Abstract
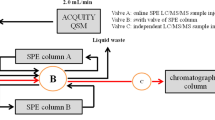
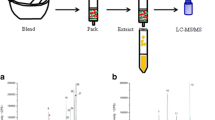
The current study presents a single step sample preparation procedure for the simultaneous determination of five antihypertensive (propranolol, losartan, irbesartan, telmisartan, and valsartan), three antiarrhythmic drugs (flecainide, dronedarone, and amiodarone), and one of their metabolites (N-desethylamiodarone) in sludge from municipal sewage treatment plants (STPs). Matrix solid-phase dispersion (MSPD) and ultra-performance liquid chromatography (UPLC) with tandem mass spectrometry (MS/MS) detection were selected as sample preparation and determination techniques, respectively. Under optimal conditions, MSPD extractions were carried out with freeze-dried samples (0.5 g) dispersed on 2 g of C18. Exhaustive extraction of target compounds was achieved with 10 mL of a methanol/acetonitrile/formic acid (30:69:1) solution. The obtained extract was ready for UPLC-MS/MS analysis without any further treatment, except filtration. The overall recoveries of the method (calculated against solvent-based standards) varied from 82 to 124%, with standard deviations in the range from 2 to 16%. Thus, the method was free of matrix effects during electrospray ionization. The achieved limits of quantification stayed between 2 and 10 ng g−1, and the linear response range extended to 5000 ng g−1. The occurrence of target compounds was investigated in sludge from 14 different STPs. High detection frequencies were observed for all compounds, with average concentrations above 100 ng g−1 for six cardiovascular drugs.

Similar content being viewed by others
References
Montero Corominas D, García del Pozo J, De Abajo Iglesias FJ. Utilización de antihipertensivos en España (1992-2006). 2009. https://www.aemps.gob.es/medicamentosUsoHumano/observatorio/docs/antihipertensivos.pdf. Accessed 2 May 2018.
Ortiz de García S, Pinto Pinto G, García Encina P, Irusta Mata R. Consumption and occurrence of pharmaceutical and personal care products in the aquatic environment in Spain. Sci Total Environ. 2013;444:451–65.
Stankiewicz A, Giebułtowicz J, Stankiewicz U, Wroczyński P, Nałecz-Jawecki G. Determination of selected cardiovascular active compounds in environmental aquatic samples—methods and results, a review of global publications from the last 10 years. Chemosphere. 2015;138:642–56.
Bueno MJM, Gomez MJ, Herrera S, Hernando MD, Agüera A, Fernández-Alba AR. Occurrence and persistence of organic emerging contaminants and priority pollutants in five sewage treatment plants of Spain: two years pilot survey monitoring. Environ Pollut. 2012;164:267–73.
Giebułtowicz J, Nałecz-Jawecki G. Occurrence of cardiovascular drugs in the sewage-impacted Vistula River and in tap water in the Warsaw region (Poland). Environ Sci Pollut Res. 2016;23:24337–49.
Bayer A, Asner R, Schüssle W, Kopf W, Weiß K, Seng M, et al. Behavior of sartans (antihypertensive drugs) in wastewater treatment plants, their occurrence and risk for the aquatic environment. Environ Sci Pollut Res. 2014;21:10830–9.
Gurke R, Robler M, Marx C, Diamond S, Schubert S, Oertel R, et al. Occurrence and removal of frequently prescribed pharmaceuticals and corresponding metabolites in wastewater of a sewage treatment plant. Sci Total Environ. 2015;532:762–70.
Huerta-Fontela M, Galcerán MT, Ventura F. Occurrence and removal of pharmaceuticals and hormones through drinking water treatment. Water Res. 2010;45:1432–42.
Godoy AA, Kummrow F, Pamplin PAZ. Occurrence, ecotoxicological effects and risk assessment of antihypertensive pharmaceutical residues in the aquatic environment-a review. Chemosphere. 2015;138:281–91.
Grabic R, Fick J, Lindberg RH, Fedorova G, Tysklind M. Multi-residue method for trace level determination of pharmaceuticals in environmental samples using liquid chromatography coupled to triple quadrupole mass spectrometry. Talanta. 2012;100:183–95.
Hörsing M, Ledin A, Grabic R, Fick J, Tysklind M, Jansen JC, et al. Determination of sorption of seventy-five pharmaceuticals in sewage sludge. Water Res. 2011;45:4470–82.
Berthod L, Whitley DC, Roberts G, Sharpe A, Greenwood R, Mills GA. Quantitative structure-property relationships for predicting sorption of pharmaceuticals to sewage sludge during waste water treatment processes. Sci Total Environ. 2017;579:1512–20.
Lajeunesse A, Smyth SA, Barclay K, Sauvé S, Gagnon C. Distribution of antidepressant residues in wastewater and biosolids following different treatment processes by municipal wastewater treatment plants in Canada. Water Res. 2012;46:5600–12.
González-Mariño I, Rodríguez I, Quintana JB, Cela R. Matrix solid-phase dispersion followed by gas chromatography-mass spectrometry for the determination of triclosan and methyl triclosan in sludge and sediments. Anal Bioanal Chem. 2010;398:2289–97.
Ivanová L, Mackulak T, Grabic R, Golovko O, Koba O, Stanová AV, et al. Pharmaceuticals and illicit drugs—a new threat to the application of sewabe sludge in agriculture. Sci Total Environ. 2018;634:606–15.
Boix C, Ibáñez M, Fabregat-Safont D, Morales E, Pastor L, Sancho JV, et al. Behaviour of emerging contaminants in sewage sludge after anaerobic digestion. Chemosphere. 2016;163:296–304.
Kuhn J, Götting C, Kleesiek K. Simultaneous measurement of amiodarone and desethylamiodarone in human plasma and serum by stable isotope dilution liquid chromatography-tandem mass spectrometry assay. J Pharm Biomed Anal. 2010;51:210–6.
Montes R, Rodríguez I, Casado J, López-Sabater MC, Cela R. Determination of the cardiac drug amiodarone and its N-desethyl metabolite in sludge samples. J Chromatogr A. 2015;1394:62–70.
Celano R, Rodríguez I, Cela R, Rastrelli L, Piccinelli AL. Liquid chromatography quadrupole time-of-flight mass spectrometry quantification and screening of organophosphate compounds in sludge. Talanta. 2014;118:312–20.
Li M, Sun Q, Li Y, Lv M, Lin L, Wu Y, et al. Simultaneous analysis of 45 pharmaceuticals and personal care products in sludge by matrix solid-phase dispersion and liquid chromatography tandem mass spectrometry. Anal Bioanal Chem. 2016;408:4953–64.
Wasik A, Kot-Wasik A, Namiesnik J. New trends in sample preparation techniques for the analysis of the residues of pharmaceuticals in environmental samples. Curr Anal Chem. 2016;12:280–302.
Baker SA. Matrix solid phase dispersion. J Biochem Biophys Methods. 2007;70:151–62.
Capriotti AL, Cavaliere C, Foglia P, Samperi R, Stampachiacchiere S, Ventura S, et al. Recent advances and developments in matrix solid-phase dispersion. TrAC Trends Anal Chem. 2015;71:186–93.
Kasprzyk-Hordern B, Dinsdale RM, Guwy AJ. The effect of signal suppression and mobile phase composition on the simultaneous analysis of multiple classes of acidic/neutral pharmaceuticals and personal care products in surface water by solid-phase extraction and ultra performance liquid chromatography. Talanta. 2008;74:1299–312.
Acknowledgements
This study has been financially supported by Xunta de Galicia and the Spanish Government (projects GRC-ED431C 2017/36 and CTQ2015-68660-P). I.C. acknowledges a postdoctoral grant to Xunta de Galicia. We also thank Indrops laboratories for providing most of the sludge samples used in this research.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
ESM 1
(PDF 707 kb)
Rights and permissions
About this article
Cite this article
Castro, G., Carpinteiro, I., Rodríguez, I. et al. Determination of cardiovascular drugs in sewage sludge by matrix solid-phase dispersion and ultra-performance liquid chromatography tandem mass spectrometry. Anal Bioanal Chem 410, 6807–6817 (2018). https://doi.org/10.1007/s00216-018-1268-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-018-1268-3