Abstract
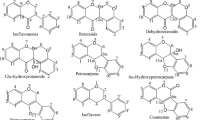
Polyphenols belonging to the class of secondary metabolites of plants and microorganisms play an important role as bioactive food constituents as well as contaminants. Structure elucidation of polyphenols in plant extracts or polyphenol metabolites, especially those arising during biotransformation, still represents a challenge for analytical chemistry. Various approaches have been proposed to utilize fragmentation reactions in connection with mass spectrometry (MS) for structural considerations on polyphenolic targets. We compiled and applied specific liquid chromatography (LC)–electrospray ionization in positive mode [ESI(+)]–tandem MS (MS/MS) and gas chromatography (GC)–(electron impact, EI)–MS/MS fragmentation reactions with a special focus on the analysis of isoflavones, whereby this technique was also found to be extendable to determine further polyphenols. For ESI(+)-MS the basic retro-Diels–Alder (rDA) fragmentation offers information about the substitution pattern in the A- and B-rings of flavonoids and the elimination of a protonated 4-methylenecyclohexa-2,5-dienone (m/z = 107) fragment can be used as a diagnostic tool for many isoflavanones. For GC-(EI)-MS/MS analysis after derivatization of the analytes to their trimethylsilyl ethers, the elimination of methyl radicals, tetramethylsilane groups or the combined loss of two methyl groups can be shown to be specific for certain substitution patterns in polyphenols. The applicability of the fragmentation reactions presented is demonstrated exemplarily for three derivatives of the isoflavone irilone. With the help of these fragmentation reactions of the two MS techniques combined, a reliable identification of polyphenols is possible. Especially in such cases where NMR cannot be utilized owing to low analyte amounts being available or prior to purification, valuable information can be obtained.







Similar content being viewed by others
Abbreviations
- AOH:
-
alternariol
- BIOA:
-
biochanin A
- BSTFA:
-
N,O-bis(trimethylsilyl)trifluoroacetamide
- DAI:
-
daidzein
- DHD:
-
dihydrodaidzein
- DHG:
-
dihydrogenistein
- EI:
-
electron impact
- ESI:
-
electrospray ionization
- ESI(+):
-
electrospray ionization in positive mode
- ESI(−):
-
electrospray ionization in negative mode
- FORM:
-
formononetin
- GC:
-
gas chromatography
- GEN:
-
genistein
- GLY:
-
glycitein
- HFM:
-
human fecal microflora
- IRI:
-
irilone
- IFA:
-
isoflavane
- IFAO:
-
isoflavanone
- IF:
-
isoflavone
- LC:
-
liquid chromatography
- MS:
-
mass spectrometry
- PRUN:
-
prunetin
- rDA:
-
retro Diels–Alder
- TMS:
-
trimethylsilyl
- ZAL:
-
zearalanol
- ZAN:
-
zearalanone
References
Bingham SA, Atkinson C, Liggins J, Bluck L, Coward A (1998) Br J Nutr 79:393–406
Kinjo J, Tsuchihashi R, Morito K, Hirose T, Aomori T, Nagao T, Okabe H, Nohara T, Masamune Y (2004) Biol Pharm Bull 27:185–188
Engelman HM, Alekel DL, Hanson LN, Kanthasamy AG, Reddy MB (2005) Am J Clin Nutr 81:590–596
van der Schouw YT, Kreijkamp-Kaspers S, Peeters PHM, Keinan-Boker L, Rimm EB, Grobbee DE (2005) Circulation 111:465–471
Wuttke W, Jarry H, Seidlova-Wuttke D (2007) Ageing Res Rev 6:150–188
Wu Q, Wang M, Simon JE (2003) J Chromatogr A 1016:195–209
Heinonen SM, Wahala K, Adlercreutz H (2004) J Agric Food Chem 52:6802–6809
Setchell KD, Brown NM, Lydeking-Olsen E (2002) J Nutr 132:3577–3584
Ruefer CE, Maul R, Donauer E, Fabian EJ, Kulling SE (2007) Mol Nutr Food Res 51:813–823
Horman I, Viani R (1971) Org Mass Spectrom 5:203–219
Smith GG, Djerassi C (1971) Org Mass Spectrom 5:487–489
Cimino CO, Shelnutt SR, Ronis MJ, Badger TM (1999) Clin Chim Acta 287:69–82
Anonymous (2006) Homöopathisches Arzneibuch. Deutscher Apotheker, Stuttgart
Kulling SE, Honig DM, Simat TJ, Metzler M (2000) J Agric Food Chem 48:4963–4972
Fabre N, Rustan I, de Hoffmann E, Quetin-Leclercq J (2001) J Am Soc Mass Spectrom 12:707–715
Barbuch RJ, Coutant JE, Welsh MB, Setchell KDR (1989) Biomed Environ Mass Spectrom 18:973–977
Antignac JP, Cariou R, Le Bizec B, Cravedi JP, Andre F (2003) Rapid Commun Mass Spectrom 17:1256–1264
Parmar VS, Sanduja SK, Jha NH, Kukla AS (1985) Org Mass Spectrom 20:265–266
Zhang L, Xu L, Xiao S-S, Liao Q-F, Li Q, Liang J, Chen X-H, Bi K-S (2007) J Pharm Biomed Anal 44:1019–1028
Creaser CS, Koupaiabyazani MR, Stephenson GR (1991) Org Mass Spectrom 26:157–160
Hvattum E, Ekeberg D (2003) J Mass Spectrom 38:43–49
March RE, Miao X-S, Metcalfe CD, Stobiecki M, Marczak L (2004) Int J Mass Spectrom 232:171–183
Rufer CE, Glatt H, Kulling SE (2006) Drug Metab Dispos 34:51–60
Locati D, Morandi S, Cupisti A, Ghiadoni L, Arnoldi A (2005) Rapid Commun Mass Spectrom 19:3473–3481
Heinonen S, Wahala K, Adlercreutz H (1999) Anal Biochem 274:211–219
Kelly GE, Nelson C, Waring MA, Joannou GE, Reeder AY (1993) Clin Chim Acta 223:9–22
Grace PB, Taylor JI, Botting NP, Fryatt T, Oldfield MF, Bingham SA (2003) Anal Biochem 315:114–21
Tekel J, Daeseleire E, Heeremans A, van Peteghem C (1999) J Agric Food Chem 47:3489–3494
Porter PM, Banwart WL, Hassett JJ (1985) Environ Exp Bot 25:229–232
Pfeiffer E, Schebb NH, Podlech J, Metzler M (2007) Mol Nutr Food Res 51:307–316
Pfeiffer E, Heyting A, Metzler M (2007) Mol Nutr Food Res 51:867–871
Creaser CS, Koupai-Abyazani MR, Stephenson GR (1992) Analyst 117:1105–1109
Heinonen SM, Hoikkala A, Wahala K, Adlercreutz H (2003) J Steroid Biochem Mol Biol 87:285–299
Woodward MD (1981) Phytochemistry 20:532–534
Castagnetta LA, Granata OM, Arcuri FP, Polito LM, Rosati F, Cartoni GP (1992) Steroids 57:437–443
Atta ur R, Nasim S, Baig I, Jalil S, Orhan I, Sener B, Choudhary MI (2003) J Ethnopharmacol 86:177–180
Acknowledgements
We acknowledge the financial support of the Studienstiftung des deutschen Volkes (Bonn, Germany) in form of a Ph.D. scholarship for N.H.S. Further we would like to thank A. Braune at the Deutsches Institut für Ernährungsforschung Potsdam-Rehbrücke for carrying out the HFM incubations and initial cleanup.
Author information
Authors and Affiliations
Corresponding author
Additional information
Ronald Maul and Nils Helge Schebb contributed equally to this work.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
ESM
(PDF 122 KB)
Rights and permissions
About this article
Cite this article
Maul, R., Schebb, N.H. & Kulling, S.E. Application of LC and GC hyphenated with mass spectrometry as tool for characterization of unknown derivatives of isoflavonoids. Anal Bioanal Chem 391, 239–250 (2008). https://doi.org/10.1007/s00216-008-1884-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-008-1884-4