Abstract
This article reviews some recent applications of surface-enhanced Raman scattering (SERS) spectroscopy in biology, biochemistry and biomedicine. We start with a short description of theoretical background of Raman scattering enhancement by the plasmonic nanoparticles. SERS is a phenomenon observed for molecules in a close proximity to the surface of metallic nanostructures. We present an overview of SERS substrates fabricated using various physical and chemical methods. SERS spectroscopy, which combines very high sensitivity with molecular specificity, is a powerful technique for studying biologically important systems, ranging from simple molecules like amino acids, to extremely complex samples such as living cells and tissues. We demonstrate great potential of SERS not only for detection and identification of (bio)molecules, but also in monitoring various biochemical processes. The strategies that are used for biosensing with the aid of SERS spectroscopy are briefly described.
Similar content being viewed by others
Keywords
- Surface-enhanced Raman scattering (SERS)
- SERS of biological molecules
- Biosensors
- Intracellular SERS
- Nanoparticle probes
1 Introduction
Inelastic scattering of light by molecules, utilized by Raman spectroscopy, is nowadays commonly used to provide valuable information on molecular structure as well as chemical composition of the studied samples. Conventional (or normal) Raman spectroscopy suffers from very low scattering cross-sections compared with other spectroscopic methods. This significantly limits its applications to investigate molecules in solutions at low concentrations, which is a common requirement in analytical chemistry and biological systems. The simplest way to increase Raman intensity is exciting the spectrum with the laser beam of the energy corresponding to the energy difference between ground and excited electronic state. This effect, called resonance Raman (RR) scattering allows us to enhance the spectrum several orders of magnitude. However, in case of colorless samples, this method requires excitation in ultraviolet (UV), while the most popular light sources in Raman spectroscopy are visible or near infrared lasers. Another, more advanced possibility to increase the Raman scattering intensity is the use of non-linear effects such as stimulated Raman scattering. However, the most effective way of amplification of the Raman scattering is offered by surface enhanced Raman scattering (SERS) spectroscopy. In SERS, Raman signal may be enhanced by as much as 11 orders of magnitude. Typical SERS enhancement factors are of order of 104–106. Further enhancement of the Raman spectrum is obtained when the laser excitation energy corresponds to electronic transition of the molecule (resonance Raman). In this effect, called surface enhanced resonance Raman scattering (SERRS), enhancement factors can reach 1011, allowing detection at a single molecule level. Combined enhancement processes provide a Raman signal of sensitivity at least equivalent or even higher than that of fluorescence. Very high sensitivity, selectivity and ability to identify molecular species, together with other advantages make SERS/SERRS spectroscopy an exceptionally attractive tool for variety of applications in bioscience. SERS may compete successfully with other spectroscopic methods in bioscience, such as fluorescence.
Giant amplification of the Raman signal in SERS is generated by metallic nanoparticles (typically silver, gold and copper), on which target molecules are adsorbed. The first surface-enhanced Raman spectrum of pyridine adsorbed on a silver electrode was observed by Fleischmann and coworkers in 1974 [1] at the University of Southampton, but in fact they did not recognize the importance of their results. The first explanation of this new phenomenon in terms of a giant surface enhancement of the Raman scattering, observed by Fleischmann et al., was reported by Jeanmaire and van Duyne in 1977 [2]. Since 1977, many papers on both theoretical and practical aspects of SERS spectroscopy have been published. In the last several years, popularity of surface-enhanced Raman spectroscopy has grown rapidly. Among a great variety of applications, these connected with biomolecule detection develop extremely quickly.
2 SERS Phenomenon—Background and Mechanism of Enhancement
Phenomena concerning interactions between matter and light might be roughly divided into three cases: absorption and emission of light, scattering—when, as opposed to emission, angle distribution of radiated photons is observed—and reflection, when the wave vector changes its direction at an interface between two media according to the laws of reflection. Raman effect falls into the second category and is defined more accurately as inelastic scattering of light, which means that the energy of the scattered photons differs from the incident ones. Thanks to this energy difference, Raman spectroscopy informs us about energies of rotational and vibrational modes of the studied molecule. Raman spectrum contains a set of bands, which is characteristic for a given molecule, thus identifies the sample, providing its so called chemical fingerprint.
Vibrational Raman effect is classically described by Placzek’s theory, which is a simplified depiction of interaction between a molecule and electromagnetic wave that induces dipole moment in the molecule. As far as oscillation Raman effect is concerned, one needs to realize that molecular vibrations influence properties of the charge distribution in the molecule. Therefore, dipole oscillates with frequencies \({{\nu }_{0}}\), \({{\nu }_{0}}-{{\nu }_{osc}}\), and \({{\nu }_{0}}+{{\nu }_{osc}}\), emitting light of frequency equal to its own (\({{\nu }_{0}}\) being frequency of incident light beam and \({{\nu }_{osc}}\): frequency of molecular vibrations).
As it follows from the classical description, two types of Raman scattering are distinguished: when the energy of the scattered photons is lower or higher than the energy of the incident ones. It is explained as losing or gaining energy to vibrational or rotational modes of the molecule.
Unfortunately, Raman effect concerns only a small number of photons; vast majority of incident photons is scattered elastically without the energy change. Raman scattering involves only 1 of about 107 photons. Such obstacle prevented this technique from effectively developing in application fields for years. It was only in 1970s when Fleischmann et al. [1] observed a sudden rise in the Raman scattering intensity of the molecules adsorbed on a roughened electrode surface. Even though the contribution of the metal surface to the enhancement was not initially recognized, this technique is fully referred to as surface-enhanced Raman scattering (SERS) nowadays. Intrinsically weak Raman scattering can be enhanced thanks to the interaction between all three components of the system: light, adsorbed molecule and the metal surface. Nature of the effect, which is a borderline case between chemistry and physics, makes it necessary to combine languages of both fields in order to fully describe its mechanism. Theories explaining SERS belong to one of the two categories: chemical and electromagnetic ones. It is now well known that the dominant contribution to the enhancement arises from the electromagnetic effect. However, some observations, such as stronger enhancement of the Raman scattering of chemically adsorbed molecules as compared to physically adsorbed ones cause that chemical mechanism should be also taken into account.
Chemical explanation of the surface-enhanced Raman scattering mechanism makes use of charge-transfer theory. It focuses on the influence of the metal surface on energy levels of adsorbed molecules [3]. According to the chemical theory, Raman intensity gain in SERS is obtained by matching the energy of the incident beam with the energy of electron transition between Fermi level of the metal and a molecular level of the adsorbate. Situation when intensity of Raman scattering rises due to laser-induced intermolecular transitions is analogous to resonance Raman mechanism. In that case, enhancement of the Raman scattering is obtained when a laser beam of energy equal to electronic transition of the molecule is applied.
Some features of SERS prove that chemical mechanism plays a role in the enhancement; potential-dependent experiments show that the phenomenon might be tuned to a certain beam energy through applying appropriate bias to the metal adsorbent. However, there are features that cannot be understood on the ground of charge-transfer theory. It turns out that every SERS-active metal has its own energy work range. It is widely known that silver nanostructures yield the best enhancement when exposed to blue/green laser beam; on the other hand, red light supports SERS on gold structures. In addition, enhancement factor depends on electric permittivity of the medium. This inconsistency led to the second theory of SERS: electromagnetic one. It explores the influence of electromagnetic wave on a roughened surface. First attempt to describe SERS electromagnetically was dipole image theory. It presented a molecule as an oscillating electric dipole. Vibrations of the induced dipole are caused by oscillating electric compound of the electromagnetic wave. As the molecule is situated near the surface, it polarizes the metal. Thus, image dipole in the metal is created. It, in turn, causes extra polarization of the molecule which completes the feedback loop of interaction leading to the enhancement of the electric field near the metal surface. One of the main flaws of this theory is that it does not answer the question why metal nanostructures on the surface are essential to Raman enhancement. More refined theory includes the idea of surface plasmon resonance (SPR). Physicists define plasmons as collective oscillations of electron plasma (collection of valence electrons in the solid) [4]. Plasmons of different energies describe different excitation modes of such oscillations. Special properties of longitudinal charge density waves propagating at a metal/dielectric interface led to distinction of a subgroup of plasmons: surface plasmons [5]. These quasi-particles can be excited only by a transverse electromagnetic wave. What is more, optical properties of the system change drastically when energies of surface plasmons and incident light beam match. In that case, giant rise in electric field intensity near the interface is observed. This phenomenon is called surface plasmon resonance. For light from near UV/VIS/near IR range (usually used to excite Raman scattering), SPR is possible to obtain for plasmons confined in small metallic nanostructures; then we talk about localized surface plasmons (more accurately localized surface plasmon-polaritons, in order to refer to coupling between plasmons and light). When SPR occurs, molecules situated in the vicinity of such a metal nanostructure are exposed to electric field of intensity magnified several orders of magnitude (in comparison to the incident beam). As a result, electromagnetic enhancement factors reach up to 1011 (see critical paper on this topic by Ru et al. [6]). Exceptionally high enhancement factors are seen on the edges of the nanostructures. Such places might be called hot-spots (Fig. 3.1). Needless to say, only selected metals enhance Raman scattering effectively in the visible and near infrared range. Coinage metals, silver, gold and copper, face all the extra requirements needed to provide SERS signal. Extensive outlook on the problem of SERS substrates is presented in sect. 3.
Mathematic modelling of electric field amplitude distribution in the vicinity of different triangular silver nanoparticles as a result of surface plasmon resonance at different excitation wavelengths: (a) 412 nm, (b) 600 nm, (c) 458 nm. (Reproduced from Ref. [7] with kind permission of Elsevier BV)
One more thing needs to be highlighted. If one is already acquainted with Raman spectroscopy and knows Raman spectra of investigated molecules, it might be tempting to compare them to SERS spectra. However, they may—and usually they do—differ. Due to adsorption onto the metal surface, energies of molecular vibrations change which leads to modification of the Raman shift of certain bands. Furthermore, selection rules that apply to electromagnetic and charge transfer mechanisms of surface enhancement make some vibrational modes selectively enhanced, dependent on the relative contribution of each mechanism to total enhancement [8]. Thus, relative intensities of the bands in the SERS spectra are also altered in comparison to the normal Raman ones. As normal Raman selection rules do not apply in SERS, some bands ascribed to forbidden transitions may appear. Significant rise in the background is usually observed in the SERS spectra. Among less apparent differences one may recognize: increase of the depolarization ratio of the bands (intensity ratio of the perpendicular to the parallel component of the Raman scattered light) and potential dependence of positions and intensity of the bands for molecules adsorbed on electrodes.
3 SERS Supports
For generating SERS spectrum, molecule must be in close contact with SERS-active support. Although many materials have been reported as providing enhancement of the Raman scattering with visible and near-infrared excitation, there are three metals that exhibit the highest enhancement—silver, gold, and copper. Because of high reactivity of Cu, silver and gold are most commonly used as the SERS supports. A key problem in a variety of applications of SERS is obtaining substrates that provide stable, robust, reproducible and effectively enhancing surface. Development of strongly enhancing and highly reproducible substrates is critical in the applications of SERS as an important and efficient spectroscopic technique for studying biomolecules. Some examples of SERS supports are shown in Fig. 3.2.
The main source of the surface enhancement is a very strong electromagnetic field generated by excitation of the localized surface plasmons in the metallic nanoparticles (NP) (see Sect. 2). It is well known that the highest enhancement of the Raman spectrum is obtained when the energy of the laser beam used to excite the spectrum is close to the energy of the surface plasmons. To fulfill the condition of the plasmon resonance for a given excitation laser line, nanoparticles should have proper size, shape and composition. Historically, first substrates used for SERS were electrochemically roughened Ag electrode surfaces [1, 2]. They are still used in some SERS experiments, especially when the applied potential has to be controlled. The advantage of the electrodes is an ease of electrochemical preparation of the rough surface, high enhancement factors and possibility to simply regenerate the surface by subsequent electrochemical dissolution—deposition of the electrode material.
Soon after discovery of the SERS effect, metal (Ag and Au) colloids have been introduced as a very effective support (see Fig. 3.2) [9, 10]. Colloidal nanoparticles are produced mainly by chemical reduction of silver or gold cations in solutions, using reducing agents such as sodium citrate, sodium borohydride or hydroxylamine hydrochloride [11]. Metallic nanoparticles in colloidal suspensions are still the most widely used SERS active substrates, being ideal and simply produced substrates for performing measurements in solution. Main drawback of the chemically produced colloidal suspensions is some irreproducibility of the enhancement factors, caused by aggregation processes [12]. To overcome this problem, various methods of fabrication of metal nanoparticles have been developed. For some applications, including those in biology and biomedicine, metal nanoparticles obtained in colloidal suspension are deposited on the solid substrates. However, when colloidal particles isolated on planar substrates were used as the enhancing medium for ultra-sensitive detection of adenine, additional anomalous bands in the background were observed. Significant reduction of the spurious signals was obtained for hydroxylamine-reduced colloid, as compared to the more common citrate- and borohydride-reduced silver colloids [13].
There are many strategies of NP immobilization on solid supports, among which chemical or electrostatic self-assembling are the most popular. Chemical methods consist of functionalization of the solid substrate (usually glass or quartz) with thiol (SH) or amine (NH2) groups using bifunctional molecules which are able to attach Ag or Au nanoparticles from the colloid suspension. This strategy was first demonstrated by Chumanov et al. in 1995 [14] and developed by group of Natan et al. [15]. The idea of depositing metal nanoparticles from colloidal suspensions was also successfully applied to the fabrication of three-dimensional (3-D) structures of NP at the solid substrates by repeating chemical binding of the subsequent layers of NP deposit [16–18]. Another way is immobilization of NP by electrostatic attraction of the charged metal particles by oppositely charged solid support. In this strategy the most popular method is covering the solid substrate with positively charged derivatized silanes (e.g. aminopropyltrimethoxysilane) [14] or polymer layer such as poly(diallydimethylammonium chloride), which, as a positively charged polymer, is able to attract the negatively charged colloids [19] or polylysine [20]. Another, less common substrates include surfaces roughened by chemical treatment of solid substrate [21], “island films” deposited on glass and films deposited by evaporation or sputtering substrates [22, 23].
Metallic nanoparticles or other nanostructures can be also fabricated directly on the surface of the solid support using advanced physical methods such as nanolithography [24–28] or micro-contact printing [29]. Thin films of silver or gold can be also vapor-deposited over polystyrene or silica nanospheres of controlled diameter (FON-film over nanospheres), deposited on solid substrate. This method results in highly reproducible and effective supports [30, 31]. Using this technique, van Duyne group developed interesting SERS support by removing the underlying nanosphere mask, thus leaving periodic structure of metal nanoparticles (Fig. 3.2e), deposited on the free space between the nanospheres. As reported [32], supports prepared in this way exhibit very high enhancement factors (of order 108).
Overview of various SERS substrates: scanning electron microscopy (SEM) (a–c, e–h) and transmission electron microscopy (TEM) (d) images of a aggregated silver nanoparticles produced by a modified Lee–Meisel protocol, b gold nanoparticles arranged in clusters, c flower-like silver composites prepared by an enzymatic silver deposition, d SERS tags for labeling in bioanalytics consisting of gold cores coated by a silica shell, e regular arranged metallic triangles by applying nanosphere lithography, f optical fibers as templates for SERS substrates, g deposited nanoparticles within the optical fiber (magnification of Fig. 3.2f), h electron beam lithographic top-down fabrication of periodically arranged gold nanorhombs. (Reproduced from Ref. [33] with kind permission of Springer-Verlag)
Plasmonic properties of metal nanoparticles can be significantly changed by tailoring their shape or size. Numerous methods of production of shaped nanoparticles such as nanorods [34, 35] triangles [36], nanocubes [37, 38], polyhedrons [39], stars [40–42] and nanoflowers [43] have been developed. Some of these nanostructures exhibit higher enhancement factors as compared to nanospheres, because of the presence of sharp edges and corners that are able to concentrate extremely strong electromagnetic fields (see Fig. 3.1). Among a great variety of nanoparticles, hollow spheres with differing cavity diameter—which allows tuning the plasmon properties—are very effective enhancers [44].
The possibility of shifting plasmon resonance to the near infrared is sometimes important in biological applications, because of problems with surface photochemistry when using visible radiation (especially on silver) and/or problems with fluorescence background. This red-shift may be also achieved in core-shell nanoparticles built of a spherical silica core of varying dimensions, covered with a thin metal shell [45, 46]. It was confirmed that spherical Au nanoshells, in which plasmon resonance frequencies are controlled by the relative inner and outer radius of the metallic shell layer (see Fig. 3.3), can be used as an effective and reproducible SERS substrate, also for large biomolecules and more complex systems such as DNA and living cells [47–50].
(Left) TEM images of a Ag nanoparticle (top) and a hollow Au/Ag nanoshell (bottom). (Right) Schematic diagram of the particle geometry. (Reproduced from Ref. [51] with kind permission of Elsevier Science)
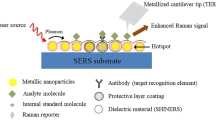
Interesting SERS substrate has been developed by Tian group [52, 53]. They coated Au nanoparticles with a very thin (2–3 nm) silica film that isolates the metal from the environment, preserving high enhancement factor of the shell-isolated nanoparticles (SHINERS—shell-isolated nanoparticle enhanced Raman spectroscopy). Protective layers of silica [54], glass [55] or polyethylene glycol [56] are also applied for metallic nanoparticles labeled with various compounds called Raman reporters. These encapsulated nanoprobes find many applications as SERS probes incorporated in a variety of biological materials (see Sect. 4.2). Larger enhancement of the Raman scattering than that produced by a single nanoparticle (NP) is created at the junction between two nanoparticles in a dimer, in which nanoparticles are separated by nanometer-scale gaps [57–60]. Such NP dimers are sometimes called hot spots. Reproducible fabrication of these nanostructures is however challenging, because the SERS enhancement factor critically depends on interparticle spacing [61].
This short review does not cover all developed SERS supports. Usually, SERS substrates are prepared in a given laboratory, but effectively enhancing gold solid support known as Klarite, or colloid suspensions are now also commercially available.
Another approach to enhance Raman scattering is offered by tip-enhanced Raman spectroscopy (TERS), reported for the first time in 2000 [62] and significantly developed in the last ten years [63–67]. In TERS, very strong electromagnetic field is located at the nanometer-scale metallic tip (Ag or Au). In an ideal case, it ends with a single metal nanoparticle. Its radius is much smaller than the diffraction limit so it allows measuring the spectrum from a much more confined spot. The Raman signal is strongly enhanced when tip is brought close to the sample irradiated by the laser beam (the movement is controlled by atomic force or scanning tunneling microscopy head) (Fig. 3.4). Therefore, TERS provides spectroscopic characterization of the sample with a nanometric spatial resolution.
Different configurations used in TERS experiments; left—side illumination, right—bottom illumination, using an inverted microscope. (Reproduced from Ref. [67] with kind permission of Elsevier BV)
To date, many TERS experiments have been reported for a variety of chemical and biochemical systems. They have been thoroughly reviewed in 2008 by Bailo and Deckert [68]. In the last ten years, TERS spectroscopy has been significantly developed and opened new possibilities of applications in a variety of fields in biology and medicine. In particular, experiments with living cells seem to be very promising. Interesting review that shows the potential and challenges of TERS applications in bioscience was published in 2010 by Deckert-Gaudig and Deckert [69].
4 SERS in Biology, Biochemistry and Biomedicine
4.1 SERS of Aminoacids, Peptides, Proteins, and Enzymes
Applications of SERS in bioscience cover very broad area, from relatively simple systems such as aminoacids and peptides through proteins, nucleic acids and enzymes, up to individual cells, living tissues, bacteria and viruses. Advantages of SERS spectroscopy in structural studies of small, biologically important molecules such as amino acids, purine and pyrimidine bases, porphyrins, flavines etc. have been shown just after discovery of the SERS phenomenon. These early studies have been reviewed by Cotton in 1988 [70]. SERS of amino acids and proteins has been observed on various SERS active substrates, among which silver and gold colloids are the most important and the most popular. However, it is well known that SERS spectra on metal colloids suffer from some irreproducibility [71–73] caused by aggregation of metallic nanoparticles. On the other hand, aggregating agents are indispensable to achieve better surface enhancement, because huge electromagnetic fields responsible for surface enhancement are created at particle junctions (nanogaps). A common problem in these experiments is the inhomogeneity of the colloids, which in turn provides local differences in the SERS enhancement factors. Most SERS studies of amino acids aimed at establishing the mode of interaction with the metal and the geometry of the molecule at the surface. SERS spectra allow determining the molecular form of amino acid present at the metal support (cationic, zwitterionic, or anionic), owing to high sensitivity of the spectrum to the molecular structure of the adsorbed species [74–79]. Some experimental SERS results are supported by theoretical calculations using DFT approach, making interpretation of the spectra more credible [80].
The most stable and reproducible SERS spectra of aminoacids have been recorded for sulphur containing molecules such as cysteine or methionine [77, 81–83]. SERS spectra of cysteine were recorded on various substrates such as colloidal nanoparticles [84] and rough silver electrodes, on which cysteine spontaneously self-assembles [81, 83]. Cysteine molecules are chiral—they can exist in one of two enantiomeric forms (L- or D-), that only differ in that their structures are mirror images of each other. Because only one enantiomer tends to be physiologically active while the other one is inactive or even toxic, drug compounds are produced in an enantiomerically pure form. Chiral surfaces may be applied as sensors for chiral recognition. In principle, both: the normal Raman and the SERS spectra are not sensitive to molecule chirality. However, in carefully designed experiments SERS spectra of cysteine monolayers on the Ag electrode surfaces have been shown to be sensitive to chirality of adjacent cysteine molecules at the metal surface [83]. It was demonstrated that there is a range of electrode potentials in which cysteine molecules are adsorbed mainly in zwitterionic form with the COO– groups close to the surface, while at more negative potentials NH3 + groups deprotonate at the surface with simultaneous weakening of the interaction of the carboxylic groups with the surface. As expected, the potential-induced effects for D-cysteine were similar to these for L-cysteine. However, for racemic mixture at acidic pH, the changes in the spectral pattern corresponding to potential-induced transition from adsorbed zwitterions to neutral molecule were considerably smaller. The origin of the observed effects were explained in terms of the most stable adsorption configuration for cysteine that involves metal-sulphur, metal-nitrogen and two hydrogen bonds between carboxylic groups (cyclic dimer) as indicated by DFT calculations [85]. In the case of LD cysteine dimers, there is a mismatch in the carboxylic bonds of the neighbouring molecules, which results in considerable weakening of the hydrogen bonds between them. The L- and D- cysteine enantiomers adsorb at the metal surface with equal probability, when adlayer is self-assembled from the solution. Thus, in spite of higher stability of the homochiral dimers, molecules of opposite chirality may exist at the surface in the immediate vicinity. In such a case intermolecular interactions between adjacent molecules are considerably weaker than for monolayer formed by the molecules of the same chirality, what is reflected in the band positions in the respective SERS spectra. Another example that confirms sensitivity of the SERS spectra to chirality is recognition of phenylalanine enantiomers by the enantiomeric cysteine monolayer deposited on a silver support [86] and SERS evidence for chiral discrimination in intermolecular interactions between adsorbed methionine molecules [87]. Further exploration of the stereospecificity of SERS may open new possibilities of applications of SERS spectroscopy in optical sensors of chiral biomolecules.
TERS is a very helpful technique in investigating molecules of biological importance. In 2009, first TERS spectra of cystine adsorbed on ultrasmooth gold substrate were reported. They indicated local variations of cystine adsorption [88]. It was also possible to observe two differently protonated histidine forms and determine two different geometries of adsorption on an atomically flat silver substrate [89]. SERS data for simple amino acids made it possible to interpret the SERS spectra of more complex systems such as peptides [90–95], dipeptides [96, 97], and proteins [98–100]. For example, it was possible to predict major features of the SERS spectra of penetratin, a 19 amino acid cell penetrating peptide, based on the SERS spectra of aromatic aminoacids such as tryptophan and phenylalanine and cysteine-containing aromatic peptides bound to Au nanoshells [91]. As may be seen in Fig. 3.5, the SERS spectrum of penetratin is dominated by features characteristic of the aromatic amino acids, greatly simplifying its interpretation.
While interpreting complex SERS spectra of peptides and proteins, it must be kept in mind that the most strongly enhanced bands in the spectrum correspond to the vibrational modes involving part of the molecule in the vicinity of the metal support. It has been proved that electromagnetic enhancement decays with distance from the surface, but is still measurable at about 10 nm [101]. Therefore, in case of very large biomolecules, spectral information may be confined to some fragments of the molecule directly interacting with the metal nanoparticle or in a short distance from the metal surface. This specificity and selectivity of the SERS spectra cause that in the case of large bio-molecules some fragments of the molecules are not “seen” in the spectra. On the other hand, this would enable characterization of the interaction between metal surfaces and biological molecule and determination of its orientation at the surface.
Comparison of empirically predicted (red) and measured (black) spectra of penetratin. (a) Molecular model of penetratin, including one phenylalanine (purple) and two tryptophan (green). (b) Raman spectra (peaks assigned to tryptophan are denoted with an asterisk). (c) SERS spectra. (Reproduced from Ref. [91] with kind permission of the American Chemical Society. Copyright 2008, American Chemical Society)
Also TERS spectra of peptides have been successfully recorded. TERS studies of peptide adsorption on metal surface (gold nanoplate) were carried out on a model molecule of oxidized glutathione and the structure of the peptide on the Au surface was determined [102]. The uniformity of the spectra measured at several points along a line across the gold substrate indicated the consistent orientation of the peptide. This result was regarded as crucial for possibility of characterizing and sequencing peptides with the help of this method.
Another problem related to SERS biosensing of proteins is their denaturation upon contact with metal nanoparticles. Thus the biocompatibility of the metal surface must be carefully controlled. The most simple way is to cover the nanoparticle surface with a self-assembled monolayer (SAM) [103] that prevents direct contact between the protein and the metal surface. There is a great variety of SAMs that could be used to prevent denaturation of the protein at the metal surface. However, the most optimal ones have to effectively bind the protein e.g. through electrostatic or covalent interaction, with simultaneous preservation of the protein properties and biological activity. The most popular SAMs are built of ω-substituted alkanethiols HS(CH2) n X, which form very stable metal-sulfur chemical bond. They create an interface with a well-defined composition and structure and with easily controlled properties achieved through the selective use of X groups. Among alkanethiols with negatively charged terminal groups, carboxylate-functionalized ones are the most frequently employed [104–109]. Sulfonate group is an example of an anionic group that can be used alternatively [110]. A great deal of work has been devoted to design biocompatible surfaces that ensure preserving the native structure of immobilized proteins. In the case of redox protein, communication between protein and electrode, which enables effective electron transfer, is also a major challenge. The case of cytochrome c (Cc) is a good illustration of the advantages for using SERS in studies of redox proteins. Numerous reports have been published on SERS of Cc immobilized on a silver or gold support modified with various monolayers [105, 108–112]. To improve intensity of the spectra, they are recorded using excitation laser line in resonance with molecular transition in Cc chromophore (SERRS spectra). The SERRS spectrum of Cc not only allows identifying the protein immobilized on the surface, but also makes it easy to determine the oxidation state and spin configuration of iron in heme groups, because Raman bands are well known markers of these properties [113, 114]. Moreover, sensitivity of the SERS spectrum to the orientation of the adsorbed molecules with respect to the surface causes that relative orientation of the heme plane may be determined by using so called surface section rules [8, 115]. Figure 3.6 shows SERRS spectrum of Cc electrostatically immobilized on silver electrode, which has been covered with a monolayer of thioglicolic acid (TGA) and resonance Raman spectrum of Cc solution. As may be seen, both spectra exhibit different frequencies and intensity pattern of the bands, which excludes risk that the bands detected in the SERRS spectrum originate from the protein in the solution. The band positions in the SERRS spectra are characteristic of the native 6cLS configuration of heme iron. Namely, the band at 1362 cm−1 indicates Fe2+ oxidation state, together with the 1493, 1542 and 1618 cm−1 bands which are markers of the native low spin (LS) configuration. On the contrary, in some cases bands assigned to oxidized form of Cc (at 1568 and 1638 cm−1) are clearly visible as for Ag support modified with mercaptoethanesulphonate (MES) (see spectrum in Fig. 3.6). The band at 1568 cm−1 indicates lots of Met-80 axial ligand leading to a non-native B2 5cHS state of heme and 1638 cm−1 feature is ascribed to the native B1 6cLS state [110].
SERRS spectra of cytochrome c adsorbed from 10−4 M aqueous solution of Cc on Ag electrode, coated with monolayer of thioglicolic acid (TGA) (upper spectrum—left panel) (A. Królikowska, unpublished results) and mercaptoethanesulphonate (MES) monolayer (right panel), as compared with the resonance Raman spectrum of 0.5 × 10−4 M Cc solution. (Reproduced from Ref. [110] with kind permission of John Wiley and Sons)
Potential-dependent SERRS studies for Cc immobilized on functionalized SAMs on Ag allow monitoring conformational changes in interfacial redox processes [111]. SERRS experiments may be performed in the stationary conditions with continuously changing electrode potential [111, 116], as well as in time-resolved mode by applying a potential jump [117]. These spectroelectrochemical experiments indicated that for the short thiol linkers the electron-transfer kinetics is determined by protein dynamics rather than by electron tunneling probabilities and that the reorientation of protein is controlled by the interfacial electric field. Thus, they show that SERRS can provide a very deep insight into dynamics of interfacial processes of proteins on the molecular level. SERRS experiments deliver much information about the heme part of the protein. A unique possibility of observing vibrational spectrum of both the heme and amino acid residues is however offered by TERS spectroscopy as demonstrated in 2008 [118].
Surface enhanced Raman scattering spectroscopy is now widely reported for enzymes. In this case, combination of SERS and resonance Raman spectroscopy is also usually applied, because it offers considerably higher sensitivity as compared to SERS. Excellent review on the development of SERS for the detection of enzyme transformations was published by Larmour et al. in 2010 [119]. Initially, SERRS was used for direct detection of enzymes. First results on SERRS of glucose oxidase has been reported at the very beginning of the SERS studies [120]. The first studies, in which biological activity of enzyme immobilized on Ag nanoparticles (Ag colloid) was monitored by SERRS spectroscopy, were published in 1993 [121]. In this report, 60–85 % retention of enzymatic activity of chlorocatechol dioxygenase in the reaction of catechol substrate with oxygen was demonstrated. The subsequent experiments for enzyme-substrate, enzyme-inhibitor, and enzyme-product complexes indicated that in general, SERRS allows probing the enzymatic processes in situ, during catalytic turnover. However, it has to be stressed that in all SERRS studies reported, some loss of the enzyme activity was observed upon contact with the metal support responsible for signal enhancement. Therefore, preparation of biocompatible substrates seems to be a key problem and important challenge in these experiments.
An alternative approach to study enzyme bioactivity with SERRS is recording the spectrum of enzymatically produced dyes, which are easily detected with SERRS. For example, azo dyes may be used in such experiments because they exhibit strong and very characteristic SERRS spectrum. SERRS signal of azoaniline adsorbed on Au nanoparticles has been applied for determination of glucose concentration [122], or adsorbed on Ag colloids for detection of antigen (mouse IgG) [123]. In this case azo dye was produced in peroxidase catalyzed oxidation reaction of o-phenylenediamine by hydrogen peroxide (Fig. 3.7).
SERS enzyme immunoassay system (a) and enzyme catalyzed reaction (b). (Reproduced from Ref. [123] with kind permission of the American Chemical Society)
The idea of utilizing SERRS spectrum of enzymatically produced dyes to detect enzymes was further developed by Stevenson et al. [124]. In a very interesting experiment, the SERRS spectrum of oxidized form of 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS), used as a substrate in an enzyme-linked immunosorbent assay (ELISA), was applied for the indirect detection of human prostate specific antigen (PSA) [124]. ABTS yields a green product upon enzymatic reaction with horseradish peroxidase and is commonly used as a colorimetric indicator of peroxidase activity. Experiments by Stevenson et al. [124] proved that SERRS provides a lower limit of detection and a wider range of linearity of the Raman signal vs. concentration as compared to the standard colorimetric ELISA approach. Another example of successful application of SERRS for detecting the presence and measuring activity of the enzyme is the work of Ruan et al. [125], in which SERRS spectrum of a blue indigo dye produced by oxidation and dimerization of 5-bromo-4-chloro-3-indolyl phosphate was utilized for monitoring activity of alkaline phosphatase.
Detection of DNA is one of the most important tasks in bioanalysis. To date, numerous SERS measurements of DNA utilizing different SERS supports have been reported. Experiments can be roughly divided into two groups. In the first group, called label-free method, DNA is directly detected with the SERS nanoprobes. For example, Halas group reported SERS spectra of DNA oligonucleotides and found that they are selectively dominated by the vibrational modes of adenine [126]. The authors also demonstrated the capability of SERS in investigating the interaction of DNA with various molecules (in this case—cisplatin). While direct SERS measurements for DNA are rather scarce, there are many reports on the indirect SERS experiments. This approach to DNA detection consists of monitoring the SERS spectrum of reporter molecules (usually dyes) that are attached to the metallic nanoparticles. This kind of SERS experiments for DNA is more extensively described in sect. 4.2.1.
4.2 SERS Biosensors
Term “biosensor” has no clear definition. Therefore, it might be applied to many different systems. Generally, biosensors provide data about the biological analyte they are exposed to, monitoring a change in their physicochemical response. If we monitor a SERS spectrum coming from the sensor in varying conditions, we deal with a SERS sensor. There is a whole range of tasks which biosensors may be employed to: monitoring a chosen kind of molecules, identification of the analyte composition, or determination of the environment parameters. Previous section of this review describes SERS experiments used for detection of certain biomolecules such as proteins or enzymes. Those examples perfectly fit in what we call a biosensor—they give information about the presence of a chosen biochemical compound in the sample. Those SERS sensors make use of various types of metallic substrates to enhance Raman scattering. However, lots of opportunities in the cell analysis open if metallic nanoparticles are engaged: thanks to their size, they can be successfully introduced into the cell in order to report the intracellular conditions from a precisely defined place.
4.2.1 Specific Sensing of Biomolecules
Previously described experiments intended to detect biomolecules were carried out in the following manner: object of the study needs to be found in the close proximity of the Raman enhancing substrate and then its characteristic SERS spectrum is collected. There are cases, however, when such a direct approach cannot be applied. Problem of glucose sensing illustrates such situation perfectly. Glucose is a molecule of great importance in human body, especially due to its relation to diabetes which has been considered a disease of civilization in the Western World for years while its incidence in developing countries has been increasing rapidly. Monitoring a concentration of glucose with SERS is hindered by its small Raman cross section and minimal adsorption on SERS-active metals. Van Duyne et al. performed a series of experiments to improve and optimize sensing parameters of glucose [127–130]. They increased its affinity to the SERS substrate by adsorbing a SAM of decanethiol on the AgFON surface [127]. It let them detect glucose over a clinically interesting concentration range. Further investigations focused both on the composition of the partition layer and the substrate. Researches showed that the sensor with (1-mercaptoundeca-11-yl)tri(ethylene glycol) as a partition layer works also in presence of interfering specimens, such as bovine serum albumin [128]. Real-time sensing was possible with a mixed SAM consisting of decanethiol and mercaptohexanol [129]. Such a device exhibited stability for 10 days and was used to determine concentration of glucose in the environment of bovine plasma. On the other hand, it was proved that replacing AgFON with AuFON and 1-mercaptoundeca-11-yl)tri(ethylene glycol) with 1-mercaptoocta-8-yltri(ethylene glycol) resulted in red-shift of plasmonic resonance which could be a step forward towards applying NIR lasers in this area. It could reduce fluorescence and background spectra of biological matrix [130].
Another example of sensing biomolecules are SERS experiments with bacterial spores. Indirect SERS detection of some bacterial spores is based on tracking down its biomarkers—molecules which are released from the spores while absent from vegetative cells. In case of bacterial genus of Bacillus, dipicolinic acid (DPA) serves as a biomarker for its spores. Sensitive DPA sensor is interesting from medical point of view, as one member of the genus, Bacillus anthracis, causes an acute disease: anthrax. AgFON substrate turned out to be applicable in this problem as well [131]. Obtained limit of detection for Bacillus subtilis spores (~ 2600 spores per 0,2 μl) was well below anthrax infectious dose of ~ 10000 spores per 0,2 μl. Similar experiment was carried out using Au-nanoparticles-based substrate [132].
Various fields of biosensing make use of so called nanotags: nanoparticles providing Raman enhancement for molecules adsorbed on them. Having labelled a biomolecule of interest with such a nanotag, one may be able to detect it indirectly: SERS spectrum of a molecular dye adsorbed on a tag indicates presence of the studied analyte.
Adenosine triphosphate (ATP) biosensor based on such nanolabels was demonstrated by Li et al. [133]. Nanotag consisted of malachite green isothiocyanate (MGITC) molecules attached to gold nanostars which were additionally covered with a layer of silica to prevent the reporter from detachment. It was functionalized with a single-stranded DNA creating the signalling probe, while the capture probe: complementary DNA strand was immobilized on a gold substrate. Under normal conditions, aptamer with the nanotag attached and the complementary strand create a duplex DNA, thus immobilizing the tag on the gold surface. However, DNA dissociates in the presence of ATP which leads to decrease in the SERS signal from the nanotag. Such a device determines not only absence or presence of ATP but also its concentration.
DNA hybridization is also exploited in detecting DNA itself (Fig. 3.8). Dye molecule might be adsorbed on a SERS inactive substrate; collecting its SERS spectrum becomes possible in the presence of a target strand after its hybridization with a capture strand on the surface and a DNA-probe functionalized with a silver nanoparticle [134].
On the other hand, reporter molecule can be adsorbed on a metal nanoparticle bound to the probe DNA strand; it is only after hybridization of the probe strand with an appropriate target strand on the sample surface when collecting the SERS spectrum of the dye is feasible [135]. Detected DNA strand might be also labeled directly with a reporter molecule whose SERRS is collected, thanks to creating a duplex DNA with a complementary strand on a SERS-active surface [136]. By modifying different strands with different dyes, multiplexed DNA detection was also obtained [137].
Examples of different approaches in DNA SERS sensing: a SERS label bound directly to the DNA strand [136], b metal nanoparticle bound to DNA single strand while the label is adsorbed on the surface [134]. (Adapted from Refs. [134, 136] with kind permission of respectively The American Chemical Society, Copyright 2007 and Royal Society of Chemistry)
SERS nanotags are widely used in immunoassays [138–140]. In this case, nanoparticles are functionalized not only with a reporter molecule but also with an antibody which introduces selectivity for a certain antigen. Antigen binds to the capture antibody immobilized on the surface and then to the antibody on the nanoprobe modified with the SERS reporter. Low limits of detection obtained in this approach make it competitive with other viral assays. It is possible to create a multisensitive assay by modifying nanotags for different antigens with different reporter molecules [141].
4.2.2 SERS Probing of Cells and Microorganisms
Very high sensitivity of SERS makes it an attractive method to study extremely complex systems such as single living cells. Compared to normal Raman spectra of the cells, SERS offers considerably shorter collection times, which is especially important in Raman mapping. Application of SERS for probing living cells was reviewed in several papers [142–145]. First SERS report, which demonstrated the capability of SERS for cell studies was published in 1991 [146], but broader interest of scientific society to this subject actually started in 2005, as shown in an interesting review by Vitol et al. [145]. In SERS experiments with cells, nanoprobes that are responsible for enhancement of the Raman signal are inserted into the cells. Therefore, the key problem in intracellular SERS is development of efficient and biocompatible metal nanoprobes that do not affect cell integrity and viability. Nanostructures used in these experiments should be small enough to be transferred through the cell membranes without damaging them. Various SERS probes have been used for intracellular experiments, starting from the most popular and easily fabricated colloidal silver and gold nanoparticles [147, 148], through nanoshells [49], nanorods [149], nanostars [150], nanoflowers [151], to more sophisticated probes such as nanopipettes [152].
Generally, there are two ways of performing intracellular SERS experiments. In the majority of experiments, metallic nanoprobes are labeled with molecules that exhibit strong SERS (or SERRS) spectrum. They are called reporter molecules. Then, the SERS spectrum of the reporter molecule (e.g. a dye), linked to the nanoprobe is recorded. In the second, label-free approach, metallic nanoprobes enhance Raman spectrum of native constituents of the cell such as DNA, RNA, lipids or aminoacids, delivering chemical structural information at the molecular level. Coupling of the modern Raman instruments with confocal microscope opened a new possibility of obtaining Raman maps that show spatial distribution of investigated species with high lateral resolution.
In case of using label-free SERS nanoprobes the signatures of biological molecules and structures constituting the cell in the immediate nanoparticle vicinity can be delivered. In SERS experiments, nanoparticles are either introduced inside the cell or located on the cell membrane surface. First important report demonstrating power of SERS in such experiments was published in 2002 by Kneipp et al. [153]. It was possible to observe vibrational bands that were ascribed to amino acids, DNA and RNA and to find their distribution inside the intestinal epithelial cells. In 2006, Kneipp et al. reported SERS spectra from endosomes in living individual epithelial and macrophage cells at different times after the uptake of gold nanoparticles. [147]. Metal particles were found to be collected in endosomes inside the cells. Spectra indicated that the molecular composition in the vicinity of the nanoprobes changes over time. Spectra also varied between different cell lines. Moreover, total SERS intensity increased due to formation of nanoaggregates of which dimers and trimers were found to exhibit the highest enhancement factors. On the other hand, it is known that the increasing distance between aggregated nanoparticles results in lowering of the electromagnetic enhancement factor, that in turn result in decreasing intensity of the spectra. Uncontrollable aggregation of nanoparticles with time is a major obstacle in using metal colloids for SERS inside the cells, because of undesirable processes that result in changes of both the spectral pattern and SERS intensity [147].
SERS is also applied to microorganisms such as bacteria. It is difficult to introduce metal nanoparticles into bacterial cells because of their size, which is much smaller in comparison to mammalian cells. Therefore, the studies of bacteria with SERS are mainly focused on the extracellular experiments. However, Jarvis et al. reported intracellular SERS of Geobacter sulfurreducens using colloidal metal nanoparticles prepared from the enzymatic reduction of silver and gold salts by these bacteria [154]. Very interesting and critical review of the problems that are connected with the interpretation of the SERS spectra of such a complex molecular systems was published by Efrima and Zeiri [155]. They paid attention to considerable differences between the SERS spectra reported for the same bacterium by various authors. Efrima and Zeiri suggested that the differences observed are due to differences in preparation procedures that may result in interactions of nanoparticles with various chemical constituents of the cells. They demonstrated clear dependence of the spectra on localization of the nanoparticles. Unusual selectivity and sensitivity of SERS spectrum toward flavins, an essential coenzyme in cells, was observed for external (on the cell walls) coating of the bacteria with the metal particles. On the contrary, when the metal colloids were produced inside the cell, flavin signatures disappeared and the bands corresponding to internal chemical constituents appeared instead. To overcome the limitations of colloidal nanoparticles for SERS of the cells and microorganisms and to gain more control upon the position of the sensor, new approaches to intracellular sensing have been proposed. The most important are: using metal-coated fiber-optic tips [156, 157], SERS-active glass nanopipettes [145, 152] and tip-enhanced Raman scattering (TERS) [158, 159].
TERS technique, briefly described in Sect. 3, is a perfect tool in biosensing. Cell walls were successfully investigated with TERS. Thanks to sub-optical spatial resolution of this technique, it is possible to distinguish between protein and lipid components of the Staphylococcus epidermidis cell wall [158, 160]. Fluctuations in the spectra collected from one spot of the outer layer of the cell may be ascribed to the bacterial surface dynamics [161]. Surface of a single tobacco mosaic virus was also characterized with TERS. Not only protein components from the capsid, but also some RNA features were seen in the spectrum (Fig. 3.9) [159]. These very promising results show that fast spectroscopic identification of single virus particles with TERS is possible.
Top: AFM image of tobacco mosaic virus (TMV); bottom: TERS spectra collected from four different points marked in the top picture. (Reproduced from Ref. [159] with kind permission of John Wiley and Sons)
Biosensors are able to look directly into the cell compartments—which used to be impossible without complex techniques—without affecting cell viability. However, uncoated nanoparticles suffer from lack of specificity. While features of the spectra could be assigned to some types of biomolecules, such a simple device fails to determine the presence of particular molecules. Rich spectra of biomolecules make it difficult to determine all the components accurately—profusion of the bands, as well as their overlapping, hinder clarity of the spectrum. In order to increase specificity, it is better to detect molecules in an indirect way. In this case, SERS signal is collected from a small reporter molecule adsorbed on a nanoparticle. Molecular dyes are frequently used as reporter molecules; as a result, even higher intensities thanks to SERRS effect are obtained.
Finding out the composition of the analyte is not always the main goal of the research. Sometimes it is more interesting to determine intracellular variables. Concept of the biosensor remains the same: surface of the nanoparticle yielding Raman enhancement is modified with reporter molecules whose SERS spectra are recorded. So that the sensor could monitor the environment, spectrum of the reporter should change with the studied parameter, such as intracellular redox potential. It plays an important role in protein interactions, apoptosis and signalling. In this approach, SERS spectra of the nanoprobe-attached molecules that exhibit SERS spectrum sensitive to oxidation state are recorded. Auchinvole et al. constructed a biosensor based on the SERS effect of two separate molecules, quinone derivatives: 1,8-diaza-4,5-dithia-1,8-di(2-chloro-[1, 4]-naphthoquinone-3-yl)octane (NQ) and 2-mercaptobenzene-1,4-diol (HQ), deposited on the surface of gold nanoshells [162]. They undergo reversible two-electron redox reactions. The nanoprobes were introduced into single fibroblast cell and the SERS spectra were collected from the cytoplasm, where nanoprobes have been located. Plots of the marker band intensity changes show sigmoidal shape for both of the sensors. Based on the intensities of the respective SERS bands, relative content of the reduced/oxidized form of probe molecules was estimated, enabling determination of the value of resting cellular potential. What is important, it was proved that the introduction of the device into the cell does not change the redox potential inside. In separate experiments, the authors also demonstrated the capability of SERS to monitor the localized response to reductive and oxidative stress.
Another essential variable of the intracellular environment is the pH value. Different compartments are characterized by different pH ranges which additionally change over time. Anomalies in the pH value might be associated with several diseases, such as kidney failure or certain cancers. Importance of acidity inside the cell led to a big collection of pH biosensors throughout the literature. Determination of pH value inside the living cell may be an interesting illustration of possible applications of labeled SERS nanoprobes [163–170]. In these experiments, gold nanoparticles are functionalized with reporter molecules that demonstrate strong SERS spectrum sensitive to pH, such as 4-mercaptobenzoic acid (PMBA), 4-mercaptopyridine [165] or 4-aminothiophenol [167]. Next, the nanosensors are introduced into the cells and the SERS spectra of a reporter molecule are collected at many points of the sample, thus probing and imaging pH values inside the individual living cells. Due to changes in their chemical structure in varying pH, intensities of the chosen marker band vary as well. Kneipp et al. employed PMBA based sensor to perform SERS mapping of the pH-value in the cell [163]. They also broadened the working range of the sensor by exploiting SEHRS (surface-enhanced hyper-Raman scattering). A mobile SERS nanosensor made of aggregates of Au nanoparticles, functionalized with PMBA enabled monitoring changes in local pH value over time and observing some metabolic changes in living NIH/3T3 cells [166]. Local pH value was also monitored inside individual cancer cells after treatment by the photodynamic therapy drug emodine [168]. In this report, a micrometer-sized silica beads covered with Ag nanoparticles functionalized with PMBA were passively embedded into the cells. The pH changes over time revealed ability of the cell to recover when emodin was activated with low light dosage (pH fluctuated slightly around the average value, about 5.5), while after treatment with a high light dosage, the intracellular pH increased substantially, triggering consecutive processes and finally cell death was observed.
This short review does not cover all applications of SERS in cellular sensing. For example, SERS nanotags were successfully applied to detect cancer markers in living cells [171, 172]. These reports clearly demonstrate perspectives of SERS spectroscopy as a promising tool in biomedicine.
5 Concluding Remarks
As shown in this review, SERS is an exceptionally selective and nondestructive spectroscopic method which offers sensitivity down to the single molecule level. It has a great potential for a variety of applications in bioscience, including in situ experiments in living cells and microorganisms. Despite great progress in bioanalytical applications of SERS spectroscopy in the last several years, there are still many obstacles that must be overcome to make this method a more universal tool. First, developing stable, reproducible, and highly enhancing substrates is still a great challenge in SERS spectroscopy of biological samples. Controlling the distribution of the SERS nanoparticles inside the living biological materials such as cells, tissues and microorganisms is another task that must be solved. In targeted sensing in biological samples, the magnetic plasmonic nanoparticles have great potential, since they enable displacing the nanoprobes in magnetic field. Developing biocompatible and non-toxic SERS nanoprobes is a very important and still unresolved problem in applications of SERS for in vivo experiments with living cells and microorganisms as well.
In the last decade, great progress has been made in combining Raman spectroscopy with scanning tunnelling (STM) or atomic force microscopy (AFM). It resulted in developing fascinating technique called TERS. This technique opens new, exciting perspectives for noninvasive imaging of a variety of biological systems, e.g. cell membranes with nanometric spatial resolution. Main challenge of TERS spectroscopy is fabrication of reproducible, high quality tips, that are necessary for effective enhancement of the Raman signal.
References
Fleischmann M, Hendra PJ, McQuillan AJ (1974) Raman spectra of pyridine adsorbed at a silver electrode. Chem Phys Lett 26(2):163–166
Jeanmaire DL, Van Duyne RP (1977) Surface Raman spectroelectrochemistry. Part I. Heterocyclic, aromatic, and aliphatic amines adsorbed on the anodized silver electrode. J Electroanal Chem Interfacial Electrochem 84(1):1–20
Lombardi JR, Birke RL, Lu T, Xu J (1986) Charge-transfer theory of surface enhanced Raman spectroscopy: Herzberg-Teller contributions. J Chem Phys 84:4174–4180
Pines D (1956) Collective energy losses in solids. Rev Mod Phys 28:184–198
Stern EA, Ferrell RA (1960) Surface plasma oscillations of a degenerate electron gas. Phys Rev 120:130–136
Le Ru EC, Blackie E, Meyer M, Etchegoin PG (2007) Surface enhanced Raman scattering enhancement factors: a comprehensive study. J Phys Chem C 111:13794–13803
Kottmann JP, Martin OJF, Smith DR, Schultz S (2001) Dramatic localized electromagnetic enhancement in plasmon resonant nanowires. Chem Phys Lett 341:1–6
Creighton JA (1988) The selection rules for surface-enhanced Raman spectroscopy. In: Clark RJH, Hester RE (eds) Spectroscopy of surfaces. Advances in Spectroscopy, vol 16. Wiley, New York, pp 37–89
Creighton A, Blatchford CG, Albrecht MG (1979) Plasma resonance enhancement of Raman scattering by pyridine adsorbed on silver and gold sol particles of size comparable to the excitation wavelength. J Chem Soc Faraday Trans 2 75:790–798
Lee PC, Meisel D (1982) Adsorption and surface-enhanced Raman of dyes on silver and gold sols. J Phys Chem 86:3391–3395
Leopold N, Lendl B (2003) A new method for fast preparation of highly surface-enhanced Raman scattering (SERS) active silver colloids at room temperature by reduction of silver nitrate with hydroxylamine hydrochloride. J Phys Chem B 107(24):5723–5727
Blatchford CG, Campbell JR, Creighton JA (1982) Plasma resonance enhanced Raman scattering by adsorbates on gold colloids: the effects of aggregation. Surf Sci 120:435–455
Larmour IA, Faulds K, Graham D (2012) SERS activity and stability of the most frequently used silver colloids. J Raman Spectrosc 43:202–206
Chumanov G, Sokolov K, Gregory BW, Cotton TM (1995) Colloidal metal films as a substrate for surface-enhanced spectroscopy. J Phys Chem 99:9466–9471
Grabar KC, Brown KR, Keating CD, Stranick SJ, Tang SL, Natan MJ (1997) Nanoscale characterization of gold colloid monolayers: a comparison of four techniques. Anal Chem 69:471–477
Fan M, Brolo AG (2009) Silver nanoparticles self assembly as SERS substrates with near single molecule detection limit. Phys Chem Chem Phys 11:7381–7389
Andrade GFS, Fan MK, Brolo AG (2010) Multilayer silver nanoparticles-modified optical fiber tip for high performance SERS remote sensing. Biosens Bioelectron 25:2270–2275
Addison CJ, Brolo AG (2006) Nanoparticle-containing structures as a substrate for surface-enhanced Raman scattering. Langmuir 22:8696–870
Li X, Xu W, Zhang J, Jia H, Yang B, Zhao B, Li B, Ozaki Y (2004) Self-assembled metal colloid films: two approaches for preparing new SERS active substrates. Langmuir 20:1298–1304
Tantra R, Brown RJC, Milton MJT, Gohil DD (2008) A practical method to fabricate gold substrates for surface-enhanced Raman spectroscopy. Appl Spectrosc Vol. 62:992–1000
Yang C, Xie YT, Yuen MMF, Xiong XM, Wong CP (2010) A facile chemical approach for preparing a SERS active silver substrate. Phys Chem Chem Phys 12(43):14459–14461
Semin DJ, Rowlen KL (1994) Influence of vapor deposition parameters on SERS active Ag film morphology and optical properties. Anal Chem 66:4324–4331
Roark SE, Rowlen KL (1994) Thin Ag films: influence of substrate and postdeposition treatment on morphology and optical properties. Anal Chem 66:261–270
Haynes CL, Van Duyne RP (2001) Nanosphere lithography: a versatile nanofabrication tool for studies of size-dependent nanoparticle optics. J Phys Chem B 105:5599–5611
Haes AJ, Chang L, Klein WL, Van Duyne RP (2005) Detection of a biomarker for Alzheimer’s disease from synthetic and clinical samples using a nanoscale optical biosensor. J Am Chem Soc 127(7):2264–2271
Stosch R, Yaghobian F, Weimann T, Brown RJC, Milton MJT, Guttler B (2011) Lithographical gap-size engineered nanoarrays for surface-enhanced Raman probing of biomarkers. Nanotechnology 22(10):105303
Abu Hatab NA, Oran JM, Sepaniak MJ (2008) Surface-enhanced Raman spectroscopy substrates created via electron beam lithography and nanotransfer printing. ACS Nano 2(2):377–385
Cialla D, Huebner U, Schneidewind H, Moeller R, Popp J (2008) Probing innovative microfabricated substrates for their reproducible SERS activity. ChemPhysChem 9(5):758–762
Porter Jr LA, Choi HC, Schmeltzer JM, Ribbe AE, Elliott LCC, Buriak JM (2002) Electroless nanoparticle film deposition compatible with photolithography, microcontact printing, and dip-pen nanolithography patterning technologies. NanoLetters 2(12):1369–1372
Dieringer JA, McFarland AD, Shah NC, Stuart DA, Whitney AV, Yonzon CR, Young MA, Zhang XY, Van Duyne RP (2006) Surface enhanced Raman spectroscopy: new materials, concepts, characterization tools, and applications. Faraday Discussions 132:9–26
Baia L, Baia M, Popp J, Astilean S (2006) Gold films deposited over regular arrays of polystyrene nanospheres as highly effective SERS substrates from visible to NIR. J Phys Chem B 110(47):23982–23986
Haynes CL, Van Duyne RP (2003) Plasmon-sampled surface-enhanced Raman excitation spectroscopy. J Phys Chem B 107:7426–7433
Cialla D, März A, Böhme R, Theil F, Weber K, Schmitt M, Popp J (2012) Surface-enhanced Raman spectroscopy (SERS): progress and trends. Anal Bioanal Chem 403:27–54
Mohapatra S, Siddhanta S, Kumar DR, Narayana C, Maji TK (2010) Facile and green synthesis of SERS active and ferromagnetic silver nanorods. Eur J Inorg Chem 31:4969–4974
Smitha SL, GopchandranKG, Ravindran TR, Prasad VS (2011) Gold nanorods with finely tunable longitudinal surface plasmon resonance as SERS substrates. Nanotechnology 22:265705
Rycenga M, Camargo PHC, Li W, Moran CH, Xia Y, (2010) Understanding the SERS effects of single silver nanoparticles and their dimers, one at a time. J Phys Chem Lett 1:696–703
Sun Y, Xia Y (2002) Shape-controlled synthesis of gold and silver nanoparticles. Science 298:2176–2179
Rycenga M, Kim MH, Camargo PHC, Cobley C, Li Z-Y, Xia Y (2009) Surface-enhanced Raman scattering: comparison of three different molecules on single-crystal nanocubes and nanospheres of silver. J Phys Chem A 113:3932–3939
Eguchi M, Mitsui D, Wu H-L, Sato R, Teranishi T (2012) Simple reductant concentration-dependent shape control of polyhedral gold nanoparticles and their plasmonic properties. Langmuir 28(24):9021–9026
Khoury CG, Vo-Dinh T (2008) Gold nanostars for surface-enhanced Raman scattering: synthesis, characterization and optimization. J PhysChem C 112(48):18849–18859
Esenturk EN, Walker ARH (2009) Surface-enhanced Raman scattering spectroscopy via gold nanostars. J Raman Spectrosc 40(1):86–91
Rodriguez-Lorenzo L, Alvarez-Puebla RA, Garcia J, de Abajo F, Liz-Marzan LM (2010) Surface enhanced Raman scattering using star-shaped gold colloidal nanoparticles. J Phys Chem C 114(16):7336–7340
Kim JH, Kang T, Yoo SM, Lee SY, Kim B, Choi YK (2009) A well-ordered flower-like gold nanostructure for integrated sensors via surface-enhanced Raman scattering. Nanotechnology 20(23):235302
Gellner M, Kustner B, Schlucker S (2009) Optical properties and SERS efficiency of tunable gold/silver nanoshells. Vib Spectrosc 50:43–47
Oldenberg SJ, Averitt RD, Westcott SL, Halas NJ (1998) Nanoengineering of Optical Resonances. Chem Phys Lett 288:243–247
Alvarez-Puebla RA, Ross DJ, Nazri GA, Aroca RF (2005) Surface-enhanced Raman scattering on nanoshells with tunable surface plasmon resonance. Langmuir 21:10504–10508
Talley CE, Jackson JB, Oubre C, Grady NK, Hollars CW, Lane SM, Huser TR (2005) Surface-enhanced Raman scattering from individual Au nanoparticles and nanoparticle dimer substrates. Nano Lett 5:1569–1574
Jackson JB, Halas NJ (2004) Surface-enhanced Raman scattering on tunable plasmonic nanoparticle substrates. Proc Natl Acad Sci USA 101:17930–17935
Ochsenkuhn MA, Jess PRT, Stoquert H, Dholakia K, Campbell CJ, (2009) Nanoshells for surface-enhanced Raman spectroscopy in eukaryotic cells: cellular response and sensor development. ACS Nano 3(11):3613–3621
Lal S, Grady NK, Goodrich GP, Halas NJ (2006) Profiling the near field of a plasmonic nanoparticle with Raman-based molecular rulers. Nano Lett 6:2338–2343
Gellner M, Küstner B, Schlücker S (2009) Optical properties and SERS efficiency of tunable gold/silver nanoshells. Vibr Spectr 50:43–47
Li JF, Huang YF, Ding Y, Yang ZL, Li SB, Zhou XS, Fan FR, Zhang W, Zhou ZY, Wu DY, Ren B, Wang ZL, Tian ZQ (2010) Shell-isolated nanoparticle-enhanced Raman spectroscopy. Nature 64(7287):392–395
Anema JR, Li JF, Yang ZL, Ren B, Tian ZQ (2011) Shell-isolated nanoparticle-enhanced Raman spectroscopy: expanding the versatility of surface-enhanced Raman scattering. Annu Rev Anal Chem 4:129–150
Kustner B, Gellner M, Schutz M, Schoppler F, Marx A, Strobel P, Adam P, Schmuck C, Schlucker S (2009) SERS labels for red laser excitation: silica-encapsulated SAMs on tunable gold/silver nanoshells. Angew Chem Int Ed 48:1950–1953
Mulvaney SP, Musick M D, Keating CD. Natan MJ (2003) Glass-coated, analyte-tagged nanoparticles: a new tagging system based on detection with surface-enhanced Raman scattering. Langmuir 19:4784–4790
Merican Z, Schiller TL, Hawker CJ, Fredericks PM, Blakey I (2007) Self-assembly and encoding of polymer-stabilized gold nanoparticles with surface-enhanced Raman reporter molecules. Langmuir 23:10539–10545
Moskovits M (2005) Surface-enhanced Raman spectroscopy: a brief retrospective. J Raman Spectrosc 36:485–496
Halas NJ, Lal S, Chang WS, Link S, Nordlander P (2011) Plasmons in strongly coupled metallic nanostructures. Chem Rev 111:3913–3939
Camden JP, Dieringer JA, Wang Y, Masiello DJ, Marks LD, Schatz GC, Van Duyne RP (2008) Probing the structure of single-molecule surface-enhanced Raman scattering hot spots. J Am Chem Soc 130:12616–12617
Rycenga M, Camargo PHC, Li W, Moran CH, Xia Y (2010) Understanding the SERS effects of single silver nanoparticles and their dimers, one at a time. J Phys Chem Lett 1:696–703
Wustholz KL, Henry AI, McMahon JM, Freeman RG, Valley N, Piotti ME, Natan MJ, Schatz GC, Van Duyne RP (2010) Structure-activity relationships in gold nanoparticle dimers and trimers for surface-enhanced Raman spectroscopy. J Am Chem Soc 132:10903–10910
Stockle RM, Suh YD, Deckert V, Zenobi R (2000) Nanoscale chemical analysis by tip-enhanced Raman spectroscopy. Chem Phys Lett 318:131–136
Pettinger B, Picardi G, Schuster R, Ertl G (2003) Surface-enhanced and STM tip-enhanced Raman spectroscopy of CN ions at gold surfaces. J Electroanal Chem 554:293–299
Picardi G, Nguyen Q, Schreiber J, Ossikovski R (2007) Comparative study of atomic force mode and tunneling mode tip-enhanced Raman spectroscopy. Eur Phys J Appl Phys 40:197–201
Zhang W, Yeo BS, Schmid T, Zenobi R (2007) Single-molecule tip-enhanced Raman spectroscopy with silver tips. J Phys Chem C 111:1733–1738
Sonntag MD, Klingsporn JM, Garibay LK, Roberts JM, Dieringer JA, Seideman T, Scheidt KA, Jensen L, Schatz GC, Van Duyne RP (2012) Single-molecule tip-enhanced Raman spectroscopy. J Phys Chem C 116:478–483
Yeo BS, Stadler J, Schmid T, Zenobi R, Zhang W (2009) Tip-enhanced Raman spectroscopy—its status, challenges and future directions. Chem Phys Lett 472:1–13
Bailo E, Deckert V (2008) Tip-enhanced Raman scattering. Chem Soc Rev 37:921–930
Deckert-Gaudig T, Deckert V (2010) Tip-enhanced Raman scattering (TERS) and high-resolution bio nano-analysis—a comparison. Phys Chem Chem Phys 12:12040–12049
Cotton TM (1988) The application of surface-enhanced Raman scattering to biochemical systems. In: Clark RJH, Hester RE (eds) Spectroscopy of surfaces. Advances in Spectroscopy, vol 16. Wiley, New York, pp 91–153
Aliaga AE, Osorio-Roman I, Leyton P, Garrido C, Carcamo J, Caniulef C, Celis F, Díaz GF, Clavijo E, Gomez-Jeria JS, Campos-Vallette MM (2009) Surface-enhanced Raman scattering study of L-tryptophan. J Raman Spectrosc 40:164–169
Aliaga AE, Osorio-Roman I, Garrido C, Leyton P, Carcamo J, Clavijo E, Gomez-Jeria JS, Diaz FG, Campos-Vallette MM (2009) Surface enhanced Raman scattering study of L-lysine. Vib Spectrosc 50(1):131–135
Yaffe NR, Blanch EW (2008) Effects and anomalies that can occur in SERS spectra of biological molecules when using a wide range of aggregating agents for hydroxylamine-reduced and citrate-reduced silver colloids. Vibr Spectrosc 48:196–201
Suh JS, Moskovits M (1986) Surface-enhanced Raman spectroscopy of amino acids and nucleotide bases adsorbed on silver. J Am Chem Soc 108:4711–4718
Lee HI, Suh SW, Kim MS (1988) Raman spectroscopy of L-tryptophan-containing peptides adsorbed on a silver surface. J Raman Spectrosc 19:491–495
Vidugiris GJA, Gudavicius AV, Razumas VJ, Kulys JJ (1989) Structure-potential dependence of adsorbed enzymes and amino acids revealed by the surface enhanced Raman effect. Eur Biophys J 17:19–23
Stewart S, Fredericks PM (1999) Surface-enhanced Raman spectroscopy of amino acids adsorbed on an electrochemically prepared silver surface. Spectrochim Acta Part A 55:1641–1660
Podstawka E, Ozaki Y, Proniewicz L (2004) Part I: surface-enhanced Raman spectroscopy investigation of amino acids and their homodipeptides adsorbed on colloidal silver. Appl Spectrosc 58:570–580
Podstawka E, Ozaki Y, Proniewicz L (2005) Part III: surface-enhanced Raman spectroscopy investigation of amino acids and their homodipeptides deposited onto colloidal Gold Surface. Appl Spectrosc 59:1516–1526
Guerrero AR, Aroca RF (2012) Surface-enhanced Raman scattering of hydroxyproline. J Raman Spectrosc 43:478–481
Brolo AG, Germain P, Hager G (2002) Investigation of the adsorption of L-Cysteine on a polycrystalline silver electrode by surface-enhanced Raman scattering (SERS) and Surface-Enhanced Second Harmonic Generation (SESHG). J Phys Chem B 106:5982–5987
Lee HI, Kim MS, Suh SW (1991) Raman spectroscopy of sulphur-containing amino acids and their derivatives adsorbed on silver. J Raman Spectrosc 22:91–96
Graff M, Bukowska J (2005) Adsorption of enantiomeric and racemic cysteine on a silver electrode—SERS sensitivity to chirality of adsorbed molecules. J Phys Chem B 109:9567–9574
Lee HI, Kim MS, Suh SW (1991) Raman spectroscopy of sulphur-containing amino acids and their derivatives adsorbed on silver. J Raman Spectrosc 22:91–96
Kühnle A, Linderoth TR, Hammer B, Besenbacher F (2002) Chiral recognition in dimerization of adsorbed cysteine observed by scanning tunneling microscopy. Nature 415:891–893
Graff M, Bukowska J (2010) Enantiomeric recognition of phenylalanine by self-assembled monolayers of cysteine: surface-enhanced Raman scattering evidence. Vibr Spectr 52:103–107
Graff M, Bukowska J (2011) Surface-enhanced Raman Scattering (SERS) spectroscopy of enantiomeric and racemic methionine on a silver electrode—evidence for chiral discrimination in interactions between adsorbed molecules. Chem Phys Lett 509:58–61
Deckert-Gaudig T, Deckert V (2009) Ultraflat transparent gold nanoplates-ideal substrates for tip-enhanced Raman scattering experiments. Small 5(4):432–436
Deckert-Gaudig T, Deckert V (2009) Tip-enhanced Raman scattering studies of histidine on novel silver substrates. J Raman Spectrosc 40:1446–1451
Stewart S, Fredericks PM (1999) Surface-enhanced Raman spectroscopy of peptides and proteins adsorbed on an electrochemically prepared silver surface. Spectrochim Acta Part A 55:1615–1640
Wei F, Zhang D, Halas NJ, Hartgerink JD (2008) Aromatic amino acids providing characteristic motifs in the Raman and SERS spectroscopy of peptides. J Phys Chem B 112:9158–9164
Herne TM, Ahern A, Garrell RL (1991) Surface-enhanced Raman spectroscopy of peptides: preferential N-terminal adsorption on colloidal silver. J Am Chem Soc 113:846–854
Podstawka E, Ozaki Y (2008) Bombesin modified 6–14 C-terminal fragments adsorption on silver surfaces: influence of a surface substrate. Biopolymers 89:941–950
Podstawka E, Niaura G, Proniewicz LM (2010) Potential-dependent studies on the interaction between phenylalanine-substituted bombesin fragments and roughened Ag, Au, and Cu electrode surfaces. J Phys Chem B 114:1010–1029
Podstawka-Proniewicz E, Kudelski A, Younkyoo Kim Y, Proniewicz L (2012) Adsorption of neurotensin-family peptides on SERS-active Ag substrate. J Raman Spectrosc 43:1196–1203
Podstawka-Proniewicz E, Andrzejak M, Kafarski P, Kim Y, Proniewicz LM (2011) Vibrational characterization of L-valine phosphonate dipeptides: FT-IR, FT-RS, and SERS spectroscopy studies and DFT calculations. J Raman Spectrosc 42(5):958–979
Małek K, Królikowska A and Bukowska J (2012) Comparative studies on IR, Raman, and Surface Enhanced Raman scattering spectroscopy of dipeptides containing ΔAla and ΔPhe. J Phys Chem B 116:1414–1425
Keating CD, Kovaleski KM, Natan MJ (1998) Protein: colloid conjugates for surface enhanced Raman scattering: stability and control of protein orientation. J Phys Chem B 102(45):9404–9413
Pavel I, McCarney E, Elkhaled A, Morrill A, Plaxco K, Moskovits M (2008) Label-free SERS detection of small proteins modified to act as bifunctional linkers. J Phys Chem C 112:4880–4883
Drachev VP, Nashine VC, Thoreson MD, Ben-Amotz D, Davisson VJ, Shalaev VM (2005) Adaptive silver films for detection of antibody-antigen binding. Langmuir 21:8368–8373
Aroca R (2006) Surface-enhanced vibrational spectroscopy. John Wiley, Chichester, pp 91–96
Deckert-Gaudig T, Bailo E, Deckert V (2009) Tip-enhanced Raman scattering (TERS) of oxidised glutathione on an ultraflat gold nanoplate. Phys Chem Chem Phys 11:7360–7362
Love JC, Estroff LA, Kriebel JK, Nuzzo RG, Whitesides GM (2005) Self-assembled monolayers of thiolates on metals as a form of nanotechnology. Chem Rev 105:1103–1169
Moyano DF, Rotello VM, (2011) Nano Meets Biology: Structure and Function at the Nanoparticle Interface. Langmuir 27:10376–10385
Królikowska A, Bukowska J (2007) Self-assembled monolayers of mercaptosuccinic acid monolayers on silver and gold surfaces designed for protein binding. Part II: vibrational spectroscopy studies on cytochrome c immobilization. J Raman Spectrosc 38:943–949
Fedurco M (2000) Redox reactions of heme-containing metalloproteins:dynamic effects of self-assembled monolayers on thermodynamics and kinetics of cytochrome c electron-transfer reactions. Coord Chem Rev 209:263–331
Hobara D, Imabayashi Si, Kakiuchi T (2002) Preferential adsorption of horse heart cytochrome c on nanometer-scale domains of a phase-separated binary self-assembled monolayer of 3-mercaptopropionic acid and 1-hexadecanethiol on Au(111). Nano Lett 2:1021–1025
Dick LA, Haes AJ, Van Duyne RP (2000) Distance and orientation dependence of heterogeneous electron transfer: a surface-enhanced resonance Raman scattering study of cytochrome c bound to carboxylic acid terminated alkanethiols adsorbed on silver electrodes. J Phys Chem B 104:11752–11762
Bonifacio A, van der Sneppen L, Gooijer C, van der Zwan G (2004) Citrate-reduced silver hydrosol modified with ö-mercaptoalkanoic acids self-assembled monolayers as a substrate for surface-enhanced resonance Raman scattering. A study with cytochrome c. Langmuir 20:5858–5864
Królikowska A, Bukowska J (2010) Surface-enhanced resonance Raman spectroscopic characterization of cytochrome c immobilized on 2-mercaptoethanesulfonate monolayers on silver. J Raman Spectrosc 41:1621–1631
Murgida DH, Hildebrandt P (2004) Electron-transfer processes of cytochrome c at interfaces. New insights by surface-enhanced resonance Raman spectroscopy. Acc Chem Res 37:854–861
Grochol J, Dronov R, Lisdat F, Hildebrandt P, Murgida DH (2007) Electron transfer in SAM/Cytochrome/Polyelectrolyte hybrid systems on electrodes: a time-resolved surface-enhanced resonance Raman study. Langmuir 23:11289–11294
Hildebrandt P, Stockburger (1986) Surface-enhanced resonance Raman spectroscopy of cytochrome c at room and low temperatures. J Phys Chem 90:6017–6024
Oellerich S, Wackerbarth H, Hildebrandt P (2002) Spectroscopic characterization of nonnative conformational states of cytochrome c. J Phys Chem B 106:6566–6580
Macdonald IDG, Smith WE (1996) Orientation of cytochrome c adsorbed on a citrate-reduced silver colloid surface. Langmuir 12:706–713
Murgida DH, Hildebrandt P (2001) Heterogenous electron transfer of cytochrome c on coated silver electrodes. Electric field effects on structure and redox potential. J Phys Chem B 105:1578–1586
Kranich A., Ly HK, Hildebrandt P, Murgida DH (2008) Direct observation of the gating step in protein electron transfer: electric-field-controlled protein dynamics. J Am Chem Soc 130:9844–9848
Yeo BS, Madler S, Schmid T, Zhang W, Zenobi R (2008) Tip-enhanced Raman spectroscopy can see more: the case of cytochrome c. J Phys Chem C 112:4867–4873
Larmour IA, Faulds K, Graham D (2010) The past, present and future of enzyme measurements using surface enhanced Raman spectroscopy. Chem Sci 1:151–160
Copeland RA, Fodor SPA, Spiro TG (1984) Surface-enhanced Raman spectra of an active flavo enzyme: glucose oxidase and riboflavin binding protein on silver particles. J Am Chem Soc 106:3872–3874
Broderick JB, Natan MJ, O’Halloran TV, Van Duyne RP (1993) Evidence for retention of biological activity of a non-heme iron enzyme adsorbed on a silver colloid: a surface-enhanced resonance Raman scattering study. Biochemistry 32:13771–13776
Wu ZS,. Zhou GZ, Jiang JH, Shen GL, Yu RQ (2006) Gold colloid-bienzyme conjugates for glucose detection utilizing surface-enhanced Raman scattering. Talanta 70:533–539
Dou X, Takama T, Yamaguchi Y, Yamamoto H, Ozaki Y (1997) Enzyme Immunoassay Utilizing Surface-Enhanced Raman Scattering of the Enzyme Reaction Product. Anal Chem 69:1492–1495
Stevenson R, Ingram A, Leung H, McMillan DC, Graham D (2009) Quantitative SERRS immunoassay for detection of human PSA. Analyst 134:842–844
Ruan CM, Wang W, Gu BH, (2006) Detection of alkaline phosphatase using surface-enhanced Raman spectroscopy. Anal Chem 78:3379–3384
Barhoumi A, Zhang D, Tam F., Halas NJ (2008) Surface-enhanced Raman spectroscopy of DNA. J Am Chem Soc 130:5523–5529
Shafer-Peltier KE, Haynes CL, Glucksberg MR, Van Duyne RP (2003) Towards a glucose biosensor based on surface-enhanced Raman scattering. J Am Chem Soc 125:588–593
Yonzon CR, Haynes CL, Zhang XY, Walsh JT, Van Duyne RP (2004) A glucose biosensor based on surface-enhanced Raman scattering: improved partition layer, temporal stability, reversibility, and resistance to serum protein interference. Anal Chem 76:78–85
Lyandres O, Shah NC, Yonzon CR, Walsh JT, Glucksberg MR, Van Duyne RP (2005) Real-time glucose sensing by surface-enhanced Raman spectroscopy in bovine plasma facilitated by a mixed decanethiol/mercaptohexanol partition layer. Anal Chem 77:6134–6139
Stuart DA, Yonzon CR, Zhang X, Lyandres O, Shah NC, Glucksberg MR, Walsh JT, Van Duyne RP (2005) Glucose sensing using near-infrared surface-enhanced Raman spectroscopy: gold surfaces, 10-day stability, and improved accuracy. Anal Chem 77:4013–4019
Zhang XY, Young MA, Lyandres O, Van Duyne RP (2005) Rapid detection of an anthrax biomarker by surface-enhanced Raman spectroscopy. J Am Chem Soc 127:4484–4489
Cheng H-W, Chen Y-Y, Lin X-X, Huan S-Y, Wu H-L, Shen G-L, Yu R-Q (2011) Surface-enhanced Raman spectroscopic detection of Bacillus subtilis spores using gold nanoparticle based substrates. Anal Chim Acta 707:155–163
Li M, Zhang J, Suri S, Sooter LJ, Ma D, Wu N (2012) Detection of adenosine triphosphate with an aptamer biosensor based on surface-enhanced Raman scattering. Anal Chem 84:2837–2842
Braun G, Lee SJ, Dante M, Nguyen T-Q, Moskovits M, Reich N (2007) Surface-enhanced Raman spectroscopy for DNA detection by nanoparticle assembly onto smooth metal films. J Am Chem Soc 129:6378–6379
Cao YC, Jin R, Mirkin CA (2002) Nanoparticles with Raman spectroscopic fingerprints for DNA and RNA detection. Science 297:1536–1540
Stokes RJ, Macaskill A, Dougan JA, Hargreaves PG, Stanford HM, Smith WE, Faulds K, Graham D (2007) Highly sensitive detection of dye-labelled DNA using nanostructured gold surfaces. Chem Commun 27:2811–2813
Faulds K, McKenzie F, Smith WE, Graham D (2007) Quantitative simultaneous multianalyte detection of DNA by dual-wavelength surface-enhanced resonance Raman scattering. Angew Chem Int Ed 46:1829–1831
Driskell JD, Kwarta KM, Lipert RJ, Porter MD (2005) Low-level detection of viral pathogens by a surface-enhanced Raman scattering based immunoassay. Anal Chem 77:6147–6154
Xu S, Ji X, Xu W, Li X, Wang L, Bai Y, Zhao B, Ozaki Y (2004) Immunoassay using probe-labelling immunogold nanoparticles with silver staining enhancement via surface-enhanced Raman scattering. Analyst 129:63–68
Yakes BJ, Lipert RJ, Bannantine JP, Porter MD (2008) Detection of mycobacterium avium subsp. paratuberculosis by a sonicate immunoassay based on surface-enhanced Raman scattering. Clin Vaccine Immunol 15:227–234
Wang G, Park HY, Lipert RJ (2009) Mixed monolayers on gold nanoparticle labels for multiplexed surface-enhanced Raman scattering based immunoassays. Anal Chem 81:9643–9650
Chourpa I, Lei FH, Dubois P, Manfaita M, Sockalingum GD (2008) Intracellular applications of analytical SERS spectroscopy and multispectral imaging. Chem Soc Rev 37:993–1000
Xie W, Su L, Shen A, Materny A, Hu J (2011) Application of surface-enhanced Raman scattering in cell analysis. J Raman Spectrosc 42:1248–1254
Kneipp J, Kneipp H, Kneipp K (2008) SERS—a single-molecule and nanoscale tool for bioanalytics. Chem Soc Rev 37:1052–1060
Vitol EA, Orynbayeva Z, Friedman G, Gogotsi Y (2012) Nanoprobes for intracellular and single cell surface-enhanced Raman spectroscopy (SERS). J Raman Spectrosc 43:817–827
Nabiev IR, Morjani H, Manfait M (1991) Selective analysis of antitumor drug interaction with living cancer cells as probed with surface-enhanced Raman spectroscopy Eur Biophys J 19:311–316
Kneipp J, Kneipp H, McLaughlin M, Brown D, Kneipp K (2006) In Vivo Molecular Probing of Cellular Compartments with Gold Nanoparticles and Nanoaggregates. Nano Letters. 6(10):2225–2231
Kneipp J, Kneipp H, Rajadurai A, Redmont RW, Kneipp K (2009) Optical probing and imaging of live cells using SERS labels. J Raman Spectrosc 40:1–5
Oyelere AK, Chen PC, Huang X, El-Sayed IH, El-Sayed MA (2007) Peptide-Conjugated Gold Nanorods for Nuclear Targeting. Bioconjugate Chem 18:1490–1497
Rodriguez-Lorenzo L, Krpetic Z, Barbosa S, Alvarez-Puebla RA, Liz-Marzan LM, Prior IA, Brust M (2011) Intracellular mapping with SERS-encoded gold stars. Integr Biol 3:922–926
Xie JP, Zhang QB, Lee JY (2008) The synthesis of SERS-active gold nanoflower tags for In Vivo applications. ACS Nano 2:2473–2480
Vitol EA, Orynbayeva Z, Bouchard MJ, Azizkhan-Clifford J, Friedman G, Gogotsi Y (2009) In Situ intracellular spectroscopy with surface enhanced Raman spectroscopy (SERS)-enabled nanopipettes. ACS Nano 3(11):3529–3536
Kneipp K, Haka AS, Kneipp H, Badizadegan K, Yoshizawa N, Boone C, Shafer-Peltier K, Motz JT, Dasari RR, Feld MS (2002) Surface-enhanced Raman spectroscopy in single living cells using gold nanoparticles. Appl Spectrosc 56:150–154
Jarvis RM, Law N, Shadi LT, O’Brien P, Lloyd JR, Goodacre R (2008) Surface-enhanced Raman scattering from intracellular and extracellular bacterial locations. Anal Chem 80:6741–6746
Efrima S, Zeiri L (2009) Understanding SERS of bacteria. J Raman Spectrosc 40:277–288
Gessner R, Winter C, Rosch P, Schmitt M, Petry R, Kiefer W, Lankers M, Popp J (2004) Identification of Biotic and Abiotic Particles by Using a Combination of Optical Tweezers and In Situ Raman Spectroscopy. ChemPhysChem 5:1159–1170
Scaffidi JP, Gregas MK, Seewaldt V, Vo-Dinh T (2009) SERS-based plasmonic nanobiosensing in single living cells. Anal Bioanal Chem 393:1135–1141
Bohme R, Richter M, Cialla D, Rosch P, Deckert V, Popp J (2009) Towards a specific characterization of components on a cell surface—combined TERS-investigations of lipids and human cells. J Raman Spectrosc 40:1452–1457
Cialla D, Deckert-Gaudig T, Budich C, Laue M, Moller R, Naumann D, Deckert V, Popp J (2009) Raman to the limit: tip-enhanced Raman spectroscopic investigations of a single tobacco mosaic virus. J Raman Spectrosc 40:240–243
Budich C, Neugebauer U, Popp J, Deckert V (2008) Cell Wall investigations utilizing tip-enhanced Raman scattering. J Microsc 229:533–539
Kozitskaya S, Deckert V, Schmitt M, Popp J (2007) Towards a detailed understanding of bacterial metabolism—spectroscopic characterization of Staphylococcus Epidermidis. ChemPhysChem 8:124–137
Auchinvole CAR, Richardson P, McGuinnes C, Mallikarjun V, Donaldson K, McNab H, Campbell CJ (2012) Monitoring intracellular redox potential changes using SERS nanosensors. ACS Nano 6(1):888–896
Kneipp J, Kneipp H, Wittig B, Kneipp K (2007) One- and two-photon excited optical pH probing for cells using surface-enhanced Raman and hyper-Raman nanosensors. Nano Lett 7:2819–2823
Talley CE, Jusinski L, Hollars CW, Lane SM, Huser T (2004) Intracellular pH sensors based on surface-enhanced Raman scattering. Anal Chem 76:7064–7068
Nowak-Lovato KL, Wilson BS, Rector KD (2010) SERS nanosensors that report pH of endocytic compartments during FcεRI transit. Anal Bioanal Chem 398:2019–2029
Kneipp J, Kneipp H, Wittig B, Kneipp K (2010) Following the dynamics of pH in endosomes of live cells with SERS nanosensors. J Phys Chem C 114:7421–7426
Zong S, Wang Z, Yang J, Cui Y (2011) Intracellular pH sensing using p-aminothiophenol functionalized gold nanorods with low toxicity. Anal Chem 83:4178–4183
Balint S, Rao S, Marro M, Miskovsky P, Petrov D (2011) Monitoring of local pH in photodynamic therapy-treated live cancer cells using surface-enhanced Raman scattering probes. J Raman Spectrosc 42:1215–1221
Bishnoi SW, Rozell CJ, Levin CS, Gheith MK, Johnson BR, Johnson DH, Halas NJ (2006) All-optical nanoscale pH meter. Nano Lett 6:1687–1692
Ando RA, Pieczonka NPW, Santos PS, Aroca RF (2009) Chromic materials for responsive surface-enhanced resonance Raman scattering systems: a nanometric pH sensor. Phys Chem Chem Phys 11:7505–7508
Qian X, Peng XH, Ansari DO, Yin-Goen Q, Chen GZ, Shin DM, Yang L, Young AN, Wang MD, Nie SM (2008) In vivo tumor targeting and spectroscopic detection with surface-enhanced Raman nanoparticle tags. Nat Biotech 26:83–90
Lee S, Kim S, Choo J, Shin SY, Lee YH, Choi HY, Ha S, Kang KH, Oh CH (2007) Biological imaging of HEK293 cells expressing PLCç1 using surface-enhanced Raman microscopy. Anal Chem 79:916–922
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Science+Business Media Dordrecht
About this chapter
Cite this chapter
Bukowska, J., Piotrowski, P. (2014). Surface-enhanced Raman Scattering (SERS) in Bioscience: A Review of Application. In: Baranska, M. (eds) Optical Spectroscopy and Computational Methods in Biology and Medicine. Challenges and Advances in Computational Chemistry and Physics, vol 14. Springer, Dordrecht. https://doi.org/10.1007/978-94-007-7832-0_3
Download citation
DOI: https://doi.org/10.1007/978-94-007-7832-0_3
Published:
Publisher Name: Springer, Dordrecht
Print ISBN: 978-94-007-7831-3
Online ISBN: 978-94-007-7832-0
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)