Abstract
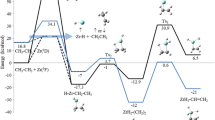
Chemiluminescence is a fundamental process of chemistry consisting in the conversion of chemical energy stored in chemical bonds into light. It is used by nature and by man-made technology, being especially relevant in chemical analysis. The understanding of the phenomenon strongly relies in the study of peroxide models such as 1,2-dioxetanones. In the present contribution, the singlet S 2 and the triplet T 2 potential energy surfaces of the unimolecular decomposition of 1,2-dioxetanone have been mapped along the O–O and C–C bond coordinates on the grounds of the multiconfigurational CASPT2//CASSCF approach. Results confirm the energy degeneracy between T 2, T 1, S 1, and S 0 at the TS region, whereas S 2 is unambiguously predicted at higher energies. Triplet-state population is also supported by the spin–orbit couplings between the singlet and triplet states partaking in the process. In particular, the first-principle calculations show that decomposition along the T 2 state is a competitive process, having a small (~3 kcal/mol) energy barrier from the ground-state TS structure. The present findings can explain the higher quantum yield of triplet-state population with respect to the excited singlet states recorded experimentally for the unimolecular decomposition of 1,2-dioxetanone models.




Similar content being viewed by others
References
Haddock SHD, Moline MA, Case JF (2010) Bioluminescence in the sea. Annu Rev Mar Sci 2:443
Contag CH, Bachmann MH (2002) Advances in vivo bioluminescence imaging of gene expression. Annu Rev Biomed Eng 4:235
Endo G, Yamagata T, Narita M, Huang CC (2003) Bioluminescence biosensor for the detection of organomercury contamination. Acta Biotechnol 23:123
Kim J-B, Urban K, Cochran E, Lee S, Ang A, Rice B, Bata A, Campbell K, Coffee R, Gorodinsky A, Lu Z, Zhou H, Kishimoto TK, Lassota P (2010) Non-invasive detection of a small number of bioluminescent cancer cells in vivo. PLoS ONE 5:e9364
Thakur MS, Ragavan KV (2013) Biosensors in food processing. J Food Sci Technol Mysore 50:625
Stannard CJ, Gibbs PA (1986) Rapid microbiology: applications of bioluminescence in the food industry—a review. J Biolumin Chemilumin 1:3–10
Specht W (1937) The chemiluminescence of hemin: an aid for finding and recognizing blood stains important for forensic purposes. Angew Chem 50:155–157
Quickenden TI, Cooper PD (2001) Increasing the specificity of the forensic luminol test for blood. Luminescence 16:251
Navizet I, Liu Y-J, Ferre N, Roca-Sanjuán D, Lindh R (2011) The chemistry of bioluminescence: an analysis of chemical functionalities. Chem Phys Chem 12:3064–3076
Schmidt SP, Schuster GB (1980) Chemiluminescence of dimethyldioxetanone. Unimolecular generation of excited singlet and triplet acetone. Chemically initiated electron-exchange luminescence, the primary light generating reaction. J Am Chem Soc 102:306–314
Schmidt SP, Schuster GB (1978) Kinetics of unimolecular dioxetanone chemiluminescence. Competitive parallel reaction paths. J Am Chem Soc 100:5559–5561
Adam W, Simpson GA, Yany F (1974) Mechanism of direct and rubrene enhanced chemiluminescence during alpha-peroxylactone decarboxylation. J Phys Chem 78:2559–2569
Adam W, Liu JC (1972) Photooxygenation (singlet oxygen) of tetrathioethylenes. J Am Chem Soc 94:1206–1209
Turro NJ, Chow MF (1980) Chemiluminescent thermolysis of alpha-peroxylactones. J Am Chem Soc 102:5058–5064
Baader WJ, Stevani CV, Bastos EL (2006) Chemiluminescence of organic peroxides. In: Rappoport Z (ed) The chemistry of peroxides. Wiley, Chichester
Adam W (1982) Determination of chemiexcitation yields in the thermal generation of electronic excitation from 1,2-dioxetanes. In: Adam W (ed) Chemical and biological generation of excited states. Academic Press, New York
Adam W, Trofimov AV (2006) Contemporary trends in dioxetane chemistry. In: Rappoport Z (ed) The chemistry of peroxides. Wiley, Chichester
Adam W, Baader WJ (1985) Effects of methylation on the thermal stability and chemiluminescence properties of 1,2-dioxetanes. J Am Chem Soc 107:410–416
Augusto FA, de Souza GA, de Souza Júnior SP, Khalid M, Baader WJ (2013) Efficiency of electron transfer initiated chemiluminescence. Photochem Photobiol 89:1299–1317
Navizet I, Roca-Sanjuán D, Yue L, Liu Y-J, Ferre N, Lindh R (2013) Are the bio- and chemiluminescence states of the firefly oxyluciferin the same as the fluorescence state? Photochem Photobiol 89:319–325
Schramm S, Weiss D, Navizet I, Roca-Sanjuán D, Brandl H, Beckert R, Goerls H (2013) Investigations on the synthesis and chemiluminescence of novel 2-coumaranones. ARKIVOC 3:174–188
Chen S-F, Navizet I, Roca-Sanjuán D, Lindh R, Liu Y-J, Ferre N (2012) Chemiluminescence of coelenterazine and fluorescence of coelenteramide: a systematic theoretical study. J Chem Theor Comput 8:2796–2807
Roca-Sanjuán D, Delcey MG, Navizet I, Ferre N, Liu Y-J, Lindh R (2012) WARNING: the light-emitting molecular structures responsible for the chemiluminescence and fluorescence phenomena are not necessarily the same! Luminescence 27:155–156
Roca-Sanjuán D, Delcey MG, Navizet I, Ferre N, Liu Y-J, Lindh R (2011) Chemiluminescence and fluorescence states of a small model for coelenteramide and cypridina oxyluciferin: a CASSCF/CASPT2 study. J Chem Theor Comput 7:4060–4069
Farahani P, Lundberg M, Lindh R, Roca-Sanjuán D (2014) CASPT2//CASSCF study of the ring-opening mechanism of dewar dioxetane. Luminescence 29:18–19
Farahani P, Roca-Sanjuán D, Zapata F, Lindh R (2013) Revisiting the nonadiabatic process in 1,2-dioxetane. J Chem Theor Comput 9:5404–5411
Aparici-Espert I, Francés-Monerris A, Rodríguez-Muñiz GM, Roca-Sanjuán D, Lhiaubet-Vallet V, Miranda MA (2016) A combined experimental and theoretical approach to the photogeneration of 5,6-dihydropyrimidin-5-yl radicals in nonaqueous media. J Org Chem 81:4031–4038
Roca-Sanjuán D, Lundberg M, Mazziotti DA, Lindh R (2012) Comment on “Density functional theory study of 1,2-dioxetanone decomposition in condensed phase.”. J Comput Chem 33:2124–2126
Yue L, Roca-Sanjuán D, Lindh R, Ferre N, Liu Y-J (2012) Can the closed-shell DFT methods describe the thermolysis of 1,2-dioxetanone? J Chem Theor Comput 8:4359–4363
da Silva LP, Esteves da Silva JC (2012) Density functional theory study of 1,2-dioxetanone decomposition in condensed phase. J Comput Chem 33:2118–2123
Augusto FA, Francés-Monerris A, Fdez. Galván I, Roca-Sanjuán D, Bastos EL, Baader WJ, Lindh R (2017) Mechanism of activated chemiluminescence of cyclic peroxides: 1,2-dioxetane and 1,2-dioxetanone. Phys Chem Chem Phys 19:3955–3962
de Oliveira MA, Bartoloni FH, Augusto FA, Monteiro Leite Ciscato LF, Bastos EL, Baader WJ (2012) Revision of singlet quantum yields in the catalyzed decomposition of cyclic peroxides. J Org Chem 77:10537–10544
Liu F, Liu Y, de Vico L, Lindh R (2009) Theoretical study of the chemiluminescent decomposition of dioxetanone. J Am Chem Soc 131(17):6181
Greenman L, Mazziotti DA (2010) Strong electron correlation in the decomposition reaction of dioxetanone with implications for firefly bioluminescence. J Chem Phys 133:164110
Greenman L, Mazziotti DA (2011) Balancing single- and multi-reference correlation in the chemiluminescent reaction of dioxetanone using the anti-Hermitian contracted Schrödinger equation. J Chem Phys 134:174110
Roca-Sanjuán D, Fdez. Galván I, Lindh R, Ya-Jun L (2015) Recent method developments and applications in computational photochemistry chemiluminescence and bioluminescence. In: Albini A, Fasani E (eds) Photochemistry, vol 42. The Royal Society of Chemistry, London
Serrano-Andrés L, Merchán M, Lindh R (2005) Computation of conical intersections by using perturbation techniques. J Chem Phys 122:104107
Serrano-Pérez JJ, Serrano-Andrés L (2012) Calculation of excited states: molecular photophysics and photochemistry on display. In: Leszczynski J (ed) Handbook of computational chemistry. Springer, Berlin
Serrano-Andrés L, Merchán M (2005) Ab initio methods for excited states. In: Olivucci M (ed) Computational photochemistry. Elsevier, Amsterdam
Roca-Sanjuán D, Francés-Monerris A, Fdez. Galván I, Farahani P, Lindh R, Liu Y-J (2017) Advances in computational photochemistry and chemiluminescence of biological and nanotechnological molecules. In: Albini A, Fasani E (eds) Photochemistry, vol 44. The Royal Society of Chemistry, London
Liu Y-J, Roca-Sanjuán D, Lindh R (2012) Computational photochemistry and photophysics: the state of the art. In: Albini A, Fasani E (eds) Photochemistry, vol 40. The Royal Society of Chemistry, London
Serrano-Andrés L, Roca-Sanjuán D, Olaso-González G (2010) Recent trends in computational photochemistry. In: Albini A (ed) Photochemistry, vol 38. The Royal Society of Chemistry, London
Malmqvist PÅ, Roos BO, Schimmelpfennig B (2002) The restricted active space (RAS) state interaction approach with spin–orbit coupling. Chem Phys Lett 357:230–240
Sergentu D-C, Maurice R, Havenith RWA, Broer R, Roca-Sanjuán D (2014) Computational determination of the dominant triplet population mechanism in photoexcited benzophenone. Phys Chem Chem Phys 16:25393–25403
Marazzi M, Mai S, Roca-Sanjuán D, Delcey MG, Lindh R, González L, Monari A (2016) Benzophenone ultrafast triplet population: revisiting the kinetic model by surface-hopping dynamics. J Phys Chem Lett 7:622–626
Sitkiewicz SP, Oliva JM, Dávalos JZ, Notario R, Saiz-López A, Alcoba DR, Oña OB, Roca-Sanjuán D (2016) Ab initio quantum–chemical computations of the electronic states in HgBr2 and IBr: molecules of interest on the Earth’s atmosphere. J Chem Phys 145:244304
Widmark PO, Malmqvist PÅ, Roos BO (1990) Density-matrix averaged atomic natural orbital (ANO) basis-sets for correlated molecular wave-functions. I. First row atoms. Theor Chim Acta 77:291
Roos BO, Lindh R, Malmqvist PÅ, Veryazov V, Widmark PO (2004) Main group atoms and dimers studied with a new relativistic ANO basis set. J Phys Chem A 108:2851–2858
Andersson K, Malmqvist PÅ, Roos BO (1992) 2nd-order perturbation-theory with a complete active space self-consistent field reference function. J Chem Phys 96:1218–1226
Finley J, Malmqvist PÅ, Roos BO, Serrano-Andrés L (1998) The multi-state CASPT2. Chem Phys Lett 228:299–306
Francés-Monerris A, Merchán M, Roca-Sanjuán D (2013) Electronic UV–Vis transient spectra of the OH reaction products of uracil, thymine, cytosine, and 5,6-dihydrouracil by using the complete active space self-consistent field second-order perturbation (CASPT2//CASSCF) theory. J Chem Phys 139:071101
Giussani A, Segarra-Martí J, Roca-Sanjuán D, Merchán M (2015) Excitation of nucleobases from a computational perspective I: reaction paths. In: Borin AC, Ullrich S, Barbatti M (eds) Photoinduced phenomena in nucleic acids I. Springer, Berlin
Roca-Sanjuán D, Aquilante F, Lindh R (2012) Multiconfiguration second-order perturbation theory approach to strong electron correlation in chemistry and photochemistry. WIREs Comput Mol Sci 2:585–603
Rubio M, Roca-Sanjuán D, Serrano-Andrés L, Merchán M (2009) Determination of the electron-detachment energies of 2′-deoxyguanosine 5′-monophosphate anion: influence of the conformation. J Phys Chem B 113:2451–2457
Segarra-Martí J, Merchán M, Roca-Sanjuán D (2012) Ab initio determination of the ionization potentials of water clusters (H2O)(n) (n = 2–6). J Chem Phys 136:244306
Ghigo G, Roos BO, Malmqvist PÅ (2004) A modified definition of the zeroth-order Hamiltonian in multiconfigurational perturbation theory (CASPT2). Chem Phys Lett 396:142–149
Forsberg N, Malmqvist PÅ (1997) Multiconfiguration perturbation theory with imaginary level shift. Chem Phys Lett 274:196–204
Roos BO, Malmqvist PÅ (2004) Relativistic quantum chemistry: the multiconfigurational approach. Phys Chem Chem Phys 6:2919–2927
Hess BA, Marian CM, Wahlgren U, Gropen O (1996) A mean-field spin–orbit method applicable to correlated wavefunctions. Chem Phys Lett 251:365–371
Christiansen O, Gauss J, Schimmelpfennig B (2000) Spin-orbit coupling constants from coupled-cluster response theory. Phys Chem Chem Phys 2:965–971
Aquilante F, De Vico L, Ferré N, Ghigo G, Malmqvist PÅ, Neogrady P, Pedersen TB, Pitonak M, Reiher M, Roos BO, Serrano-Andrés L, Urban M, Veryazov V, Lindh R (2010) Software news and update MOLCAS 7: the next generation. J Comput Chem 31:224–247
Aquilante F, Pedersen TB, Veryazov V, Lindh R (2013) MOLCAS-a software for multiconfigurational quantum chemistry calculations. WIREs Comput Mol Sci 3:143–149
González-Luque R, Climent T, González-Ramírez I, Merchán M, Serrano-Andrés L (2010) Singlet–triplet states interaction regions in DNA/RNA nucleobase hypersurfaces. J Chem Theor Comput 6:2103–2114
de Vico L, Liu Y-J, Krogh JW, Lindh R (2007) Chemiluminescence of 1,2-dioxetane reaction mechanism uncovered. J Phys Chem A 111:8013–8019
Farahani P, Lundberg M, Lindh R, Roca-Sanjuán D (2015) Theoretical study of the dark photochemistry of 1,3-butadiene via the chemiexcitation of dewar dioxetane. Phys Chem Chem Phys 17:18653–18664
Acknowledgements
This research was supported by Projects CTQ2014-58624-P of the Spanish MINECO/FEDER. D.R.-S. thanks the “Juan de la Cierva” program of the Spanish MINECO (Ref. JCI-2012-13431). A.F.-M. thanks BES-2011-048326 FPI Grant and the EEBB-I-14-08821 mobility program (MINECO). I.F.G. and R.L. acknowledge financial support from the Swedish Research Council (Grant No. 2012-3910), the eSSENCE program, and Uppsala University.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Published as part of the special collection of articles derived from the 10th Congress on Electronic Structure: Principles and Applications (ESPA-2016).
Rights and permissions
About this article
Cite this article
Francés-Monerris, A., Fdez. Galván, I., Lindh, R. et al. Triplet versus singlet chemiexcitation mechanism in dioxetanone: a CASSCF/CASPT2 study. Theor Chem Acc 136, 70 (2017). https://doi.org/10.1007/s00214-017-2095-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00214-017-2095-x