospital rooms of patients with acute COVID-19. J Hosp Infect. 2022; 126:116–22.
Kanaujia R, Biswal M, Angrup A, Ray P. Inhale, then exhale: start afresh to diagnose severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) by non-invasive face-mask sampling technique. Clin Microbiol Infect. 2020;26(12):1701–2.
Wang X, Grobe N, Haq Z, Thwin O, Fuentes LR, Maddux D, Kotanko P. Testing of worn face masks for timely diagnosis of SARS-CoV-2 in hemodialysis patients. J Am Soc Nephrol. 2021;32(11):2728–30.
Mello VM, Eller CM, Salvio AL, Nascimento FF, Figueiredo CM, Silva E, Sousa PSF, Costa PF, Paiva AAP, Mares-Guias M, Lemos ERS, Horta MAP. Effectiveness of face masks in blocking the transmission of SARS-CoV-2: a preliminary evaluation of masks used by SARS-CoV-2-infected individuals. PLoS ONE. 2022;17(2): e0264389.
Johnson TJ, Nishida RT, Sonpar AP, Lin YJ, Watson KA, Smith SW, Conly JM, Evans DH, Olfert JS. Viral load of SARS-CoV-2 in droplets and bioaerosols directly captured during breathing, speaking and coughing. Sci Rep. 2022;12(1):3484.
de Man P, Ortiz MA, Bluyssen PM, de Man SJ, Rentmeester MJ, van der Vliet M, Wils EJ, Ong DSY. Airborne SARS-CoV-2 in home and hospital environments investigated with a high-powered air sampler. J Hosp Infect. 2022; 119:126–31.
Smolinska A, Jessop DS, Pappan KL, De Saedeleer A, Kang A, Martin AL, Allsworth M, Tyson C, Bos MP, Clancy M, Morel M, Cooke T, Dymond T, Harris C, Galloway J, Bresser P, Dijkstra N, Jagesar V, Savelkoul PHM, Beuken EVH, Nix WHV, Louis R, Delvaux M, Calmes D, Ernst B, Pollini S, Peired A, Guiot J, Tomassetti S, Budding AE, McCaughan F, Marciniak SJ, van der Schee MP. The SARS-CoV-2 viral load in COVID-19 patients is lower on face mask filters than on nasopharyngeal swabs. Sci Rep. 2021;11(1):13476.
Delanghe L, Cauwenberghs E, Spacova I, De Boeck I, Van Beeck W, Pepermans K, Claes I, Vandenheuvel D, Verhoeven V, Lebeer S. Cotton and surgical face masks in community settings: bacterial contamination and face mask hygiene. Front Med. 2021; 8:732047.
Davies A, Thompson KA, Giri K, Kafatos G, Walker J, Bennett A. Testing the efficacy of homemade masks: would they protect in an influenza pandemic? Disaster Med Public Health Prep. 2013;7(4):413–8.
Gund MP, Naim J, Hannig M, Halfmann A, Gartner B, Boros G, Rupf S. CHX and a face shield cannot prevent contamination of surgical masks. Front Med. 2022; 9:896308.
Chan W, Jin L, Sun Z, Griffith SM, Yu JZ. Fabric masks as a personal dosimeter for quantifying exposure to airborne polycyclic aromatic hydrocarbons. Environ Sci Technol. 2021;55(8):5128–35.
Chan W, Guo W, Yu JZ. Polyurethane-based face mask as a sampling device for environmental tobacco smoke. Anal Chem. 2021;93(41):13912–8.
Sun Z, Guo W, Chan CK, Jin L, Griffith SM, Yu JZ, Chan W. Polyurethane foam face masks as a dosimeter for quantifying personal exposure to airborne volatile and semi-volatile organic compounds. Chem Res Toxicol. 2022; 35(9):1604–13.
Cai SH, Di D, Yuan ZC, Chen W, Hu B. Paper-in-facemask device for direct mass spectrometry analysis of human respiratory aerosols and environmental exposures via wearable continuous-flow adsorptive sampling: a proof-of-concept study. Anal Chem. 2021;93(41):13743–8.
Liu R, Mabury SA. Single-use face masks as a potential source of synthetic antioxidants to the environment. Environ Sci Technol Lett. 2021;8(8):651–5.
Xie H, Han W, Xie Q, Xu T, Zhu M, Chen J. Face mask—a potential source of phthalate exposure for human. J Hazard Mater. 2022; 422:126848.
Chang Y, Huang R-J, Cheng K, Lin C, Ling Q, Haque MM, Ovadnevaite J, O’Dowd C. Highly time-resolved and nontargeted characterization of volatile organic compound emissions from face masks. Environ Sci Technol Lett. 2022; 9(12):1007–13.
Yuan ZC, Li W, Wu L, Huang D, Wu M, Hu B. Solid-phase microextraction fiber in face mask for in vivo sampling and direct mass spectrometry analysis of exhaled breath aerosol. Anal Chem. 2020;92(17):11543–7.
Locatelli M, Tartaglia A, Ulusoy HI, Ulusoy S, Savini F, Rossi S, Santavenere F, Merone GM, Bassotti E, D’Ovidio C, Rosato E, Furton KG, Kabir A. Fabric-phase sorptive membrane array as a noninvasive in vivo sampling device for human exposure to different compounds. Anal Chem. 2021;93(4):1957–61.
Guallar-Hoyas C, Turner MA, Blackburn GJ, Wilson ID, Thomas CP. A workflow for the metabolomic/metabonomic investigation of exhaled breath using thermal desorption GC–MS. Bioanalysis. 2012;4(18):2227–37.
Gahleitner F, Guallar-Hoyas C, Beardsmore CS, Pandya HC, Thomas CP. Metabolomics pilot study to identify volatile organic compound markers of childhood asthma in exhaled breath. Bioanalysis. 2013;5(18):2239–47.
Woodfield G, Belluomo I, Laponogov I, Veselkov K, Cross AJ, Hanna GB, Group CW. Diagnostic performance of a noninvasive breath test for colorectal cancer: COBRA1 study. Gastroenterology. 2022; 163(6):1447–9.
Bhavra KK, Wilde M, Richardson M, Cordell R, Thomas P, Zhao B, Bryant L, Brightling CE, Ibrahim W, Salman D, Siddiqui S, Monks P, Gaillard EA. The utility of a standardised breath sampler in school age children within a real-world prospective study. J Breath Res. 2022;16(2): 027104.
Khan MS, Cuda S, Karere GM, Cox LA, Bishop AC. Breath biomarkers of insulin resistance in pre-diabetic Hispanic adolescents with obesity. Sci Rep. 2022;12:339.
Jin L, Griffith SM, Sun Z, Yu JZ, Chan W. On the flip side of mask wearing: increased exposure to volatile organic compounds and a risk-reducing solution. Environ Sci Technol. 2021;55(20):14095–104.
Jankovic J, Jones W, Burkhart J, Noonan G. Environmental study of firefighters. Ann Occup Hyg. 1991;35(6):581–602.
Mogridge R, Stacey P, Forder J. A new miniature respirable sampler for in-mask sampling: Part 2—Tests performed inside the mask. Ann Occup Hyg. 2016;60(9):1084–91.
Yuan ZC, Zhang Y, Cai SH, Chen W, Hu B. Solid phase microextraction for human breath analysis of environmental and occupational exposures: a review. Advances in Sample Preparation. 2022; 3:100023.
Kennedy M, Ramsheh MY, Williams CML, Auty J, Haldar K, Abdulwhhab M, Brightling CE, Barer MR. Face mask sampling reveals antimicrobial resistance genes in exhaled aerosols from patients with chronic obstructive pulmonary disease and healthy volunteers. BMJ Open Respir Res. 2018;5(1): e000321.
Shaikh A, Sriraman K, Vaswani S, Oswal V, Mistry N. Detection of Mycobacterium tuberculosis RNA in bioaerosols from pulmonary tuberculosis patients. Int J Infect Dis. 2019; 86:5–11.
Al-Taie A, Pan J, Polak P, Barer MR, Han X, Abbott AP. Mechanical properties of 3-D printed polyvinyl alcohol matrix for detection of respiratory pathogens. J Mech Behav Biomed Mater. 2020; 112:104066.
Huynh KN, Oliver BG, Stelzer S, Rawlinson WD, Tovey ER. A new method for sampling and detection of exhaled respiratory virus aerosols. Clin Infect Dis. 2008;46(1):93–5.
Kim HS, Lee H, Park J, Abbas N, Kang S, Hyun H, Seong H, Yoon JG, Noh JY, Kim WJ, Shin S. Collection and detection of SARS-CoV-2 in exhaled breath using face mask. PLoS ONE. 2022;17(8): e0270765.
Pan D, Williams CM, Decker J, Fletcher E, Sze S, Assadi S, Haigh R, Saleem B, Nazareth J, Garton NJ, Pareek M, Barer MR. Exhaled SARS-CoV-2 RNA viral load kinetics measured by facemask sampling associates with household transmission. Clin Microbiol Infect. 2023; 29(2):254.e1-254.e6.
Gallichotte EN, Windsor W, Watts S, Sexton N, Henry C, Jaenisch T, Lamb MM, Winstanley G, Adams B, Chu M, Ebel GD. Detection of SARS-CoV-2 in exhaled air using non-invasive embedded strips in masks. Am J Infect Control. 2022;50(8):890–7.
Williams CM, Pan D, Decker J, Wisniewska A, Fletcher E, Sze S, Assadi S, Haigh R, Abdulwhhab M, Bird P, Holmes CW, Al-Taie A, Saleem B, Pan J, Garton NJ, Pareek M, Barer MR. Exhaled SARS-CoV-2 quantified by face-mask sampling in hospitalised patients with COVID-19. J Infect. 2021;82(6):253–9.
Verma R, Kim E, Degner N, Walter KS, Singh U, Andrews JR. Variation in severe acute respiratory syndrome coronavirus 2 bioaerosol production in exhaled breath. Open Forum Infect Dis. 2022; 9(1): ofab600.
Sriraman K, Shaikh A, Parikh S, Udupa S, Chatterjee N, Shastri J, Mistry N. Non-invasive adapted N-95 mask sampling captures variation in viral particles expelled by COVID-19 patients: implications in understanding SARS-CoV2 transmission. PLoS ONE. 2021;16(4):e0249525.
Daniels J, Wadekar S, DeCubellis K, Jackson GW, Chiu AS, Pagneux Q, Saada H, Engelmann I, Ogiez J, Loze-Warot D, Boukherroub R, Szunerits S. A mask-based diagnostic platform for point-of-care screening of Covid-19. Biosens Bioelectron. 2021; 192:113486.
Ng DHL, Sim MY, Huang HH, Sim JXY, Low JGH, Lim JKS. Feasibility and utility of facemask sampling in the detection of SARS-CoV-2 during an ongoing pandemic. Eur J Clin Microbiol Infect Dis. 2021;40(12):2489–96.
Jung S, Yang JY, Byeon EY, Kim DG, Lee DG, Ryoo S, Lee S, Shin CW, Jang HW, Kim HJ, Lee S. Copper-coated polypropylene filter face mask with SARS-CoV-2 antiviral ability. Polymers. 2021;13(9):1367.
Orton CM, Symons HE, Moseley B, Archer J, Watson NA, Philip KEJ, Sheikh S, Saccente-Kennedy B, Costello D, Browne WJ, Calder JD, Bzdek BR, Hull JH, Reid JP, Shah PL. A comparison of respiratory particle emission rates at rest and while speaking or exercising. Commun Med (Lond). 2022; 2(1):44.
Bruderer T, Gaisl T, Gaugg MT, Nowak N, Streckenbach B, Müller S, Moeller A, Kohler M, Zenobi R. On-line analysis of exhaled breath. Chem Rev. 2019;119(19):10803–28.
Basanta M, Koimtzis T, Singh D, Wilson I, Thomas CL. An adaptive breath sampler for use with human subjects with an impaired respiratory function. Analyst. 2007;132(2):153–63.
Basanta M, Jarvis RM, Xu Y, Blackburn G, Tal-Singer R, Woodcock A, Singh D, Goodacre R, Thomas CL, Fowler SJ. Non-invasive metabolomic analysis of breath using differential mobility spectrometry in patients with chronic obstructive pulmonary disease and healthy smokers. Analyst. 2010;135(2):315–20.
Xu Y, Fowler SJ, Bayat A, Goodacre R. Chemometrics models for overcoming high between subject variability: applications in clinical metabolic profiling studies. Metabolomics. 2014;10(3):375–85.
Basanta M, Ibrahim B, Dockry R, Douce D, Morris M, Singh D, Woodcock A, Fowler SJ. Exhaled volatile organic compounds for phenotyping chronic obstructive pulmonary disease: a cross-sectional study. Respir Res. 2012;13(1):72.
Chen WN, Yuan ZC, Cai SH, Zou YT, Xin GB, Di DD, Song X, Zhao P, Wu MM, Hu B. Gas chromatography-mass spectrometry analysis of human exhaled volatile organic compounds via wearable facemask microextraction sampling. Chin J Anal Chem 2022; 50(3):445–53.
De Vietro N, Aresta AM, Picciariello A, Altomare DF, Lucarelli G, Di Gilio A, Palmisani J, De Gennaro G, Zambonin C. Optimization of a breath analysis methodology to potentially diagnose transplanted kidney rejection: a preclinic study. Appl Sci. 2023;13(5):2852.
Rattray NJW, Hamrang Z, Trivedi DK, Goodacre R, Fowler SJ. Taking your breath away: metabolomics breathes life in to personalized medicine. Trends Biotechnol. 2014;32(10):538–48.
Boots AW, Bos LD, van der Schee MP, van Schooten F-J, Sterk PJ. Exhaled molecular fingerprinting in diagnosis and monitoring: validating volatile promises. Trends Mol Med. 2015;21(10):633–44.
Hu B. Face mask microextraction sampling of human exhaled breath aerosol for multidimensional mass spectrometry analysis. The 32 CCS Congress, CCS, Zhuhai, China, 2021, pp. 47-O-007.
Kuo T-H, Dutkiewicz EP, Pei J, Hsu C-C. Ambient ionization mass spectrometry today and tomorrow: embracing challenges and opportunities. Anal Chem. 2020;92(3):2353–63.
Williams CM, Cheah ES, Malkin J, Patel H, Otu J, Mlaga K, Sutherland JS, Antonio M, Perera N, Woltmann G, Haldar P, Garton NJ, Barer MR. Face mask sampling for the detection of Mycobacterium tuberculosis in expelled aerosols. PLoS ONE. 2014;9(8): e104921.
Williams C, Haldar P, Barer M. Mask sampling in pulmonary tuberculosis. Thorax. 2013;68(Suppl 3):A89–A89.
Williams CM, Abdulwhhab M, Birring SS, De Kock E, Garton NJ, Townsend E, Pareek M, Al-Taie A, Pan J, Ganatra R, Stoltz AC, Haldar P, Barer MR. Exhaled Mycobacterium tuberculosis output and detection of subclinical disease by face-mask sampling: prospective observational studies. Lancet Infect Dis. 2020;20(5):607–17.
Choi JR. Development of point-of-care biosensors for COVID-19. Front Chem. 2020;8:517.
del Valle M. Sensors as green tools in analytical chemistry. Curr Opin Green Sustain Chem. 2021;31:100501.
Bordbar MM, Samadinia H, Hajian A, Sheini A, Safaei E, Aboonajmi J, Arduini F, Sharghi H, Hashemi P, Khoshsafar H, Ghanei M, Bagheri H. Mask assistance to colorimetric sniffers for detection of Covid-19 disease using exhaled breath metabolites. Sens Actuators B Chem. 2022; 369:132379.
Di Zazzo L, Magna G, Lucentini M, Stefanelli M, Paolesse R, Di Natale C. Sensor-embedded face masks for detection of volatiles in breath: a proof of concept study. Chemosensors. 2021;9(12):356.
Jin Z, Jorns A, Yim W, Wing R, Mantri Y, Zhou J, Zhou J, Wu Z, Moore C, Penny WF, Jokerst JV. Mapping aerosolized saliva on face coverings for biosensing applications. Anal Chem. 2021;93(31):11025–32.
Nguyen PQ, Soenksen LR, Donghia NM, Angenent-Mari NM, de Puig H, Huang A, Lee R, Slomovic S, Galbersanini T, Lansberry G, Sallum HM, Zhao EM, Niemi JB, Collins JJ. Wearable materials with embedded synthetic biology sensors for biomolecule detection. Nat Biotechnol. 2021;39(11):1366–74.
Peveler WJ, Yazdani M, Rotello VM. Selectivity and specificity: pros and cons in sensing. ACS Sensors. 2016;1(11):1282–5.
Mogridge R, Stacey P, Forder J. A new miniature respirable sampler for in-mask sampling: Part 2-Tests performed inside the mask. Ann Occup Hyg. 2016;60(9):1084–91.
Funding
This work was supported by the National Natural Science Foundation of China (No. 22127810) and the Double Hundred Talents Program of Jinan University (No. DHT/XP2021289).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Published in the topical collection Young Investigators in (Bio-)Analytical Chemistry 2023 with guest editors Zhi-Yuan Gu, Beatriz Jurado-Sánchez, Thomas H. Linz, Leandro Wang Hantao, Nongnoot Wongkaew, and Peng Wu.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, X., Hu, B. Mask device as a new wearable sampler for breath analysis: what can we expect in the future?. Anal Bioanal Chem 415, 3759–3768 (2023). https://doi.org/10.1007/s00216-023-04673-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-023-04673-z