Abstract
Six automated, headspace-based sample preparation techniques were used to extract volatile analytes from water with the goal of establishing a systematic comparison between commonly available instrumental alternatives. To that end, these six techniques were used in conjunction with the same gas chromatography instrument for analysis of a common set of volatile organic carbon (VOC) analytes. The methods were thereby divided into three classes: static sampling (by syringe or loop), static enrichment (SPME and PAL SPME Arrow), and dynamic enrichment (ITEX and trap sampling). For PAL SPME Arrow, different sorption phase materials were also included in the evaluation. To enable an effective comparison, method detection limits (MDLs), relative standard deviations (RSDs), and extraction yields were determined and are discussed for all techniques. While static sampling techniques exhibited sufficient extraction yields (approx. 10–20 %) to be reliably used down to approx. 100 ng L−1, enrichment techniques displayed extraction yields of up to 80 %, resulting in MDLs down to the picogram per liter range. RSDs for all techniques were below 27 %. The choice on one of the different instrumental modes of operation (aforementioned classes) was thereby the most influential parameter in terms of extraction yields and MDLs. Individual methods inside each class showed smaller deviations, and the least influences were observed when evaluating different sorption phase materials for the individual enrichment techniques. The option of selecting specialized sorption phase materials may, however, be more important when analyzing analytes with different properties such as high polarity or the capability of specific molecular interactions.

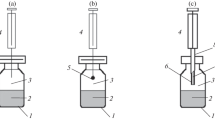
PAL SPME Arrow during the extraction of volatile analytes from the headspace of an aqueous sample



Similar content being viewed by others
References
Aguinaga N, Campillo N, Viñas P, Hernández-Córdoba M. A headspace solid-phase microextraction procedure coupled with gas chromatography–mass spectrometry for the analysis of volatile polycyclic aromatic hydrocarbons in milk samples. Anal Bioanal Chem. 2008;391(3):753–8.
Kolb B, Ettre LS. Static headspace-gas chromatography: theory and practice. New York: Wiley-VCH; 1997.
Pawliszyn J, Zhang Z. Headspace solid-phase microextraction. Anal Chem. 1993;65:1843–52.
Curvers J, Noy T, Cramers C, Rijks J. Possibilities and limitations of dynamic headspace sampling as a pre-concentration technique for trace analysis of organics by capillary gas chromatography. J Chromatogr A. 1984;289:171–82.
Reinert KH, Hunter JV, Sabatino T. Dynamic heated headspace analyses of volatile organic compounds present in fish tissue samples. J Agric Food Chem. 1983;31(5):1057–60.
Van Durme J, Demeestere K, Dewulf J, et al. Accelerated solid-phase dynamic extraction of toluene from air. J Chromatogr A. 2007;1175(2):145–53.
Bigus P, Tsakovski S, Simeonov V, Namieśnik J, Tobiszewski M. Hasse diagram as a green analytical metrics tool: ranking of methods for benzo[a]pyrene determination in sediments. Anal Bioanal Chem. 2016;408(14):3833–41.
Flórez Menéndez JC, Fernández Sánchez ML, Sánchez Urı́a JE, Fernández Martı́nez E, Sanz-Medel A. Static headspace, solid-phase microextraction and headspace solid-phase microextraction for BTEX determination in aqueous samples by gas chromatography. Anal Chim Acta. 2000;415(1–2):9–20.
Miller ME, Stuart JD. Comparison of gas-sampled and SPME-sampled static headspace for the determination of volatile flavor components. Anal Chem. 1999;71(1):23–7.
Povolo M, Contarini G. Comparison of solid-phase microextraction and purge-and-trap methods for the analysis of the volatile fraction of butter. J Chromatogr A. 2003;985(1-2):117–25.
Schulz K, Dreßler J, Sohnius E-M, Lachenmeier DW. Determination of volatile constituents in spirits using headspace trap technology. J Chromatogr A. 2007;1145(1–2):204–9.
Westland JL, Dorman FL. Comparison of SPME and static headspace analysis of blood alcohol concentration utilizing two novel chromatographic stationary phases. Forensic Sci Int. 2013;231(1–3):E50–6.
Spietelun A, Kloskowski A, Chrzanowski W, Namieśnik J. Understanding solid-phase microextraction: key factors influencing the extraction process and trends in improving the technique. Chem Rev. 2012;113(3):1667–85.
Kremser A, Jochmann MA, Schmidt TC. PAL SPME arrow—evaluation of a novel solid-phase microextraction device for freely dissolved PAHs in water. Anal Bioanal Chem. 2016;408(3):943–52.
Jochmann MA, Yuan X, Schilling B, Schmidt TC. In-tube extraction for enrichment of volatile organic hydrocarbons from aqueous samples. J Chromatogr A. 2008;1179(2):96–105.
Laaks J, Jochmann MA, Schilling B, Schmidt TC. In-tube extraction of volatile organic compounds from aqueous samples: an economical alternative to purge and trap enrichment. Anal Chem. 2010;82(18):7641–8.
Laaks J, Jochmann MA, Schilling B, Schmidt TC. Optimization strategies of in-tube extraction (ITEX) methods. Anal Bioanal Chem. 2015;407(22):6827–38.
Endo S, Brown TN, Goss K-U. General model for estimating partition coefficients to organisms and their tissues using the biological compositions and polyparameter linear free energy relationships. Environ Sci Technol. 2013;47(12):6630–9.
Endo S, Schmidt TC. Prediction of partitioning between complex organic mixtures and water: application of polyparameter linear free energy relationships. Environ Sci Technol. 2006;40(2):536–45.
Sprunger L, Proctor A, Acree WL, Abraham MH. Characterization of the sorption of gaseous and organic solutes onto polydimethyl siloxane solid-phase microextraction surfaces using the Abraham model. J Chromatogr A. 2007;1175:162–73.
Zimmermann T, Ensinger WJ, Schmidt TC. Depletion solid-phase microextraction for the evaluation of fiber-sample partition coefficients of pesticides. J Chromatogr A. 2006;1102(1–2):51–9.
Lamani X, Horst S, Zimmermann T, Schmidt TC. Determination of aromatic amines in human urine using comprehensive multi-dimensional gas chromatography mass spectrometry (GCxGC-qMS). Anal Bioanal Chem. 2015;407(1):241–52.
Ridgway K, Lalljie SPD, Smith RM. Use of in-tube sorptive extraction techniques for determination of benzene, toluene, ethylbenzene and xylenes in soft drinks. J Chromatogr A. 2007;1174(1–2):20–6.
Namieśnik J, Zygmunt B, Jastrzebska A. Application of solid-phase microextraction for determination of organic vapours in gaseous matrices. J Chromatogr A. 2000;885(1–2):405–18.
Keith LH, Crummett W, Deegan Jr J, Libby RA, Taylor JK, Wentler G. Principles of environmental analysis. Anal Chem. 1983;55(14):2210–8.
Cavalcante RM, de Andrade MVF, Marins RV, Oliveira LDM. Development of a headspace-gas chromatography (HS-GC-PID-FID) method for the determination of VOCs in environmental aqueous matrices: Optimization, verification and elimination of matrix effect and VOC distribution on the Fortaleza Coast, Brazil. Microchem J. 2010;96(2):337–43.
DIN 32465:2008–11, Chemische Analytik - Nachweis-, Erfassungs- und Bestimmungsgrenze unter Wiederholbedingungen - Begriffe, Verfahren, Auswertung. 2008.
Arbeitskreis NA 119-01-03-02 AK. Validierungsdokument zur Norm DIN 38 407–41. Primäre Validierung genormter Verfahren zur Wasser-, Abwasser- und Schlammuntersuchung. 2011.
Barani F, Dell’Amico N, Griffone L, Santoro M, Tarabella C. Determination of volatile organic compounds by headspace trap. J Chromatogr Sci. 2006;44(10):625–30.
Lord HL, Zhan W, Pawliszyn J. Fundamentals and applications of needle trap devices: a critical review. Anal Chim Acta. 2010;677(1):3–18.
Alonso M, Cerdan L, Godayol A, Antico E, Sanchez JM. Headspace needle-trap analysis of priority volatile organic compounds from aqueous samples: application to the analysis of natural and waste waters. J Chromatogr A. 2011;1218(45):8131–9.
Laaks J. Development and validation of novel solventless microextraction techniques in gas chromatography. Ph.D. Thesis, University of Duisburg-Essen: Essen, Germany; 2013.
Wardencki W, Curyło J, Namieśnik J. Trends in solventless sample preparation techniques for environmental analysis. J Biochem Biophys Methods. 2007;70(2):275–88.
Jakubowska N, Polkowska Ż, Kujawski W, Konieczka P, Namieśnik J. A comparison of three solvent-free techniques coupled with gas chromatography for determining trihalomethanes in urine samples. Anal Bioanal Chem. 2007;388(3):691–8.
Meyer-Monath M, Beaumont J, Morel I, Rouget F, Tack K, Lestremau F. Analysis of BTEX and chlorinated solvents in meconium by headspace-solid-phase microextraction gas chromatography coupled with mass spectrometry. Anal Bioanal Chem. 2014;406(18):4481–90.
Jeleń HH, Wlazły K, Wa̧sowicz E, Kamiński E. Solid-phase microextraction for the analysis of some alcohols and esters in beer: comparison with static headspace method. J Agric Food Chem. 1998;46(4):1469–73.
Sriseadka T, Wongpornchai S, Kitsawatpaiboon P. Rapid method for quantitative analysis of the aroma impact compound, 2-acetyl-1-pyrroline, in fragrant rice using automated headspace gas chromatography. J Agric Food Chem. 2006;54(21):8183–9.
U.S. Pharmacopeial Convention. Method <467 > Residual Solvents. 2007.
Konieczka P, Namieśnik J. Estimating uncertainty in analytical procedures based on chromatographic techniques. J Chromatogr A. 2010;1217(6):882–91.
Acknowledgments
We gratefully acknowledge support by CTC Analytics AG concerning the autosamplers that were used in this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No conflict of interest influenced the work presented herein. The findings and conclusions in this report are those of the authors and do not represent the official views or positions of the supporting company.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 1156 kb)
Rights and permissions
About this article
Cite this article
Kremser, A., Jochmann, M.A. & Schmidt, T.C. Systematic comparison of static and dynamic headspace sampling techniques for gas chromatography. Anal Bioanal Chem 408, 6567–6579 (2016). https://doi.org/10.1007/s00216-016-9843-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-016-9843-y