Abstract
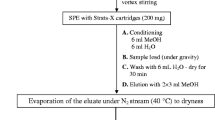
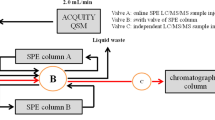
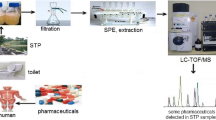
An automated method was developed using on-line solid-phase extraction (SPE) as a sample preparation step, coupled to liquid chromatography-tandem mass spectrometry (LC-MS/MS), for determination of pharmaceutical compounds in wastewater samples at nanograms per liter to micrograms per liter concentration levels. This method is suitable for use in routine analysis, especially in environmental applications, avoiding cross-contamination and requiring minimal sample handling. Results can be obtained rapidly, with a chromatographic run time of only 24 min (including sample preparation and chromatographic analysis). Using a 50 μL injection volume, the method was validated according to international guidelines, considering parameters included in terms of method detection (MDL) and quantification limit (MQL), linearity, inter-day and intra-day precisions, and matrix effects. Assessment of chromatographic efficiency considered peak resolution and asymmetry, and carryover was evaluated to ensure analytical reliability and the ability to reuse the SPE cartridge. The intra- and inter-day precisions were lower than 10 and 17%, respectively. The MDL values ranged from 1×10-6 to 1 μg L-1, while the MQL values were from 0.001 to 3 μg L-1. Matrix effects were minimized by isotope dilution calibration. Application of the method to 20 wastewater samples showed that caffeine was the most frequently detected compound, with the highest concentration of 715 μg L-1, while other pharmaceutical compounds were detected in fewer samples and at lower concentrations (up to 8.51 μg L-1).
Graphical abstract






Similar content being viewed by others
References
Chinnaiyan P, Thampi SG, Kumar M, Mini KM. Pharmaceutical products as emerging contaminant in water: relevance for developing nations and identification of critical compounds for Indian environment. Environ Monit Assess. 2018;190:288.
Mohapatra DP, Kirpalani DM. Advancement in treatment of wastewater: fate of emerging contaminants. Can J Chem Eng. 2019;97:2621–31.
Marasco Júnior CA, Luchiari NDC, Lima Gomes PCF. Occurrence of caffeine in wastewater and sewage and applied techniques for analysis: a review. Eclética Quím J. 2019;44:11–26.
U.S. GAO Government Accountability Office Toxic Chemicals - High Risk Issue. https://www.gao.gov/key_issues/toxic_chemicals/issue_summary. Accessed on March 16, 2020.
Mattarozzi M, Careri M. Liquid chromatography/mass spectrometry in environmental analysis. In: Meyers RA, editor. Encyclopedia of analytical chemistry. Chichester, UK: John Wiley & Sons, Ltd; 2015. p. 1–41.
Loos G, Van Schepdael A, Cabooter D. Quantitative mass spectrometry methods for pharmaceutical analysis. Philos Trans R Soc A Math Phys Eng Sci. 2016;374:20150366.
Pérez-Fernández V, Mainero Rocca L, Tomai P, Fanali S, Gentili A. Recent advancements and future trends in environmental analysis: sample preparation, liquid chromatography and mass spectrometry. Anal Chim Acta. 2017;983:9–41.
Silva CGA, Collins CH. Aplicações de cromatografia líquida de alta eficiência para o estudo de poluentes orgânicos emergentes. Quím Nova. 2011;34:665–76.
Pavlović DM, Babić S, Horvat AJM, Kaštelan-Macan M. Sample preparation in analysis of pharmaceuticals. TrAC Trends Anal Chem. 2007;26:1062–75.
Zhong M, Wang T, Qi C, Peng G, Lu M, Huang J, et al. Automated online solid-phase extraction liquid chromatography tandem mass spectrometry investigation for simultaneous quantification of per- and polyfluoroalkyl substances, pharmaceuticals and personal care products, and organophosphorus flame retardants in environmental waters. J Chromatogr A. 1602;2019:350–8.
Montesdeoca-Esponda S, Sosa-Ferrera Z, Santana-Rodríguez JJ. On-line solid-phase extraction coupled to ultra-performance liquid chromatography with tandem mass spectrometry detection for the determination of benzotriazole UV stabilizers in coastal marine and wastewater samples. Anal Bioanal Chem. 2012;403:867–76.
Lima Gomes PCF, Tomita IN, Santos-Neto AJ, Zaiat M. Rapid determination of 12 antibiotics and caffeine in sewage and bioreactor effluent by online column-switching liquid chromatography/tandem mass spectrometry. Anal Bioanal Chem. 2015;407:8787–801.
Andrade-Eiroa A, Canle M, Leroy-Cancellieri V, Cerdà V. Solid-phase extraction of organic compounds: a critical review (Part I). TrAC Trends Anal Chem. 2016;80:641–54.
Buszewski B, Szultka M. Past, present, and future of solid phase extraction: a review. Crit Rev Anal Chem. 2012;42:198–213.
Valsecchi S, Polesello S, Mazzoni M, Rusconi M, Petrovic M. On-line sample extraction and purification for the LC–MS determination of emerging contaminants in environmental samples. Trends Environ Anal Chem. 2015;8:27–37.
Fan X, Gao J, Li W, Huang J, Yu G. Determination of 27 pharmaceuticals and personal care products (PPCPs) in water: the benefit of isotope dilution. Front Environ Sci Eng. 2020;14:8.
Huntscha S, Singer HP, McArdell CS, Frank CE, Hollender J. Multiresidue analysis of 88 polar organic micropollutants in ground, surface and wastewater using online mixed-bed multilayer solid-phase extraction coupled to high performance liquid chromatography–tandem mass spectrometry. J Chromatogr A. 2012;1268:74–83.
Petrie B, McAdam EJ, Scrimshaw MD, Lester JN, Cartmell E. Fate of drugs during wastewater treatment. TrAC Trends Anal Chem. 2013;49:145–59.
Gros M, Pizzolato T-M, Petrović M, Alda MJL, Barceló D. Trace level determination of β-blockers in waste waters by highly selective molecularly imprinted polymers extraction followed by liquid chromatography–quadrupole-linear ion trap mass spectrometry. J Chromatogr A. 2008;1189:374–84.
Matuszewski BK, Constanzer ML, Chavez-Eng CM. Strategies for the assessment of matrix effect in quantitative bioanalytical methods based on HPLC−MS/MS. Anal Chem. 2003;75:3019–30.
Stuber M, Reemtsma T. Evaluation of three calibration methods to compensate matrix effects in environmental analysis with LC-ESI-MS. Anal Bioanal Chem. 2004;378:910–6.
Azzouz A, Souhail B, Ballesteros E. Continuous solid-phase extraction and gas chromatography–mass spectrometry determination of pharmaceuticals and hormones in water samples. J Chromatogr A. 2010;1217(17):2956–63.
Fontanals N, Marcé RM, Borrull F. New hydrophilic materials for solid-phase extraction. TrAC, Trends Anal. Chem. 2005;24(5):394–406.
Buerge IJ, Poiger T, Müller MD, Buser H-R. Caffeine, an anthropogenic marker for wastewater contamination of surface waters. Environ Sci Technol. 2003;37:691–700.
Hai F, Yang S, Asif M, Sencadas V, Shawkat S, Sanderson-Smith M, et al. Carbamazepine as a possible anthropogenic marker in water: occurrences, toxicological effects, regulations and removal by wastewater treatment technologies. Water. 2018;10(2):107.
Millar NL, Siebert S, McInnes IB. Europe rules on harm from fluoroquinolone antibiotics. Nature. 2019;566:1.
ClinCalc, The top 300 of 2020. URL https://clincalc.com/DrugStats/Top300Drugs.aspx. Accessed on August 4, 2020.
Haddad PR. In: Meyers, R. A. (Ed.), Column theory and resolution in liquid chromatography, Encyclopedia of analytical chemistry. John Wiley & Sons, Ltd, Chichester, UK 2009, pp. 1-9.
da Silva BF, Souza JC, Zanoni MVB. Corantes: Caracterização Química, Toxicológica, Métodos de Detecção e Tratamento. São Paulo: Cultura Acadêmica; 2016. p. 347.
Gómez-Ramos MDM, Rajski Ł, Lozano A, Fernández-Alba AR. The evaluation of matrix effects in pesticide multi-residue methods via matrix fingerprinting using liquid chromatography electrospray high-resolution mass spectrometry. Anal Methods. 2016;8:4664–73.
Martins GS, Luchiari NC, Lamarca RS, Silva BF, Gomes PCFL. Removal of sulfamethoxazol and trimethoprim using horizontal-flow anaerobic immobilized bioreactor. Sci Chromatogr. 2017;9:253–64.
Dethy J-M, Ackermann BL, Delatour C, Henion JD, Schultz GA. Demonstration of direct bioanalysis of drugs in plasma using nanoelectrospray infusion from a silicon chip coupled with tandem mass spectrometry. Anal Chem. 2003;75:805–11.
Clouser-Roche A, Johnson K, Fast D, Tang D. Beyond pass/fail: a procedure for evaluating the effect of carryover in bioanalytical LC/MS/MS methods. J Pharm Biomed Anal. 2008;47:146–55.
ANVISA Agência Nacional de Vigilância Sanitária, RESOLUÇÃO DA DIRETORIA COLEGIADA - RDC No 166, DE 24 DE JULHO DE 2017. Guia para validação de métodos analíticos - Julho, 2017. http://www.in.gov.br/materia/-/asset_publisher/Kujrw0TZC2Mb/content/id/19194581/do1-2017-07-25-resolucao-rdc-n-166-de-24-de-julho-de-2017-19194412 (in Portuguese).
INMETRO Instituto Nacional de Metrologia, Normalização e Qualidade Industrial, Orientação Sobre Validação de Métodos Analíticos - DOC - CGCRE - 008. 2018. http://www.inmetro.gov.br/Sidoq/Arquivos/CGCRE/DOQ/DOQ-CGCRE-8_07.pdf (in Portuguese).
IUPAC International Union of Pure and Applied Chemistry. Harmonized guidelines for single- laboratory validation of methods of analysis. 2002;74:835–55 http://publications.iupac.org/pac/2002/pdf/7405x0835.pdf.
FDA - Food and Drug Administration, Analytical procedures and methods validation for drugs and biologics. 2015. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/analytical-procedures-and-methods-validation-drugs-and-biologics
Choi DY, Row KH. Theoretical analysis of chromatographic peak asymmetry and sharpness by the moment method using two peptides. Biotechnol Bioprocess Eng. 2004;9:495–9.
CDER Center for Drug Evaluation and Research, Reviewer guidance: validation of chromatographic methods. 1994. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/reviewer-guidance-validation-chromatographic-methods.
Shabir GA. Validation of high-performance liquid chromatography methods for pharmaceutical analysis. Understanding the differences and similarities between validation requirements of the US Food and Drug Administration, the US Pharmacopeia and the International Conference on Harmonization. J Chromatogr A. 2003;978:57–66.
López-Serna R, Pérez S, Ginebreda A, Petrović M, Barceló D. Fully automated determination of 74 pharmaceuticals in environmental and waste waters by online solid phase extraction–liquid chromatography-electrospray–tandem mass spectrometry. Talanta. 2010;83:410–24.
Gros M, Rodríguez-Mozaz S, Barceló D. Rapid analysis of multiclass antibiotic residues and some of their metabolites in hospital, urban wastewater and river water by ultra-high-performance liquid chromatography coupled to quadrupole-linear ion trap tandem mass spectrometry. J Chromatogr A. 2013;1292:173–88.
Heuett NV, Ramirez CE, Fernandez A, Gardinali PR. Analysis of drugs of abuse by online SPE-LC high resolution mass spectrometry: communal assessment of consumption. Sci Total Environ. 2015;511:319–30.
Gracia-Lor E, Sancho JV, Hernández F. Multi-class determination of around 50 pharmaceuticals, including 26 antibiotics, in environmental and wastewater samples by ultra-high performance liquid chromatography–tandem mass spectrometry. J Chromatogr A. 2011;1218:2264–75.
EC - European Commission Document n° SANTE/11813/2017, Guidance document on analytical quality control and method validation procedures for pesticide residues analysis in food and feed. https://www.eurl-pesticides.eu/docs/public/tmplt_article.asp?CntID=727.
Axel M, Ewelina K, Jenny-Maria B, Leif K. An online SPE LC-MS/MS method for the analysis of antibiotics in environmental water. Environ Sci Pollut Res. 2017;24:8692–9.
Trenholm RA, Vanderford BJ, Snyder SA. On-line solid phase extraction LC–MS/MS analysis of pharmaceutical indicators in water: a green alternative to conventional methods. Talanta. 2017;79(5):1425–32.
Scheurer M, et al. The challenge of analyzing beta-blocker drugs in sludge and wastewater. Anal Bioanal Chem. 2010;396(2):845–56.
Milhome MAL, Vieira SKV, Reges BM, Fernandes DR, Uchoa MLP, Pinheiro AI, et al. Multiresidue analysis and evaluation of the matrix effect on 20 pesticides in Brazilian maize (Zea mays L.) flour. J. Environ. Sci. Health Part B. 2019;54:892–7.
Gonçalves ES, Rodrigues SV, Silva-Filho EV. The use of caffeine as a chemical marker of domestic wastewater contamination in surface waters: seasonal and spatial variations in Teresópolis, Brazil. Ambiente E Agua - Interdiscip J Appl Sci. 2017;12:192.
Gros M, Petrović M, Barceló D. Development of a multi-residue analytical methodology based on liquid chromatography–tandem mass spectrometry (LC–MS/MS) for screening and trace level determination of pharmaceuticals in surface and wastewaters. Talanta. 2006;70:678–90.
Pedrouzo M, Reverté S, Borrull F, Pocurull E, Marcé RM. Pharmaceutical determination in surface and wastewaters using high-performance liquid chromatography-(electrospray)-mass spectrometry. J Sep Sci. 2007;30:297–303.
Biel-Maeso M, Baena-Nogueras RM, Corada-Fernández C, Lara-Martín PA. Occurrence, distribution and environmental risk of pharmaceutically active compounds (PhACs) in coastal and ocean waters from the Gulf of Cadiz (SW Spain). Sci Total Environ. 2018;612:649–59.
Campanha MB, Awan AT, de Sousa DNR, Grosseli GM, Mozeto AA, Fadini PS. A 3-year study on occurrence of emerging contaminants in an urban stream of São Paulo State of Southeast Brazil. Environ Sci Pollut Res. 2015;22:7936–47.
Chen H, Jing L, Teng Y, Wang J. Characterization of antibiotics in a large-scale river system of China: occurrence pattern, spatiotemporal distribution and environmental risks. Sci Total Environ. 2018;618:409–18.
Paíga P, Santos LHMLM, Ramos S, Jorge S, Silva JG, Delerue-Matos C. Presence of pharmaceuticals in the Lis river (Portugal): sources, fate and seasonal variation. Sci Total Environ. 2016;573:164–77.
Gusmaroli L, Insa S, Petrovic M. Development of an online SPE-UHPLC-MS/MS method for the multiresidue analysis of the 17 compounds from the EU “Watch list”. Anal Bioanal Chem. 2018;410:4165–76.
Ferrer-Aguirre A, Romero-González R, Martínez Vidal JL, Frenich AG. Simple and quick determination of analgesics and other contaminants of emerging concern in environmental waters by on-line solid phase extraction coupled to liquid chromatography–tandem mass spectrometry. J Chromatogr A. 2016;1446:27–33.
Chaves MJS, Barbosa SC, Malinowski MM, Volpato D, Castro IB, Franco TCRS, et al. Pharmaceuticals and personal care products in a Brazilian wetland of international importance: occurrence and environmental risk assessment. Sci Total Environ. 2020;734:139374.
de Almeida CAA, Oliveira MS, Mallmann CA, Martins AF. Determination of the psychoactive drugs carbamazepine and diazepam in hospital effluent and identification of their metabolites. Environ Sci Pollut Res. 2015;22:17192–201.
Funding
The authors received funding from Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP, grants 16/03369-3, 18/11700-7, 18/22393-8, and INCT-DATREM 14/50945-4), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, grant 465571/2014-0), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES, 88887136426/2017/00), and Instituto de Química – Universidade Estadual Paulista (IQ/UNESP).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 120 kb)
Rights and permissions
About this article
Cite this article
Marasco Júnior, C.A., da Silva, B.F., Lamarca, R.S. et al. Automated method to determine pharmaceutical compounds in wastewater using on-line solid-phase extraction coupled to LC-MS/MS. Anal Bioanal Chem 413, 5147–5160 (2021). https://doi.org/10.1007/s00216-021-03481-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-021-03481-7