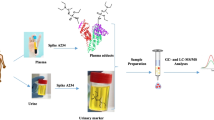
Abstract
Organophosphorus nerve agent (OPNA) adducts to butyrylcholinesterase (BChE) can be used to confirm exposure in humans. A highly accurate method to detect G- and V-series OPNA adducts to BChE in 75 μL of filtered blood, serum, or plasma has been developed using immunomagnetic separation (IMS) coupled with liquid chromatography tandem mass spectrometry (LC-MS/MS). The reported IMS method captures > 88 % of the BChE in a specimen and corrects for matrix effects on peptide calibrators. The optimized method has been used to quantify baseline BChE levels (unadducted and OPNA-adducted) in a matched-set of serum, plasma, and whole blood (later processed in-house for plasma content) from 192 unexposed individuals to determine the interchangeability of the tested matrices. The results of these measurements demonstrate the ability to accurately measure BChE regardless of the format of the blood specimen received. Criteria for accepting or denying specimens were established through a series of sample stability and processing experiments. The results of these efforts are an optimized and rugged method that is transferrable to other laboratories and an increased understanding of the BChE biomarker in matrix.


Similar content being viewed by others
Abbreviations
- A:
-
Alanine
- Ab:
-
Antibody
- AChE:
-
Acetylcholinesterase
- BChE:
-
Butyrylcholinesterase
- ChE:
-
Cholinesterase
- CLSI:
-
Clinical and Laboratory Standards Institute
- E:
-
Glutamic acid
- F:
-
Phenylalanine
- G:
-
Glycine
- GB:
-
Sarin
- IMS:
-
Immunomagnetic separation
- LC-MS/MS:
-
Liquid chromatography tandem mass spectrometry
- LOD:
-
Limit of detection
- LOQ:
-
Limit of quantitation
- LRL:
-
Lowest reportable limit
- MeP:
-
Methylphosphonic acid
- MRM:
-
Multiple reaction monitoring
- OP:
-
Organophosphorus
- OPNA:
-
Organophosphorus nerve agents
- QC:
-
Quality control
- QCH:
-
Quality control high
- QCL:
-
Quality control low
- RSD:
-
Relative standard deviation
- S:
-
Serine
- TNO:
-
The Netherlands Organisation for Applied Scientific Research
References
Convention of the prohibition of the development, production, stockpiling, and use of chemical weapons and their destruction (2005) Organisation for the Prohibition of Chemical Weapons, Netherlands. http://www.opcw.org/chemical-weapons-convention/annex-on-chemicals/b-schedules-of-chemicals/schedule-1. Assessed 16 August 2013
Nicolet Y, Lockridge O, Masson P, Fontecilla-Camps JC, Nachon F (2003) J Biol Chem 278:41141–41147
Grob D, Harvey JC (1957) J Clin Invest 37:350–368
Nagao M, Takatori T, Matsuda Y, Nakajima M, Iwase H, Iwadate K (1997) Toxicol Appl Pharm 144:198–203
Black RM, Read RW (2007) Toxin Rev 26:275–298
Report on the alleged use of chemical weapons in the Ghouta Area of Damascus on 21 August 2013 (2013) Council on Foreign Relations, Netherlands. http://www.un.org/disarmament/content/ slideshow/Secretary_General_Report_of_CW_Investigation.pdf. Accessed 17 Sep 2013
Ellman G, Courtney D, Andres V, Featherstone R (1961) Biochem Pharm 7:88–95
Worek F, Mast U, Kidelen D, Diepold C, Eyer P (1999) Clin Chim Acta 288:73–90
Gallo MA, Lawryk NJ (1991) In: Hayes WJ Jr, Laws ER Jr (eds) Handbook of Pesticide Toxicology. Acedemic Press, New York
Noort D, Benschop HP, Black RM (2002) Toxicol Appl Pharm 184:116–126
Swaim LL, Johnson RC, Zhou Y, Sandlin C, Barr JR (2008) J Anal Toxicol 32:774–777
Adams TK, Capacio BR, Smith JR, Whalley CE, Korte WD (2005) Drug Chem Toxicol 27:77–91
Li B, Ricordel I, Schopfer LM, Baud F, Mégarbane B, Nachon F, Masson P, Lockridge O (2010) Toxicol Sci 116:23–31
Black RM, Noort D (2005) In: Mesilaakso M (ed) Chemical weapons convention analysis: sample collection, preparation, and analytical methods. John Wiley & Sons, England
Riches J, Morton I, Read RW, Black RM (2005) J Chromatogr B 816:251–258
Degenhardt C, Pleijsier K, Van der Schans MJ, Langenberg JP, Preston KE, Solano MI, Maggio VL, Barr JR (2004) J Anal Toxicol 28:364–371
Sporty JLS, Lemire SW, Jakubowski EM, Renner JA, Evans RA, Williams RF, Schmidt JG, Van der Schans MJ, Noort D, Johnson RC (2010) Anal Chem 82:6593–6600
Williams NH, Harrison JM, Read RW, Black RM (2007) Arch Toxicol 81:627–639
Li B, Nachon F, Froment MT, Verdier L, Debouzy JC, Brasme B, Gillon E, Schopfer LM, Lockridge O, Masson P (2008) Chem Res Toxicol 21:421–431
Nigg HN, Knaak JB (2000) Rev Environ Contam Toxicol 163:29–111
Black RM (2010) J Chromatogr B 878:1207–1215
Aurbek N, Thiermann H, Eyer F, Eyer P, Worek F (2009) Toxicol 259:133–139
Fidder A, Hulst AG, Noort D, de Ruiter R, van der Schans MJ, Benschop HP, Langenberg JP (2002) Chem Res Toxicol 15:582–590
Knaack JS, Zhou Y, Abney CW, Jacob JT, Prezioso SM, Hardy K, Lemire SW, Thomas J, Johnson RC (2012) Anal Chem 84:9470–9477
Taylor JK (1987) Quality assurance of chemical measurements. Lewis Publishers, Michigan
Westgard JO, Barry PL, Hunt MR (1981) Clin Chem 27:493–508
Caudill SP, Schleicher RL, Pirkle JL (2008) Stat Med 27:4094–4106
Carter MD, Crow BS, Pantazides BG, Watson CM, DeCastro BR, Thomas JD, Blake TA, Johnson RC (2013) J Biomol Screen. doi:10.1177/1087057113497799
Vesper H, Emons H, Gnezda M, Jain CP, Miller WG, Rej R, Schumann G, Tate J, Thienpont L, Vaks JE (2010) Characterization and qualification of commutable reference materials for laboratory medicine; approved guidelines (C53-A). Clinical and Laboratory Standards Institute, PA
Carter MD, Crow BS, Pantazides BG, Watson CM, Thomas JD, Blake TA, Johnson RC (2013) Anal Chem. doi:10.1021/ac4029714
Acknowledgments
The authors would like to thank the members of the Analytical Response and Chemical Laboratory Response Network Laboratories for technical assistance and thoughtful discussion during the preparation of this manuscript. Specifically, the authors would like to thank Ms. Chariety Sapp of the Centers for Disease Control and Prevention’s Incident Response Laboratory for aliquoting and distributing the convenience set specimens and Ms. Karen Shields for thoughtful input to the manuscript.
Funding information
This work was funded by the Centers for Disease Control and Prevention, Defense Threat Reduction Agency, and Oak Ridge Institute for Science and Education.
Disclaimer
The findings and conclusions in this report are those of the authors and do not necessarily represent the views of the Centers for Disease Control and Prevention. Use of trade names is for identification only and does not imply endorsement by the Centers for Disease Control and Prevention, the Public Health Service, or the US Department of Health and Human Services.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in the topical collection Analysis of Chemicals Relevant to the Chemical Weapons Convention with guest editors Marc-Michael Blum and R. V. S. Murty Mamidanna.
Rights and permissions
About this article
Cite this article
Pantazides, B.G., Watson, C.M., Carter, M.D. et al. An enhanced butyrylcholinesterase method to measure organophosphorus nerve agent exposure in humans. Anal Bioanal Chem 406, 5187–5194 (2014). https://doi.org/10.1007/s00216-014-7718-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-014-7718-7