Abstract
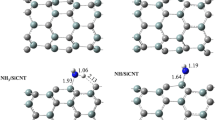
The reactivity of H2O and the Si-terminated silicon carbide surface (001) was investigated on the triplet potential energy surface with the combined first principle and molecular mechanics ONIOM(CASSCF:AM1:UFF) method for the (SiC)192·H2O model. It was found that the H2O molecule and the surface can form three physisorption complexes and follow five reaction paths to produce eight products, in which there are five main products having necessary energy barriers less than 300 kJ mol−1. Compared with that on the C-terminated surface, the interaction with the Si-terminated surface has stronger physisorption energy, smaller lowest necessary energy barrier, more main and more stable products.



Similar content being viewed by others
References
Liu Y, Su KH, Wang X, Wang YL, Zeng QF, Cheng LF, Zhang LT (2010) Chem Phys Lett 501:87
Cicero G, Galli G, Catellani A (2004) J Phys Chem B 108:16518
Cicero G, Galli G, Catellani A (2004) Phys Rev Lett 93:016102/1
Wang JJ, Zhang LT, Zeng QF, Vignoles LG, Guette A (2009) Chin Sci Bull 54:1487
Wang JJ, Zhang LT, Zeng QF, Vignoles LG, Cheng LF, Guette A (2009) Phys Rev B 79:125304/1
Maeda M, Nakamura K, Ohkubo T (1988) J Mater Sci 23:3933
Wang JJ, Zhang LT, Zeng QF, Vignoles LG, Guette A (2008) J Am Ceram Soc 91:1665
Bermudez VM (1989) J Appl Phys 66:6084
Bermudez VM (1995) Appl Sur Sci 84:45
Amy F, Chabal YJ (2003) J Chem Phys 119:6201
Ventra MD, Pantelides ST (1999) Phys Rev Lett 83:1624
Wu SJ, Cheng LF, Zhang LT, Xu YD, Zhang Q (2006) Mater Sci Eng B 130:215
Fukushima M, Zhou Y, Yoshizawa YI, Hirao K (2008) J Eur Ceram Soc 28:1043
Okawa T, Fukuyama R, Hoshino Y, Nishimura T, Kido Y (2007) Sur Sci 601:706
Voegeli W, Akimoto K, Urata T, Nakatani S, Sumitani K, Takahashi T, Hisada Y, Mitsuoka Y, Mukainakano S, Suguyama H, Zhang XW, Kawata H (2007) Sur Sci 601:1048
Hoshino Y, Okawa T, Shibuya M, Nishimura T, Kido Y (2008) Sur Sci 602:3253
Li SW, Feng ZD, Mei H, Zhang LT (2008) Mater Sci Eng A 487:424
Liu CD, Cheng LF, Mei H, Luan XG (2009) Ceram Int 35:1397
Yin XW, Cheng LF, Zhang LT, Xu YD (2003) Mater Sci Eng A 348:47
Eaton HE, Linsey GD (2002) J Eur Ceram Soc 22:2741
Irene EA, Ghez R (1977) J Electrochem Soc 124:1757
Opila EJ (1999) J Am Ceram Soc 82:625
Opila EJ Jr, Hann RE (1997) J Am Ceram Soc 80:197
Opila EJ, Fox DS, Jacobson NS (1997) J Am Ceram Soc 80:1009
Opila EJ, Smialek JL, Robinson RC, Fox DS, Jacobson NS (1999) J Am Ceram Soc 82:1826
Dapprich S, Komáromi I, Byun KS, Morokuma K, Frisch MJ (1999) J Mol Struct (Theochem) 461–462:1
Svensson M, Humbel S, Froese RDJ, Matsubara T, Sieber S, Morokum K (1996) J Phys Chem 100:19357
Svensson M, Humbel S, Morokuma K (1996) J Chem Phys 105:3654
Tamura H, Gordon MS (2003) J Chem Phys 119:10318
Hegarty D, Robb MA (1979) Mol Phys 38:1795
Eade RHA, Robb MA (1981) Chem Phys Lett 83:362
Schlegel HB, Robb MA (1982) Chem Phys Lett 93:43
Bernardi F, Bottini A, McDougall JJW, Robb MA, Schlegel HB (1984) Far Symp Chem Soc 19:137
Yamamoto N, Vreven T, Robb MA, Frisch MJ, Schlegel HB (1996) Chem Phys Lett 250:373
Frisch MJ, Ragazos IN, Robb MA, Schlegel HB (1992) Chem Phys Lett 189:524
Dewar M, Thiel W (1977) J Am Chem Soc 99:4499
Dewar MJS, McKee ML, Rzepa HS (1978) J Am Chem Soc 100:3607
Dewar MJS, Zoebisch EG, Healy EF (1985) J Am Chem Soc 107:3902
Rappé AK, Casewit CJ, Colwell KS, Goddard WA III, Skiff WM (1992) J Am Chem Soc 114:10024
Maseras F, Morokuma K (1995) J Comput Chem 16:1170
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA Jr, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas O, Foresman JB, Ortiz JV, Cioslowsk J, Fox DJ (2009) Gaussian 09, Revision A.02, Gaussian Inc., Wallingford, CT
Pollmann J, Kruger P (2004) J Phys Condens Matt 16:S1659
Powers JM, Wander A, Van Hove MA, Somorjai GA (1992) Surf Sci Lett 260:L7
Foster JP, Weinhold F (1980) J Am Chem Soc 102:7211
Reed AE, Weinstock RB, Weinhold F (1985) J Chem Phys 83:735
Acknowledgments
Part of the calculations was performed in the High Performance Computation Center of the Northwestern Polytechnical University. Supports by the National Natural Science Foundation of China (No. 50572089) and the Chinese 973 Fundamental Researches are greatly acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, Y., Su, KH., Zeng, QF. et al. Reactivity of H2O and the Si-terminated surface of silicon carbide studied with ONIOM method. Theor Chem Acc 131, 1101 (2012). https://doi.org/10.1007/s00214-012-1101-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00214-012-1101-6