Abstract
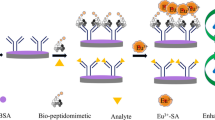
Abrin is a highly toxic ribosome-inactivating protein, which could be used as a biological warfare agent and terrorist weapon, and thus needs to be detected efficiently and accurately. Affibodies are a new class of engineered affinity proteins with small size, high affinity, high stability, favorable folding and good robustness, but they have rarely played a role in biological detection. In this work, we establish a novel electrochemiluminescence (ECL) method for abrin detection with a phage display affibody as the specific probe for the first time, to our knowledge, and a portable biosensor based on a screen-printed electrode (SPE) as the testing platform. On the basis of the double antibody sandwich structure in our previous work, we used a phage display affibody instead of monoclonal antibody as a new specific labeled probe. Due to numerous signal molecules labeled on M13 phages, significant signal amplification was achieved in this experiment. Under optimized conditions, a linear dependence was observed from 0.005 to 100 ng/mL with a limit of detection (LOD) of 5 pg/mL. This assay also showed good reproducibility and specificity, and performed well in the detection of simulated samples. Considering its high sensitivity, interference resistance and convenience, this new biosensing system based on phage display affibodies and a portable ECL biosensor holds promise for in situ detection of toxins and pollutants in different environments.
Graphical abstract








Similar content being viewed by others
References
Olsnes S, Sandvig K, Eiklid K, Pihl A. Properties and action mechanism of the toxic lectin modeccin: interaction with cell lines resistant to modeccin, abrin, and ricin. J Supramol Struct. 1978;9:15–25.
Audi J, Belson M, Patel M, Schier J, Osterloh J. Ricin poisoning: a comprehensive review. JAMA. 2005;294:2342–51.
Moran GJ. Threats in bioterrorism. II: CDC category B and C agents. Emerg Med Clin North Am. 2002;20:311–30.
Bradberry SM, Dickers KJ, Rice P, Griffiths GD, Vale JA. Ricin poisoning. Toxicol Rev. 2003;22:65–70.
Dickers KJ, Bradberry SM, Rice P, Griffiths GD, Vale JA. Abrin poisoning. Toxicol Rev. 2003;22:137–42.
Garber EA. Toxicity and detection of ricin and abrin in beverages. J Food Prot. 2008;71:1875–83.
Garber EA, Walker JL, O’Brien TW. Detection of abrin in food using enzyme-linked immunosorbent assay and electrochemiluminescence technologies. J Food Prot. 2008;71:1868–74.
Zhou Y, Tian XL, YS, Pan FG, Zhang YY, Zhang JH, et al. Development of a monoclonal antibody-based sandwich-type enzyme-linked immunosorbent assay (ELISA) for detection of abrin in food samples. Food Chem. 2012;135:2661–5.
Tang JJ, Yu T, Guo L, Xie JW, Shao NS, He ZK. In vitro selection of DNA aptamer against abrin toxin and aptamer-based abrin direct detection. Biosens Bioelectron. 2007;22:2456–63.
Mu XH, Zhou ZQ, Tong ZY, Liu B, Hao LQ. Detection of Abrin by Piezoelectric Immunosensor Based on Biotin-Avidin System. Chin J Anal Chem. 2009;37:1499–502.
Gao S, Nie C, Wang JH, Wang J, Kang L, Zhou Y, et al. Colloidal Gold-Based Immunochromatographic Test Strip for Rapid Detection of Abrin in Food Samples. J Food Prot. 2012;75:112–7.
Yang H, Deng M, Ga S, Chen SH, Kang LK, Wang JH, et al. Capillary-driven surface-enhanced Raman scattering (SERS)-based microfluidic chip for abrin detection. Nanoscale Res Lett. 2014;9:138.
Liu S, Tong ZY, Mu XH, Liu B, Du B, Liu ZW, et al. Detection of Abrin by Electrochemiluminescence Biosensor Based on Screen Printed Electrode. Sensors. 2018;18:357.
Liu B, Tong ZY, Liu W, Hao LQ, Mu XH, Huang QB. Determination of Abrin by Electrochemiluminescence Immunosensor Based on Phage-displayed Antibody. Chin J Anal Chem. 2013;41:1449–53.
Ge L, Yan JX, Song XR, Yan M, Ge SG, Yu JH. Three-dimensional paper-based electrochemiluminescence immunodevice for multiplexed measurement of biomarkers and point-of-care testing. Biomaterials. 2012;33:1024–31.
Wang S, Ge L, Yan M, Yu JH, Song XR, Ge SG, et al. 3D microfluidic origami electrochemiluminescence immunodevice for sensitive point-of-care testing of carcinoma antigen 125. Sensors Actuators B Chem. 2013;176:1–8.
Yan JX, Yan M, Ge L, Ge SG, Yu JH. An origami electrochemiluminescence immunosensor based on gold/graphene for specific, sensitive point-of-care testing of arcinoembryonic antigen. Sensors Actuators B Chem. 2014;193:247–54.
Spehar-Délèze AM, Julich S, Gransee R, Tomaso H, Dulay SB, O'Sullivan CK. Electrochemiluminescence (ECL) immunosensor for detection of Francisella tularensis on screen-printed gold electrode array. Anal Bioanal Chem. 2016;408:7147–53.
Zhang M, Liu H, Chen L, Yan M, Ge L, Ge SG, et al. A disposable electrochemiluminescence device for ultrasensitive monitoring of K562 leukemia cells based on aptamers and ZnO@carbon quantum dots. Biosens Bioelectron. 2013;49:79–85.
Feng XB, Gan N, Zhang HR, Yan Q, Li TH, Cao YT, et al. A novel “dual-potential” electrochemiluminescence aptasensor array using CdS quantum dots and luminol-gold nanoparticles as labels for simultaneous detection of malachite green and chloramphenicol. Biosens Bioelectron. 2015;74:587–93.
Leca BD, Verdier AM, Blum LJ. Screen-printed electrodes as disposable or reusable optical devices for luminol electrochemiluminescence. Sensors Actuators B Chem. 2001;74:190–3.
Corgier BP, Marquette CA, Blum LJ. Screen-printed electrode microarray for electrochemiluminescent measurements. Anal Chim Acta. 2005;538:1–7.
Ballesta-Claver J, Velazquez PS, Valencia-Miron MC, Capitan-Vallvey LF. SPE biosensor for cholesterol in serum samples based on electrochemiluminescent luminol copolymer. Talanta. 2011;86:178–85.
Ballesta-Claver J, Rodríguez-Gómez R, Capitán-Vallvey LF. Disposable biosensor based on cathodic electrochemiluminescence of tris(2,2-bipyridine)ruthenium(II) for uric acid determination. Anal Chim Acta. 2013;770:153–60.
Zhou J, Gan N, Hu FT, Li TH, Zhou HK, Li X, et al. A single antibody sandwich electrochemiluminescence immunosensor based on protein magnetic molecularly imprinted polymers mimicking capture probes. Sensors Actuators B Chem. 2013;186:300–7.
Nilsson B, Moks T, Jansson B, Abrahmsen L, Elmblad A, Holmgren E. A synthetic Ig G-binding domain based on staphylococcal protein A. Protein Eng. 1987;1:107–13.
Brausted AC, Wells JA. Minimizing a binding domain from protein A. Proc Natl Acad Sci U S A. 1996;93:5688–92.
Zhang L, Lu Y, Zhong YQ. Affibody molecules: a new class of ligands with high affinity. J Int Pharm Res. 2012;2:127–31.
Li S, Hao ZM. An Engineered Affinity Protein-affibody. Prog Biochem Biophys. 2012;39:137–41.
Nord K, Nilsson J, Nilsson B, Uhlen M, Nygren PA. A combinatorial library of an alpha-helical bacterial receptor domain. Protein Eng. 1995;8:601–8.
Mu XH, Tong ZY, Huang QB, Liu B, Liu ZW, Hao LQ, et al. An electrochemiluminescence immunosensor based on gold-magnetic nanoparticles and phage displayed antibodies. Sensors. 2016;16:308.
Sun X, Fei RH, Zhang L, Huo BY, Wang Y, Peng Y, et al. Bio–barcode triggered isothermal amplification in a fluorometric competitive immunoassay for the phytotoxin abrin. Microchim Acta. 2020;187:2456–63.
Reuven R, Paula D, Bradley H. CCD Based Detector for Detection of Abrin Toxin Activity. Toxins. 2020;12:120.
Hansbauer EM, Worbs S, Volland H, Simon S, Junot C, Fenaille F, et al. Rapid detection of abrin toxin and its isoforms in complex matrices by immuno-extraction and quantitative high resolution targeted mass spectrometry. Anal Chem. 2017;89:11719–27.
van Wezenbeek PM, Hulsebos TJ, Schoenmakers JG. Nucleotide sequence of the flamentous bacteriophage M13 DNA genome: Comparison with phage fd. Gene. 1980;11:129–48.
Rakonjac J, Bennett NJ, Spagnuolo J, Gagic D, Russel M. Filamentous bacteriophage: Biology, phage display and nano-technology applications. Curr Issues Mol Biol. 2011;13:51–76.
Li K, Chen Y, Li SQ, Nguyen HG, Niu ZW, You SJ, et al. Chemical Modification of M13 Bacteriophage and Its Application in Cancer Cell Imaging. Bioconjug Chem. 2010;21:1369–77.
Acknowledgments
This paper was supported by the National Key R&D Plan of China (2016YFF0103103).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of Interest
The authors declare no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 266 kb)
Rights and permissions
About this article
Cite this article
Liu, S., Gao, C., Tong, Z. et al. A highly sensitive electrochemiluminescence method for abrin detection by a portable biosensor based on a screen-printed electrode with a phage display affibody as specific labeled probe. Anal Bioanal Chem 414, 1095–1104 (2022). https://doi.org/10.1007/s00216-021-03735-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-021-03735-4