Abstract
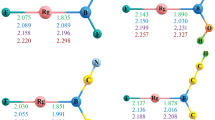
An in silico strategy of handling the thermochemical stability of XOPO(OH)2 compounds (X = F, Cl, Br) is performed by insertion of noble gas (Ng) atoms (Ng = Kr, Xe and Rn) within the X–O bond. The theoretical prediction of the set of new compounds, XNgOPO(OH)2 (X = F, Cl, Br; Ng = Kr, Xe, Rn) and their thermochemical stability are investigated using both ab initio and density-functional theory techniques considering different possible dissociation channels. The Ng (Kr–Rn) inserted analogues show that these compounds exist in their corresponding minima on their respective potential-energy surfaces. Most importantly, the release of Ng atom resulting in the formation of the bare XOPO(OH)2 and free Ng is thermochemically favorable. However, this process has very high activation energy barriers, thus kinetically protecting it from undergoing the said dissociation at room temperature. All other possible two-body and three-body ionic as well as neutral dissociation pathways are endergonic at 298 K. The generation of new Ng-based insertion compounds offers a hitherto unknown strategy of the metastable behavior of these compounds. A thorough description of the X–Ng and Ng–O bonds in XNgOPO(OH)2 compounds is provided with the help of natural bond orbital, Wiberg bond index, electron density, and energy decomposition analyses, and the more favorable representation of the compounds is proclaimed in our present discussion.



Similar content being viewed by others
References
Pauling L (1933) The formulas of antimonic acid and the antimonates. J Am Chem Soc 55:1895–1900
Bartlett N (1962) Proc Chem Soc Lond 218:197–236
Khriachtchev L, Pettersson M, Runeberg N, Lundell J, Rasänen M (2000) A stable argon compound. Nature (Lond, UK) 406:874–876
Khriachtchev L, Pettersson M, Lignell A, Rasänen M (2001) A more stable configuration of HArF in solid argon. J Am Chem Soc 123:8610–8611
Thompson CA, Andrews L (1994) Noble gas complexes with BeO: infrared spectra of Ng–BeO (Ng = Ar, Kr, Xe). J Am Chem Soc 116:423–424
Thompson CA, Andrews L (1994) Reactions of laser ablated Be atoms with O2: infrared spectra of beryllium oxides in solid argon. J Chem Phys 100:8689–8699
Evans CJ, Gerry MCL (2000) The microwave spectra and structures of Ar–AgX (X = F, Cl, Br). J Chem Phys 112:1321–1329
Evans CJ, Gerry MCL (2000) Noble gas–metal chemical bonding? The microwave spectra, structures, and hyperfine constants of Ar–CuX (X = F, Cl, Br). J Chem Phys 112:9363–9374
Evans CJ, Rubinoff DS, Gerry MCL (2000) Noble gas-metal chemical bonding: the microwave spectra geometries and nuclear hyperfine constants of Ar–AuF and Ar–AuBr. Phys Chem Chem Phys 2:3943–3948
Evans CJ, Lesarri A, Gerry MCL (2000) Noble gas–metal chemical bonds. microwave spectra, geometries and nuclear quadrupole coupling constants of Ar–AuCl and Kr–AuCl. J Am Chem Soc 122:6100–6105
Reynard LM, Evans CJ, Gerry MCL (2001) Microwave spectrum, structure and hyperfine constants of Kr–AgCl: formation of a weak Kr–Ag covalent bond. J Mol Spectrosc 206:33–40
Walker NR, Reynard LM, Gerry MCL (2002) The microwave spectrum and structure of KrAgF. J Mol Struct 612:109–116
Michaud JM, Cooke SA, Gerry MCL (2004) Rotational spectra, structures, hyperfine constants, and the nature of the bonding of KrCuF and KrCuCl. Inorg Chem 43:3871–3881
Thomas JM, Walker NR, Cooke SA, Gerry MCL (2004) Microwave spectra and structures of KrAuF, KrAgF and KrAgBr; 83Kr nuclear quadrupole coupling and the nature of noble gas–noble metal halide bonding. J Am Chem Soc 126:1235–1246
Cooke SA, Gerry MCL (2004) Insights into the xenon–silver halide interaction from a rotational spectroscopic study of XeAgF and XeAgCl. Phys Chem Chem Phys 6:3248–3256
Cooke SA, Gerry MCL (2004) XeAuF. J Am Chem Soc 126:17000–17008
Michaud JM, Gerry MCL (2006) XeCu covalent bonding in XeCuF and XeCuCl, characterised by fourier transform microwave spectroscopy supported by quantum chemical calculations. J Am Chem Soc 128:7613–7621
Seidel S, Seppelt K (2000) Xenon as a complex ligand: the tetra Xenono gold (II) cation in AuXe42+ (Sb2F11−)2. Science 290:117–118
Feldman VI, Sukhov FF, Orlov AY, Tyulpina IV (2003) Experimental evidence for the formation of HXeCCH: the first hydrocarbon with an inserted rare-gas atom. J Am Chem Soc 125:4698–4699
Khriachtchev L, Tanskanen H, Lundell J, Pettersson M, Kiljunen H, Räsänen M (2003) Fluorine-free organoxenon chemistry: HXeCCH, HXeCC, and HXeCCXeH. J Am Chem Soc 125:4696–4697
Khriachtchev L, Domanskaya A, Lundell J, Akimov A, Räsänen M, Misochko E (2010) Matrix-isolation and ab initio study of HNgCCF and HCCNgF molecules (Ng = Ar, Kr, and Xe). J Phys Chem A 114:4181–4187
Zhu C, Räsänen M, Khriachtchev L (2015) Matrix-isolation and ab initio study of HKrCCCl and HXeCCCl. J Chem Phys 143:244319
Khriachtchev L, Tanskanen H, Cohen A, Gerber RB, Lundell J, Pettersson M, Kiljunen H, R-s-nen M (2003) A gate to organokrypton chemistry: HKrCCH. J Am Chem Soc 125:6876
Tanskanen H, Khriachtchev L, Lundell J, Räsänen M (2004) Organo-noble-gas hydride compounds HKrCCH, HXeCCH, HXeCC, and HXeCCXeH: formation mechanisms and effect of 13 C isotope substitution on the vibrational properties. J Chem Phys 121:8291–8298
Arppe T, Khriachtchev L, Lignell A, Domanskaya AV, Räsänen M (2012) Halogenated xenon cyanides ClXeCN, ClXeNC, and BrXeCN. Inorg Chem 51:4398–4402
Pettersson M, Lundell J, Khriachtchev L, Räsänen M (1998) Neutral rare-gas containing charge-transfer molecules in solid matrices III. HXeCN, HXeNC, and HKrCN in Kr and Xe. J Chem Phys 109:618–625
Tanskanen H, Khriachtchev L, Lundell J, Kiljunen H, Räsänen M (2003) Chemical compounds formed from diacetylene and rare-gas atoms: HKrC4H and HXeC4H. J Am Chem Soc 125:16361–16366
Khriachtchev L, Lignell A, Tanskanen H, Lundell J, Kiljunen H, Räsänen M (2006) Insertion of noble gas atoms into cyanoacetylene: An ab initio and matrix isolation study. J Phys Chem A 110:11876–11885
Jana G, Pal R, Chattaraj PK (2021) XNgNSi (X = HCC, F; Ng = Kr, Xe, Rn): A new class of metastable insertion compounds containing Ng–C/F and Ng–N bonds and possible isomerization therein. J Phys Chem A 125:10514–10523
Willmann K, Vent-Schmidt T, Räsänen M, Riedel S, Khriachtchev L (2015) Matrix-isolation and computational study of the HKrCCH⋯HCCH complex. RSC Adv 5:35783–35791
Zhu C, Tsuge M, Räsänen M, Khriachtchev L (2015) Experimental and theoretical study of the HXeI⋯HCl and HXeI⋯HCCH complexes. J Chem Phys 142:144306
Duarte L, Khriachtchev L (2017) Matrix-isolation and theoretical study of the HXeCCXeH⋯HCCH and HXeCC⋯HCCH complexes. RSC adv 7:813–820
Pan S, Jana G, Ravell E, Zarate X, Osorio E, Merino G, Chattaraj PK (2018) Stable NCNgNSi (Ng = Kr, Xe, Rn) compounds with covalently bound C–Ng–N unit: possible isomerization of NCNSi through the release of the noble gas atom. Chem A Eur J 24:2879–2887
Mercier HPA, Breddemann U, Brock DS, Bortolus MR, Schrobilgen GJ (2019) Syntheses, structures and bonding of NgF2·CrOF4, NgF2·2CrOF4 (Ng = Kr, Xe), and (CrOF4)∞. Chem Eur J 25:12105–12119
Grochala W, Khriachtchev L, Räsänen M (2011) Noble-gas chemistry. In: Khriachtchev L (ed) Physics and chemistry at low temperatures. CRC Press, Boca Raton, pp 419–446
Eisenberg M, Desmarteau DD (1972) Xenon (II) difluorophosphates. Preparations, properties, and evidence for the difluorophosphate free radical. Inorg Chem 11:1901–1904
Musher JI (1968) Organic esters of xenon. J Am Chem Soc 90:7371–7372
Bartlett N, Wechsberg M, Jones GR, Burbank RD (1972) Crystal structure of xenon (II) fluoride fluorosulfate, FXeOSO2F. Inorg Chem 11:1124–1127
Wechsberg M, Bulliner PA, Sladky FO, Mews R, Bartlett N (1972) Fluorosulfates and perchlorates of xenon (II) and the salt [(FXeO) 2S (O) F]+[AsF6]-. Inorg Chem 11:3063–3070
Naumann D, Tyrra W, Gnann R, Pfolk DJ (1994) Synthesis of arylxenon trifluoromethanesulfonates via electrophilic substitution of F- and CF3-substituted aromatics. Chem Soc Chem Commun 22:2651–2653
Syvret RG, Schrobilgen GJ (1989) FXeOIOF4 and Xe (OIOF4) 2: preparation and study by xenon-129 and fluorine-19 NMR spectroscopy and Raman spectroscopy and NMR characterization of LXeOIOF4 (L = − OTeF5, − OSO2F). Inorg Chem 28:1564–1573
Seppelt K, Nothe D (1973) Stability of xenon (II) compounds. Pentafluorooxyselenium and pentafluorooxytellurium radicals. Bis (pentafluorotellurium) peroxide and chlorine pentafluoroorthotellurate. Inorg Chem 12:2727–2730
Seppelt K, Lentz D, Klöter G, Schack CJ (1986) Selenium tetrafluoride, selenium difluoride oxide (seleninyl fluoride), and xenon bis [pentafluorooxoselenate (VI)]. Inorg Synth 24:27–31
Sladky F (1970) Zur Chemie des Xenons, 1. Mitt.: Xenon (II)-bis (pentafluoro-orthotellurat), Xe (OTeF5)2. Monatsh Chem 101:1559–1570
Sladky F (1970) Zur Chemie des Xenons, 2. Mitt.: Xenon (II)-fluorid-pentafluoro-orthotellurat, FXeOTeF5 und das System XeF2–CF3COOH. Monatsh Chem 101:1571–1577
Schumacher GA, Schrobilgen GJ (1984) Preparation of O2XeF2-x (OTeF5) x, OXeF4-y (OTeF5) y and XeF4-y (OTeF5) y (x= 0–2, y= 0–4) and study by xenon-129 and fluorine-19 NMR and Raman spectroscopy: the oxygen primary isotopic effect in the xenon-129 NMR spectra of xenon (VI) oxyfluorides (XeO2F2 and XeOF4). Inorg Chem 23:2923–2929
Jacob E, Lentz D, Seppelt K, Simon A (1981) Edelgasverbindungen mit dem Liganden–OTeF5. Z Anorg Allg Chem 472:7–25
Moran MD, Brock DS, Mercier HP, Schrobilgen GJ (2010) Xe3OF3+, a precursor to a noble-gas nitrate; syntheses and structural characterizations of FXeONO2, XeF2·HNO3, and XeF2·N2O4. J Am Chem Soc 132:13823–13839
Catti M, Ferraris G (1974) Hydrogen bonding in the crystalline state. NaH2PO4, a crystal structure with a short symmetrical hydrogen bond. Acta Cryst. Sec B: Struct Cryst Cryst Chem 30:1–6
Bader RFW (1990) Atoms in molecules a quantum theory. Oxford University Press, Oxford
Reed AE, Weinhold F (1983) Natural bond orbital analysis of near Hartree–Fock water dimer. J Chem Phys 78:4066–4073
Glendening ED, Landis CR, Weinhold F (2013) NBO 60: Natural bond orbital analysis program. J Comput Chem 34:1429–1437
Morokuma K (1977) Why do molecules interact? The origin of electron donor-acceptor complexes, hydrogen bonding and proton affinity. Acc Chem Res 10:294–300
Ziegler T, Rauk A (1977) On the calculation of bonding energies by the Hartree Fock Slater method. Theor Chim Acta 46:1–10
Hopffgarten MV, Frenking G (2012) Energy decomposition analysis. Wiley Interdiscip Rev Comput Mol Sci 2:43–62
Zhao Y, Truhlar DG (2004) Hybrid meta density functional theory methods for thermochemistry, thermochemical kinetics, and noncovalent interactions: the MPW1B95 and MPWB1K models and comparative assessments for hydrogen bonding and van der Waals interactions. J Phys Chem A 108:6908–6918
Møller C, Plesset MS (1934) Note on an approximation treatment for many-electron systems. Phys Rev 46:618
Weigend F, Ahlrichs R (2005) Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: design and assessment of accuracy. Phys Chem Chem Phys 7:3297–3305
Cundari TR (1994) Calculation of a methane carbon-hydrogen oxidative addition trajectory: comparison to experiment and methane activation by high-valent complexes. J Am Chem Soc 116:340–347
Song J, Hall MB (1993) Theoretical studies of inorganic and organometallic reaction mechanisms. 6. Methane activation on transient cyclopentadienylcarbonylrhodium. Organometallics 12:3118–3126
Koga N, Morokuma K (1990) Ab initio potential energy surface and electron correlation effect in CH activation of methane by coordinatively unsaturated chlorodiphosphinerhodium (I). J Phys Chem 94:5454–5462
Wiberg KB (1968) Application of the pople-santry-segal CNDO method to the cyclopropylcarbinyl and cyclobutyl cation and to bicyclobutane. Tetrahedron 24:1083–1096
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Petersson GA, Nakatsuji H et al (2016) Gaussian 16, Rev. B.01; Gaussian, Inc.: Wallingford, CT
Baerends EJ, Ziegler T, Autschbach J, Bashford D, Bérces A, Bickelhaupt FM, Bo C, Boerrigter PM, Cavallo L, Chong DP, et al (2014) ADF2013.01, SCM; Theoretical Chemistry, Vrije Universiteit, Amsterdam, 2014.01
Huzinaga S, Miguel B (1990) A comparison of the geometrical sequence formula and the well-tempered formulas for generating GTO basis orbital exponents. Chem Phys Lett 175:289–291
Huzinaga S, Klobukowski M (1993) Well-tempered Gaussian basis sets for the calculation of matrix Hartree–Fock wavefunctions. Chem Phys Lett 212:260–264
Lu T, Chen F (2012) Multiwfn: a multifunctional wavefunction analyzer. J Comput Chem 33:580–592
Te Velde GT, Bickelhaupt FM, Baerends EJ, Fonseca Guerra C, van Gisbergen SJ, Snijders JG, Ziegler T (2001) Chemistry with ADF. J Comput Chem 22:931–967
Perdew JP, Burke K, Ernzerhof M (1996) Generalized gradient approximation made simple. Phys Rev Lett 77:3865
Grimme S, Antony J, Ehrlich S, Krieg H (2010) A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J Chem Phys 132:154104
Grimme S, Ehrlich S, Goerigk L (2011) Effect of the damping function in dispersion corrected density functional theory. J Comput Chem 32:1456–1465
Nemec N, Towler MD, Needs RJ (2010) Benchmark all-electron ab initio quantum Monte Carlo calculations for small molecules. J Chem Phys 132:034111
van Lenthe E, Ehlers A, Baerends EJ (1999) Geometry optimizations in the zero order regular approximation for relativistic effects. J Chem Phys 110:8943–8953
van Lenthe E, Baerends EJ, Snijders JG (1993) Relativistic regular two-component Hamiltonians. J Chem Phys 99:4597–4610
van Lenthe E, Baerends EJ, Snijders JG (1994) Relativistic total energy using regular approximations. J Chem Phys 101:9783–9792
van Lenthe E, Snijders J, Baerends E (1996) The zero-order regular approximation for relativistic effects: the effect of spin–orbit coupling in closed shell molecules. J Chem Phys 105:6505–6516
Van Lenthe E, Van Leeuwen R, Baerends E, Snijders J (1996) Relativistic regular two-component Hamiltonians. Int J Quantum Chem 57:281–293
Macchi P, Proserpio DM, Sironi A (1998) Experimental electron density in a transition metal dimer: metal–metal and metal–ligand bonds. J Am Chem Soc 120:13429–13435
Macchi P, Garlaschelli L, Martinengo S, Sironi A (1999) Charge density in transition metal clusters: supported vs unsupported metal–metal interactions. J Am Chem Soc 121:10428–10429
Cremer D, Kraka E (1984) Chemical bonds without bonding electron density ← does the difference electron-density analysis suffice for a description of the chemical bond? Angew Chem Int Ed Engl 23:627–628
Ziółkowski M, Grabowski SJ, Leszczynski J (2006) Cooperativity in hydrogen-bonded interactions: ab initio and “atoms in molecules” analyses. J Phys Chem A 110:6514–6521
Pan S, Saha R, Chattaraj PK (2015) Exploring the nature of silicon-noble gas bonds in H3SiNgNSi and HSiNgNSi compounds (Ng = Xe, Rn). Int J Mol Sci 16:6402–6418
Tonner R, Frenking G (2008) Divalent carbon (0) chemistry, part 1: parent compounds. Chem Eur J 14:3260–3272
Acknowledgements
PKC thanks 'Theoretical Chemistry Accounts' for publishing a “Festschrift" in his honor, on the occasion of his sixty-fifth birthday. He thanks Dr. Utpal Sarkar for kindly inviting him to contribute an article to the same. He also thanks DST, New Delhi, for the J. C. Bose National Fellowship, grant number SR/S2/JCB-09/2009. RP thanks CSIR for her fellowship, and GJ thanks IIT Kharagpur. We acknowledge National Supercomputing Mission (NSM) for providing computing resources of ‘PARAM Shakti’ at IIT Kharagpur, which is implemented by C-DAC and supported by the Ministry of Electronics and Information Technology and Department of Science and Technology (DST), Government of India.
Author information
Authors and Affiliations
Contributions
R.P. and G.J. conceptualized the work, performed the calculations, wrote the main manuscript text, and prepared figures and tables. P.K.C. supervised the work, edited the final manuscript. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest regarding the publication of this article, financial, and/or otherwise.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pal, R., Jana, G. & Chattaraj, P.K. Structure and stability of a new set of noble gas insertion compounds, XNgOPO(OH)2 (X = F, Cl, Br; Ng = Kr, Xe, Rn): an in silico investigation. Theor Chem Acc 142, 34 (2023). https://doi.org/10.1007/s00214-023-02973-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00214-023-02973-2