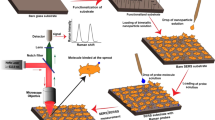
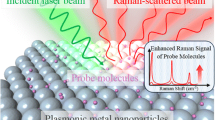
Abstract
Surface-enhanced Raman scattering (SERS) enhancement and the reproducibility of the SERS signal strongly reflect the quality and nature of the SERS substrates because of diverse localized surface plasmon resonance (LSPR) excitations excited at interstitials or sharp edges. LSPR excitations are the most important ingredients for achieving huge enhancements in the SERS process. In this report, we introduce several gold and silver nanoparticle-based SERS-active substrates developed solely by us and use these substrates to investigate the influence of LSPR excitations on SERS. SERS-active gold substrates were fabricated by immobilizing colloidal gold nanoparticles on glass slides without using any surfactants or electrolytes, whereas most of the SERS-active substrates that use colloidal gold/silver nanoparticles are not free of surfactant. Isolated aggregates, chain-like elongated aggregates and two-dimensional (2D) nanostructures were found to consist mostly of monolayers rather than agglomerations. With reference to correlated LSPR and SERS, combined experiments were carried out on a single platform at the same spatial position. The isolated aggregates mostly show a broadened and shifted SPR peak, whereas a weak blue-shifted peak is observed near 430 nm in addition to broadened peaks centered at 635 and 720 nm in the red spectral region in the chain-like elongated aggregates. In the case of 2D nanostructures, several SPR peaks are observed in diverse frequency regions. The characteristics of LSPR and SERS for the same gold nanoaggregates lead to a good correlation between SPR and SERS images. The elongated gold nanostructures show a higher enhancement of the Raman signal than the the isolated and 2D samples. In the case of SERS-active silver substrates for protein detection, a new approach has been adopted, in contrast to the conventional fabrication method. Colloidal silver nanoparticles are immobilized on the protein functionalized glass slides, and further SERS measurements are carried out based on LSPR excitations. A new strategy for the detection of biomolecules, particularly glutathione, under aqueous conditions is proposed. Finally, supramolecular J-aggregates of ionic dyes incorporated with silver colloidal aggregates are characterized by SERS measurements and correlated to finite-difference time-domain analysis with reference to LSPR excitations.

SPR and SERS images for isolated, elongated and two-dimensional gold nanostructures










Similar content being viewed by others
References
Siebert F, Hildebrandt P (2008) Vibrational spectroscopy in life science. Wiley-VCH, Darmstadt
Stiles PL, Dieringer FA, Shah NC, Van Duyne RP (2008) Annu Rev Anal Chem 1:601
Otto A (2002) J Raman Spectrosc 33:593
Yongan Y, Alexander B, Klaus K (2007) J Solid State Electrochem 11:150
Etchegoin PG, Le Ru EC (2008) Phys Chem Chem Phys 10:6079
Shalaev VM, Sarychev AK (1998) Phys Rev B 57:13265
Campion A, Kambhampati P (1998) Chem Soc Rev 27:241
Le Ru EC, Etchegoin PG (2006) Chem Phys Lett 423:63
Moskovits M, Jeong DH (2004) Chem Phys Lett 397:91
Kuncicky DM, Prevo BG, Velev OD (2006) J Mat Chem 16:1207
Fleischmann M, Hendra PJ, McQuillan AJ (1974) Chem Phys Lett 26:163
Moskovits M (1985) Rev Mod Phys 57:783
Schatz GC, Van Duyne RP (2002) Handbook of vibrational spectroscopy. Wiley, Chichester
Compton AH (1923) Phys Rev 21:483
Cardona M, Güntherodt G (1984) Light scattering in solids vol IV. Springer, Berlin
Anker JA, Hall WP, Lyandres O, Shah NC, Zhao J, Van Duyne RP (2008) Nat Mat 7:442
Kneipp K, Moskovits M, Kneipp H (2006) Surface-enhanced Raman scattering: physics and applications. Springer, Berlin
Ditlbacher H, Krenn JR, Felidj N, Lamprecht B, Schider G, Salerno M, Leitner A, Aussenegg FR (2002) Appl Phys Lett 80:404
Stefan AM, Kik PG, Atwater HA, Meltzer S, Harel E, Koel BE, Requicha AAG (2003) Nat Mat 2:229
Brongersma ML, Kik PG (2007) Surface plasmon nanophotonics. Springer, Dordrecht
Raether H (1986) Surface plasmons. Springer, New York
Haes J, Van Duyne RP (2004) Expert Rev Mol Diag 4:527
Itoh T, Biju V, Ishikawa M, Kikkawa Y, Hashimoto K, Ikehata A, Ozaki Y (2006) J Chem Phys 124:134708
Pettinger B (1986) J Chem Phys 85:7442
Itoh T, Yoshida K, Biju V, Kikkawa Y, Ishikawa M, Ozaki Y (2007) Phys Rev B 76:085405
Michaels AM, Nirmal M, Brus LE (1999) J Am Chem Soc 121:9932
Etchegoin P, Liem H, Maher RC, Cohen LF, Brown RJC, Hartigan H, Milton MJT, Gallop JC (2002) Chem Phys Lett 366:115
Otto A, Mrozek I, Grabhorn H, Akemann W (1992) J Phys Cond Mat 4:1143
Otto A (2003) Indian J Phys 77B:63
Persson N (1981) J Chem Phys Lett 82:561
Otto A (1991) J Raman Spectrosc 22:743
Maier SA, Atwater HA (2005) J Appl Phys 98:011101–1
Zeng J, Jia H, An J, Han X, Wu W, Zhao B, Ozaki Y (2008) J Raman Spec 39:1673
Jain PK, El-Sayed IH, El-Sayed MA (2007) Nanotoday 2:18
Hossain MK, Shimada T, Kitajima M, Imura K, Okamoto H (2008) Langmuir 24:9241
Imura K, Okamoto H, Hossain MK, Kitajima M (2005) Chem Lett 35:78
Murphy CJ, Sau TK, Gole AM, Orendroff CJ, Gao J, Gou L, Hunyadi SE, Li T (2005) J Phys Chem 109:13857
Min Y, Akbulut M, Kristiansen K, Golan Y, Israelachvili J (2008) Nature Mat 7:527
Xia Y, Yin Y, Lu Y, McLellan J (2003) Adv Func Mat 13:907
Maier SA, Brongersma ML, Kik PG, Meltzer S, Requicha AAG, Atwater HA (2001) Adv Mater 13:1501
Wu Y, Livneh T, Zhang YX, Cheng G, Wang J, Tang J, Moskovits M, Stucky GD (2004) Nano Lett 4:2337
Le F, Brandl DW, Urzhumov YA, Wang H, Kundu J, Halas NJ, Aizpurua J, Nordlander P (2008) ACS Nano 2:708
Wei A (2006) e-J Surf Sci Nanotech 4:9
Hossain MK, Shimada T, Kitajima M, Imura K, Okamoto H (2008) J Microsc 229:327
Olson LG, Uibel RH, Harris JM (2004) Appl Spectrosc 58:1394
Bulte JWM, Modo MMJ (2008) Nanoparticles in biomedical imaging: emerging technologies and applications. Springer, New York.
Imura K, Okamoto H, Hossain MK, Kitajima M (2006) Nano Lett 6:2173
Lu Y, Liu GL, Lee LP (2005) Nano Lett 5:5
Shafer-Peltier KE, Haynes CL, Glucksberg MR, Van Duyne RP (2003) J Am Chem Soc 125:588
Itoh T, Hashimoto K, Ozaki Y (2003) Appl Phys Lett 83:2274
Han XX, Kitahama Y, Tanaka Y, Guo J, Xu WQ, Zhao B, Ozaki Y (2008) Anal Chem 80:6567
Itoh T, Hashimoto K, Biju V, Ishikawa M, Wood BR, Ozaki Y (2006) J Phys Chem B 110:9579
Shimada T, Imura K, Hossain MK, Kitajima M, Okamoto H (2008) J Phys Chem C 112:4033
Wang H, Kundu J, Halas NJ (2007) Angew Chem Int Ed 46:9040
Jeanmaire DL, Van Duyne RP (1977) J Electroanal Chem 84:1
Nicolai SHA, Rubim JC (2003) Langmuir 19:4291
Chao Y, Zhou Q, Li Y, Yan Y, Wu Y, Zheng J (2007) J Phys Chem C 111:6990
Aroca RF, Constantino CJL (2000) Langmuir 16:5425
Drachev VP, Nashine VC, Thoreson MD, Ben-Amotz D, Davisson VJ, Shalaev VM (2005) Langmuir 21:8368
Li X, Xu W, Zhang J, Jia H, Yang B, Zhao B, Li B, Ozaki Y (2004) Langmuir 20:1298
Wei G, Wang L, Liu Z, Song Y, Sun L, Yang T, Li Z (2005) J Phys Chem B 109:23941
Xue G, Dong J (1991) Anal Chem 63:2393
Ni F, Cotton TM (1986) Anal Chem 58:3159
Wu Y, Zhao B, Xu W, Li B, Jung YM, Ozaki Y (1999) Langmuir 15:4625
Li F, Cao Q, Lu Y, Xue G (1997) Spectrosc Lett 30:451
Song W, Li W, Cheng Y, Jia H, Zhao G, Zhou Y, Yang B, Xu W, Tian W, Zhao B (2006) J Raman Spectrosc 37:755
Ruan W, Wang C, Ji N, Lu Z, Zhou T, Zhao B, Lombardi JR (2008) Langmuir 24:8417
Yao JL, Xu X, Wu DY, Xie Y, Ren B, Tian ZQ, Pan GP, Sun DM, Xue KH (2000) Chem Commun 1627
Carney J, Braven H, Seal J, Whitworth E (2006) IVD Technol 11:41
Han XX, Jia HY, Wang YF, Lu ZC, Wang CX, Xu WQ, Zhao B, Ozaki Y (2008) Anal Chem 80:2799
Han XX, Cai LJ, Guo J, Wang CX, Ruan WD, Han WY, Xu WQ, Zhao B, Ozaki Y (2008) Anal Chem 80:3020
Hossain MK, Shibamoto K, Ishioka K, Kitajima M, Mitani T, Nakashima S (2007) J Lumin 122–123:792
Lee PC, Meisel DJ (1982) J Phys Chem 86:3391
Aroca RF, Alvarez-Puebla RA, Pieczonka N, Sanchez-Cortez S, Garcia-Ramos JV (2005) Adv Colloid Interf Sci 116:45
Doering WE, Piotti ME, Natan MJ, Freeman RG (2007) Adv Mater 19:3100
Tian ZQ (2005) J Raman Spectrosc 36:466
Niemeyer CM, Mirkin CA (2004) Nanobiotechnology: concepts, applications and perspectives. Wiley, Denmark
Ooka AK, Kuhar KA, Cho N, Garrel RL (1999) Biospectrosc 5:9
Arenas JF, Castro JL, Otero JC, Marcos JI (2001) Biopolymers 62:241
Suh JS, Moskovits M (1986) J Am Chem Soc 108:4711
Singha A, Dasgupta S, Roy A (2006) Biophys Chem 120:215
Herne TM, Ahern AM, Garrell RL (1991) J Am Chem Soc 113:846
Podstawka E, Borszowska R, Grabowska M, Drag M, Kafarski P, Proniewicz LM (2005) Surf Sci 599:207
Stosch R, Henrion A, Schiel D, Guttler B (2005) Anal Chem 77:7386
Giese B, McGaughton D (2002) J Phys Chem B 106:1461
Sakar J, Chowdhury J, Ghosh M, De R, Talapatra GB (2005) J Phys Chem B 109:12861
Nirode WF, Devault GL, Sepaniak J (2000) Anal Chem 72:1866
Kneipp K, Wang Y, Dasari RR, Feld MS (1995) Spectrochim Acta 51A:481
Faulds K, Stewart L, Smith WE, Graham D (2005) Talanta 67:667
Bell SEJ, Sirimuthu NMS (2006) J Am Chem Soc 128:15580
Faulds K, Smith WE, Graham D (2004) Anal Chem 76:412
Breuzard G, Millot J-M, Riou J-F, Manfait M (2003) Anal Chem 75:4305
Rospendowski B, Kelly K, Wolf CR, Smith WE (1991) J Am Chem Soc 113:1217
Bizzarri AR, Cammostraro S (2002) Appl Spectrosc 56:1531
Feng M, Tachikawa H (2008) J Am Chem Soc 130:7443
Kumar GVP, Reddy BAA, Arif M, Kundu TK, Narayana C (2006) J Phys Chem B 110:16787
Pavel I, McCarney E, Elkhaled A, Morrill A, Plaxco K, Moskovits M (2008) J Phys Chem C 112:4880
Dou X, Takama T, Yamaguchi Y, Yamamoto H, Ozaki Y (1997) Anal Chem 69:1492
Cui Y, Ren B, Yao J-L, Gu R-A, Tian Z-Q (2006) J Phys Chem B 110:4002
Sengupta A, Laucks ML, Davis EJ (2005) Appl Spectrosc 59:1016
Jarvis RM, Goodacre R (2004) Anal Chem 76:40
Jarvis RM, Law N, Shadi IT, O’Brien P, Lloyd JR, Goodacre R (2008) Anal Chem 80:6741
Dijkstra RJ, Scheenen WJJM, Dam N, Roubos EW, Meulen JJt (2007) J Neurosci Meth 159:43
Sengupta A, Thai CK, Sastry MSR, Matthaei JF, Schwartz DT, Davis EJ, Baneyx F (2008) Langmuir 24:2000
Dou X, Jung Y-M, Cao Z-Q, Ozaki Y (1999) Appl Spectrosc 53:1440
Podstawka E, Ozaki Y, Proniewicz LM (2004) Appl Spectrosc 58:570
Podstawka E, Ozaki Y, Proniewicz LM (2004) Appl Spectrosc 58:1147
Podstawka E, Ozaki Y, Proniewicz LM (2008) Langmuir 24:10807
Xu S, Ji X, Xu W, Zhao B, Dou X, Bai Y, Ozaki Y (2005) J Biomed Opt 10:031112/1
Tamaru H, Kuwata H, Miyazaki HT, Miyano K (2002) Appl Phys Lett 80:1826
Hao E, Schatz GC (2004) J Chem Phys 120:357
Kobayashi T (1996) J-aggregates. World Scientific, Singapore
MacRae EG, Kasha M (1958) J Chem Phys 28:721
Kasha M, Rawls HR, El-Bayoumi MA (1965) Pure Appl Chem 11:371
Kasha M (1976) Spectroscopy of the excited state. Plenum, New York
Minoshima K, Taiji M, Misawa K, Kobayashi T (1994) Chem Phys Lett 218:67
Higgins DA, Reid PJ, Barbara PF (1996) J Phys Chem 100:1174
Kuhlbrandt W (1995) Nature 374:497
Akins DL, Özçelik S, Zhu HR, Guo C (1996) J Phys Chem 100:14396
Kano H, Saito T, Kobayashi T (2002) J Phys Chem A 106:3445
Ohno O, Kaizu Y, Kobayashi H (1993) J Chem Phys 99:4128
Wang J, Zhang P, He T, Xin H, Liu FC (1988) J Phys Chem 92:1942
Kneipp K, Kneipp H, Rentsch M (1987) J Mol Struc 156:331
Yao H, Kitamura S, Kimura K (2001) Phys Chem Chem Phys 3:4560
Acknowledgement
A KAKENHI (Grant-in-Aid for Scientific Research) on the Priority Area “Strong Photon-Molecule Coupling Fields (No. 470, 20043032)” from the Ministry of Education, Culture, Sports, Science and Technology of Japan (MEXT) is acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hossain, M.K., Kitahama, Y., Huang, G.G. et al. Surface-enhanced Raman scattering: realization of localized surface plasmon resonance using unique substrates and methods. Anal Bioanal Chem 394, 1747–1760 (2009). https://doi.org/10.1007/s00216-009-2762-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-009-2762-4