Abstract
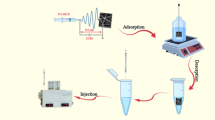
This study aimed to rapidly determine 13 representative sulfonamide (SA) residues in pork by using a surface-modified hydrophilic polystyrene sulfonic acid (PSSA) electrospun nanofibrous membrane as the solid-phase extraction (SPE) pretreatment sorbent, followed by ultra-performance liquid chromatography (UPLC) analysis. The highly hydrophilic nature of PSSA nanofibrous membrane created by vacuum plasma treatment was characterized using Fourier transform infrared spectroscopy (FT-IR), field emission scanning electron microscopy (FE-SEM), water contact angle, and X-ray photoelectron spectroscopy (XPS) measurements. In the pretreatment procedures, 13 SA standards, which were spiked in the fatty pork samples, were extracted, enriched, and purified by the SPE procedure based on the principle of ion exchange with the sulfonic groups on the PSSA chains. Under the optimized conditions, the calibration curves of 13 SA compounds showed good linearities with correlation coefficients (r) of more than 0.99 in the range of 50.0–200 μg kg−1. The mean recoveries of 13 SAs at the spiked concentrations of 50, 100, and 200 μg kg−1 were in the range of 70.3–92.5 % with average RSDs (n = 6) of less than 15 % (except for sulfacetamide, 56.9–61.6 %). Compared with other pretreatment methods reported previously, less organic solvent (especially without degreasing the extract with n-hexane) was used in this time-saving SPE procedure, which avoids the possibility of emulsification and therefore enhances the recoveries. The developed and validated analysis method was sensitive, accurate, rapid, convenient, environmentally friendly, and was successfully applied for the detection of 13 SA residues in commercially available pork samples.





Similar content being viewed by others
Abbreviations
- BE:
-
Binding energy
- DMF:
-
N,N-dimethylformamide
- EDTA:
-
Ethylenediaminetetraacetic acid
- FE-SEM:
-
Field emission scanning electron microscopy
- FT-IR:
-
Fourier transform infrared spectroscopy
- HLB:
-
Hydrophilic-lipophilic balance
- HPLC:
-
High-performance liquid chromatography
- LOD:
-
Limit of detection
- LOQ:
-
Limit of quantitation
- MCX:
-
Mixed-mode cation exchange
- MIP:
-
Molecularly imprinted polymer
- MRL:
-
Maximum residue limit
- MSPD:
-
Matrix solid-phase dispersion
- MWCNT:
-
Multi-walled carbon nanotube
- PP:
-
Polypropylene
- PS:
-
Polystyrene
- PSSA:
-
Polystyrene sulfonic acid
- QC:
-
Quality control
- SFA:
-
Sulfacetamide
- SA:
-
Sulfonamide
- SDM:
-
Sulfadimethoxine
- SIZ:
-
Sulfisoxazole
- SM1 :
-
Sulfamerazine
- SM2 :
-
Sulfamethazine
- SML:
-
Sulfabenzamide
- SMM:
-
Sulfamonomethoxine
- SMP:
-
Sulfamethoxypyridazine
- SMZ:
-
Sulfamethoxazole
- SP:
-
Sulfapyridine
- SPD:
-
Sulfachlorpyridazine
- SPE:
-
Solid-phase extraction
- SPP:
-
Sulfaphenazole
- SX:
-
Sulfamoxol
- THF:
-
Tetrahydrofuran
- UPLC:
-
Ultra-performance liquid chromatography
- XPS:
-
X-ray photoelectron spectroscopy
References
Kim K-R, Owens G, Kwon S-I, So K-H, Lee D-B, Ok YS. Occurrence and environmental fate of veterinary antibiotics in the terrestrial environment. Water, Air, Soil Pollut. 2011;214(1–4):163–74. doi:10.1007/s11270-010-0412-2.
Bialk-Bielinska A, Stolte S, Arning J, Uebers U, Boschen A, Stepnowski P, et al. Ecotoxicity evaluation of selected sulfonamides. Chemosphere. 2011;85(6):928–33. doi:10.1016/j.chemosphere.2011.06.058.
Baran W, Adamek E, Ziemianska J, Sobczak A. Effects of the presence of sulfonamides in the environment and their influence on human health. J Hazard Mater. 2011;196:1–15. doi:10.1016/j.jhazmat.2011.08.082.
Wang X, Li K, Shi D, Xiong N, Jin X, Yi J, et al. Development of an immunochromatographic lateral-flow test strip for rapid detection of sulfonamides in eggs and chicken muscles. J Agric Food Chem. 2007;55:2072–8. doi:10.1021/jf062523h.
Determination of sulfonamides residues in animal-derived food by high-performance liquid chromatographic method established by the Ministry of Agriculture of the People's Republic of China, GB 29694–2013.
Samanidou VF, Tolika EP, Papadoyannis IN. Chromatographic residue analysis of sulfonamides in foodstuffs of animal origin. Sep Purif Rev. 2008;37(4):325–71. doi:10.1080/15422110802177472.
Nebot C, Regal P, Martinez B, Miranda J, Cepeda A, Fente C. Confirmatory method for nine sulfonamides in miniature bovine muscle samples using HPLC/MS/MS without using SPE. J Food Drug Anal. 2010;18(3):191–201.
Hoff RB, Barreto F, Kist TB. Use of capillary electrophoresis with laser-induced fluorescence detection to screen and liquid chromatography-tandem mass spectrometry to confirm sulfonamide residues: validation according to European Union 2002/657/EC. J Chromatogr A. 2009;1216(46):8254–61. doi:10.1016/j.chroma.2009.07.074.
D’Orazio G, Rocchi S, Fanali S. Nano-liquid chromatography coupled with mass spectrometry: Separation of sulfonamides employing non-porous core–shell particles. J Chromatogr A. 2012;1255:277–85. doi:10.1016/j.chroma.2012.03.032.
Gamba V, Terzano C, Fioroni L, Moretti S, Dusi G, Galarini R. Development and validation of a confirmatory method for the determination of sulphonamides in milk by liquid chromatography with diode array detection. Anal Chim Acta. 2009;637(1–2):18–23. doi:10.1016/j.aca.2008.09.022.
Kishida K, Furusawa N. Matrix solid-phase dispersion extraction and high-performance liquid chromatographic determination of residual sulfonamides in chicken. J Chromatogr A. 2001;937:49–55.
Fang GZ, He JX, Wang S. Multiwalled carbon nanotubes as sorbent for on-line coupling of solid-phase extraction to high-performance liquid chromatography for simultaneous determination of 10 sulfonamides in eggs and pork. J Chromatogr A. 2006;1127(1–2):12–7. doi:10.1016/j.chroma.2006.06.024.
Gao R, Zhang J, He X, Chen L, Zhang Y. Selective extraction of sulfonamides from food by use of silica-coated molecularly imprinted polymer nanospheres. Anal Bioanal Chem. 2010;398(1):451–61. doi:10.1007/s00216-010-3909-z.
He J, Tang H, You L, Zhan H, Zhu J, Lu K. Fragment-imprinted microspheres for the extraction of sulfonamides. Microchim Acta. 2013;180(9–10):903–10. doi:10.1007/s00604-013-1011-x.
Karimi M, Aboufazeli F, Zhad HRLZ, Sadeghi O, Najafi E. Determination of sulfonamides in chicken meat by magnetic molecularly imprinted polymer coupled to HPLC–UV. Food Anal Methods. 2014;7(1):73–80. doi:10.1007/s12161-013-9600-1.
Wan Ibrahim WA, Abd Ali LI, Sulaiman A, Sanagi MM, Aboul-Enein HY. Application of solid-phase extraction for trace elements in environmental and biological samples: a review. Crit Rev Anal Chem. 2014;44(3):233–54. doi:10.1080/10408347.2013.855607.
Buszewski B, Szultka M. Past, present, and future of solid phase extraction: a review. Crit Rev Anal Chem. 2012;42(3):198–213. doi:10.1080/07373937.2011.645413.
Dmitrienko SG, Kochuk EV, Apyari VV, Tolmacheva VV, Zolotov YA. Recent advances in sample preparation techniques and methods of sulfonamides detection–a review. Anal Chim Acta. 2014;850:6–25. doi:10.1016/j.aca.2014.08.023.
http://www.waters.com/waters/en_US/Oasis-Sample-Extraction-Products
Dragan ES, editor. Advanced separations by specialized sorbents. Boca Raton: CRC; 2014. ISBN: 978-1-4822-2055-1.
Chigome S, Torto N. Electrospun nanofiber-based solid-phase extraction. TrAC, Trends Anal Chem. 2012;38:21–31. doi:10.1016/j.trac.2012.04.011.
Teo W-E, Inai R, Ramakrishna S. Technological advances in electrospinning of nanofibers. Sci Technol Adv Mater. 2011;12(1):013002. doi:10.1088/1468-6996/12/1/013002.
Kang X, Pan C, Xu Q, Yao Y, Wang Y, Qi D, et al. The investigation of electrospun polymer nanofibers as a solid-phase extraction sorbent for the determination of trazodone in human plasma. Anal Chim Acta. 2007;587(1):75–81. doi:10.1016/j.aca.2007.01.021.
Anyakora, Ifegwu C, Torto N, Chigome S. Application of nanofiber-packed SPE for determination of urinary 1-hydroxypyrene level using HPLC. Anal Chem Insights. 2014:17. doi:10.4137/aci.s13560.
Qi D, Kang X, Chen L, Zhang Y, Wei H, Gu Z. Electrospun polymer nanofibers as a solid-phase extraction sorbent for the determination of trace pollutants in environmental water. Anal Bioanal Chem. 2008;390(3):929–38. doi:10.1007/s00216-007-1747-4.
Hu WY, Kang XJ, Zhang C, Yang J, Ling R, Liu EH, et al. Packed-fiber solid-phase extraction coupled with high performance liquid chromatography-tandem mass spectrometry for determination of diethylstilbestrol, hexestrol, and dienestrol residues in milk products. J Chromatogr, B. 2014;957:7–13. doi:10.1016/j.jchromb.2014.02.036.
Ahmed FE, Lalia BS, Hashaikeh R. A review on electrospinning for membrane fabrication: challenges and applications. Desalination. 2015;356:15–30. doi:10.1016/j.desal.2014.09.033.
Xu Q, Zhang N, Yin X, Wang M, Shen Y, Xu S, et al. Development and validation of a nylon6 nanofibers mat-based SPE coupled with HPLC method for the determination of docetaxel in rabbit plasma and its application to the relative bioavailability study. J Chromatogr, B. 2010;878(26):2403–8. doi:10.1016/j.jchromb.2010.07.011.
Xu Q, Yin X, Wu S, Wang M, Wen Z, Gu Z. Determination of phthalate esters in water samples using Nylon6 nanofibers mat-based solid-phase extraction coupled to liquid chromatography. Microchim Acta. 2010;168(3–4):267–75. doi:10.1007/s00604-010-0290-8.
Cho E, Kim C, Park J-Y, Hwang CH, Kim JH, Kim YA, et al. Surface modification of electrospun polyvinylidene fluoride nanofiber membrane by plasma treatment for protein detection. J Nanosci Nanotechnol. 2013;13(1):674–7. doi:10.1166/jnn.2013.6925.
Yan D, Jones J, Yuan XY, Xu XH, Sheng J, Lee JC, et al. Plasma treatment of electrospun PCL random nanofiber meshes (NFMs) for biological property improvement. J Biomed Mater Res A. 2013;101(4):963–72. doi:10.1002/jbm.a.34398.
Kim JH, Chainey M, Elaasser MS, Vanerhoff JW. Preparation of highly sulfonated polystyrene model colloids. J Poly Sci A. 1989;27(10):3187–99. doi:10.1002/pola.1989.080271001.
Sunkara HB, Jethmalani JM, Ford WT. Synthesis of cross-linked of poly(styrene-co-sodium styrenesulfonate) latexes. J Poly Sci A. 1994;32(8):1431–5. doi:10.1002/pola.1994.080320804.
Chen R, Yang Y, Wang N, Hao L, Li L, Guo X, et al. Application of packed porous nanofibers-solid-phase extraction for the detection of sulfonamide residues from environmental water samples by ultra high performance liquid chromatography with mass spectrometry. J Sep Sci. 2015;38(5):749–56. doi:10.1002/jssc.201400982.
Tiwari A, Terada D, Yoshikawa C, Kobayashi H. An enzyme-free highly glucose-specific assay using self-assembled aminobenzene boronic acid upon polyelectrolytes electrospun nanofibers-mat. Talanta. 2010;82:1725–32. doi:10.1016/j.talanta.2010.07.078.
Safinia L, Wilson K, Mantalaris A, Bismarck A. Through-thickness plasma modification of biodegradable and nonbiodegradable porous polymer constructs. J Biomed Mater Res A. 2008;87(3):632–42. doi:10.1002/jbm.a.31731.
Jönsson SKM, Salaneck WR, Fahlman M. X-ray photoelectron spectroscopy study of the metal/polymer contacts involving aluminum and poly(3,4–ethylenedioxythiophene) – poly(styrenesulfonic acid) derivatives. J Mater Res. 2003;18(5):1219–26. doi:10.1557/JMR.2003.0167.
Acknowledgements
The project was sponsored by the Jiangsu Province Science Foundation for Youths (BK20130644) and the Fundamental Research Funds for the Central Universities (JKQZ2013006). Dr. Rong Chen would also like to acknowledge the China Scholarship Council (CSC 201407060048) for granting a scholarship that enabled her to pursue this work at National University of Singapore, Singapore.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Chen, R., Yang, Y., Qu, B. et al. Rapid determination of sulfonamide residues in pork by surface-modified hydrophilic electrospun nanofibrous membrane solid-phase extraction combined with ultra-performance liquid chromatography. Anal Bioanal Chem 408, 5499–5511 (2016). https://doi.org/10.1007/s00216-016-9648-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-016-9648-z