Abstract
We discuss Ab Initio approaches to calculate the energy lowering (stabilisation) due to aromaticity. We compare the valence bond method and the block-localised wave function approaches to calculate the resonance energy. We conclude that the valence bond approach employs a Pauling–Wheland resonance energy and that the block-localised approach employs a delocalisation criterion. The latter is shown to be more basis set dependent in a series of illustrative calculations.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
The extra-energetic stabilisation of aromatic compounds, e.g. benzene, compared to their non-aromatic counterparts is a subject of considerable interest in chemistry. Many approaches for its calculation have been suggested in the literature, both empirical [1–3] and employing Ab Initio quantum chemistry [4–8]. Historically, Pauling and Wheland [9] have defined the resonance energy of an aromatic compound as the difference in energy between the resonating multi-structure valence bond wave function and the lowest contributing structure. This definition has been widely used by VB calculations [5–8]. Pauling and Sherman [10] have also published another way to calculate the resonance energy from thermochemical data. These can be obtained by using a hypothetical, non-aromatic molecule 1,3,5-cyclohexatriene in a relaxed, D3h symmetry and comparing it to the real benzene. It is also possible to mimic the thermochemical approach computationally, by calculating the parts of a molecule (non-aromatic counterpart) and the whole (real molecule). That has been defined in valence bond as a theoretical resonance energy (TRE) [7]—the energy difference between the resonating, multi-structure VB wave function used for the description of benzene (real molecule) and the single structure VB wave function that describes its non-aromatic counterpart 1,3,5-cyclohexatriene in a relaxed D3h symmetry. In contrast, in the block-localised wave function (BLW) [11–13] approach, the resonance energy is defined as the energy that is gained by the delocalisation of the localised constituent parts (blocks), computed using a BLW wave function (non-aromatic counterpart), to the delocalised molecule, calculated using the Hartree–Fock wave function (real molecule). In the BLW approach, the delocalisation energy is thus calculated. Of course other methods like DFT could be used as well for BLW, instead of Hartree–Fock [14, 15]. In any case, the hypothetical molecules are only accessible computationally by restricting the variational space in which the wave function is expanded or by the use of fixed (non-optimised) orbitals. Pauling and Wheland [9] comment on the Pauling and Sherman [10] approach in their paper: “the resonance energy calculated by Pauling and Sherman is not strictly comparable with that found by the present method”.
The proposed Pauling–Wheland and Pauling–Sherman approaches are two completely different ways to consider the extra-stabilisation energies of aromatic compounds, which are often rather loosely mixed [11, 13], but not strictly comparable, due to the different basic approaches. When comparing a non-aromatic system, like three ethene molecules, with its aromatic counterpart benzene, both effects may be invoked to explain the extra-stabilisation (vide infra).
In this paper, we study and compare the basis set dependence of both approaches. Benzene is the archetype of aromatic molecules, and its aromatic stabilisation is still a matter of dispute [7, 11, 13]. To illustrate our considerations, we compare the resonance energies, obtained using the BLW approach and the VB approach, using different orbital models i.e. VB-local and VB-delocal [6–8]. The interpretation of the obtained results is discussed.
To illustrate the basis set dependence of the results, we performed calculations on H6 in a benzene arrangement. For this system, a wide range of basis sets of increasing size may be used. We present both BLW and VB results and show that especially the former shows a significant basis set dependence, as do the VB-local results. In contrast, the VB calculations without restrictions on the one-electron space (VB-delocal) are shown to be remarkably independent of the one-electron basis.
2 BLW versus VB
In the VB picture, the extra-stabilisation is brought upon by the mixing of different valence bond structures, e.g. in the case of benzene, the mixing of primarily the two Kekulé structures (Fig. 1).
In the BLW approach, the energy of the most stable resonance structure is calculated the way in which the primitive basis functions are partitioned into several subgroups, and each block-localised orbital is expanded in only one subgroup. The energy of the real molecule is evaluated using the Hartree–Fock method (Fig. 2). Consequently, the energy difference between the HF wave function, where all electrons are free to delocalise in the whole system, and the block-localised wave function, where electrons are confined to specific zones of the system, can be defined generally as the electron delocalisation energy [11, 13, 16, 17]. Note the similarity between the BLW approach and the calculation of the resonance energy of benzene using Hückel theory, where the resonance energy is defined as the energy difference between three doubly occupied ethene bonds and the ‘true’ Hückel benzene energy.
There is, however, a parallel between the ways the delocalisation energy and the resonance energy are calculated. Both the BLW and the VB approach rely on a restriction of the variational space, in order to define the subunits for which the energies are calculated, which are subsequently compared to that of the real system.
In the BLW approach, the orbital space available to each subunit is restricted to the orbitals of that subunit, i.e. the atomic orbitals of the bonding atoms. This BLW approach suffers from a dependence on the one-electron basis, as it is used to define the restriction. The same applies to our strictly atomic (VB-local) model (vide infra) [18]. These orbital restrictions are untenable in the limit of a complete basis set [7], which for the sake of argument may be chosen to be centred on just one atom of the subunit. In that case, the calculated delocalisation energy will be zero as both the subunit and the molecule share the same basis. The delocalisation energy for the other subunits in the molecule will defy calculation, as no orbitals are available to describe its wave function. Of course part of the basis may be assigned to each subunit, and then the result will depend on the choices made. Thus, the calculation of the delocalisation energy is by the design dependent on the chosen partition of the one-electron basis set.
The VB approach suffers from no such dependence on the one-electron basis. Even in the limit of a complete one-atom basis, the resonance energy will be retained. This does not imply that it does not depend on the basis set at all as it still has the normal dependence on the basis any calculation has. However, as the restriction used to determine the resonance energy now is at the many-electron level, an extension of the reference model beyond a simple VB model will give n-electron basis–dependent results. As a consequence, the limit of a full CI (full VB) description of the subunit will be identical to the description of the real molecule, resulting in zero resonance energy in this case. Thus, any definition of resonance energy or delocalisation energy will depend on a restriction of the variational space. Still, other choices have been made in the literature to describe 1,3,5-cyclohexatriene and benzene, in order to estimate the extra-stabilisation of benzene [19, 20].
Whatever model is chosen for the description of the molecule and its localised counterpart, it is imperative that the answers obtained are not too heavily dependent on the chosen one-electron basis set. The different electron space restrictions in case of BLW (one-electron basis) and VB (n-electron basis) determine the difference between the delocalisation and resonance energies and should not be mixed [11, 13].
The resonance energies are all evaluated at one geometry, usually the geometry of the real molecule, and are referred to in the literature, either as the vertical resonance energy (VRE) [21] as in block-localised wave function approach or as the Pauling–Wheland resonance energy (PRE) within our VBSCF method.
3 Calculations
All calculations were performed with TURTLE [22], as implemented in GAMESS-UK [23]. All VB calculations have a multi-structure character, and all the energies obtained come from the same calculations. For comparison, various basis sets were used for benzene BLW and VB calculations, in order of increasing number of orbitals 6-31G [24] (66 AOs), 6-311+G** [25] (174 AOs), aug-cc-pVTZ [26] (474 AOs) and aug-cc-pVQZ [26] (954 AOs). Two different orbital models were used for the VB calculations: (1) the strictly atomic model (VB-local), in which the p π-orbitals are restricted to remain localised on one atom and (2) the delocal model (VB-delocal), where the p π-orbitals are not restricted at all. The VB wave functions of benzene consisted of the two Kekulé structures and the three Dewar structures. The benzene geometry used throughout all BLW and VB calculations was optimised at the RHF/6-311+G** [25] level with the R C–C bond lengths equal to 1.386 Å and was applied to both the real molecules and their non-aromatic counterparts. All σ-orbitals of benzene were kept frozen during the BLW and VB calculations and were taken from a preceding RHF calculation.
In the case of H6, we used a hexagonal arrangement with a fixed H–H bond length of 0.74 Å. The basis sets used were, in order of increasing number of orbitals, 6-31G [24] (12 σ AOs), 6-311++G** [25] (36 σ AOs), aug-cc-pVTZ [26] (108 σ AOs) and a specially engineered ANO type basis ANO-3s2p1d1f1 g [27] (156 σ AOs).
4 Results and discussion
4.1 Benzene
Table 1 reports the vertical resonance energies (VRE) as obtained using the BLW description vs. Hartree–Fock benzene [11–13], and the Pauling–Wheland resonance energies (PRE) using the VB by multi-structure (Kekulé + Dewar) calculations, with different orbital optimisation models [7, 8].
The BLW VRE is significantly higher than the VB PRE ones. Apparently, this is due to the heavy restriction imposed on the one-structure calculation. The BLW one-structure has an energy (E BLW-1) that is even higher than the VB-local (E VB-local-1). The VB-delocal calculations give the lowest total energies for both benzene and the single structure. The interpretation of the different wave functions is straightforward: the block-localised wave function corresponds to a Hartree–Fock-like description of both compounds, while the valence bond delocal wave functions include static correlation and resemble MCSCF wave functions for both compounds.
A comprehensive overview of different approaches to calculate the resonance energy [13] compares various values obtained for benzene. Our reported PREs of benzene (Table 1) differ significantly from the literature values; however, the very different nature of all the techniques, reported here, prohibits a strict comparison. The PREs of benzene from the VB-delocal calculations suggest an extra-stabilisation of approximately 20 kcal/mol for benzene with respect to the single Kekulé structure description. In the delocal model, the one-structure description can be interpreted chemically as a (closed) polyene-like reference: delocalisation of the atomic orbitals to neighbouring atoms still occurs. This interpretation is further corroborated by the results of geometry optimisation of benzene using a single-structure wave function: alternating bond lengths of 1.369 and 1.433 Å are obtained [7], which are close to the bond lengths found in for example 1,3-butadiene.
4.2 Basis set dependence
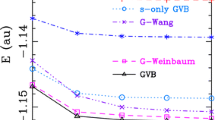
To study the basis set dependence of the various resonance energies, we have calculated the resonance energies for hexagonal H6. In principle, restrictions on the one-electron basis vanish for a complete basis set, and even a one-centre basis set expansion would suffice to describe a molecule. Thus, the completeness of the one-electron basis can be probed by the calculation of the energy of H2, where the basis set is expanded on only one hydrogen atom. In Table 2, the energies of H2 are presented for different basis sets. For the biggest basis, the error in the one-centre description of H2 is only ca. 13 kcal/mol. The better the one-centre expansion for H2 is, the smaller is the difference in energy between a one- and two-centre basis set expansion. In the limit of a complete set, that is when the Hartree–Fock limit is reached for the one-centre expansion on H2, this difference will be zero.
We present the resonance energies for hexagonal H6 in Table 3. The ANO basis set [27] was engineered in such a way that it would give a reasonable description of the H2 molecule with the basis set expanded on only one hydrogen atom. If the bonding would be recovered using this basis set and the one-centre expansion in H2, the basis would also recover the bonding in H6 while the restrictions of the BLW wave function are in effect; hence, the use of this basis set exemplifies the convergence of the BLW energy to the HF energy in case of large (complete) basis sets. The BLW values show a similar dependence on the chosen basis set as we find in the H2 one-centre expansion case. In the limit of a complete one-electron basis set, the BLW wave function has to converge to the benzene solution, with the (undesirable) result that the resonance energy approaches zero!
The VB-local resonance energies show a matching behaviour, though they do not approach zero. Ultimately, they have to converge to the results obtained by the VB-delocal approach instead. The only approach yielding consistent resonance energies over the whole range of basis sets is the VB-delocal approach. The delocal VB resonance energies are indeed remarkably basis independent. The VB-delocal model will still give a non-zero (Pauling–Wheland) resonance energy in the limit, as the required spin-couplings schemes to get the benzene wave function are absent in the one-structure wave function.
The basis dependence is already obvious from the differences between BLW and RHF energies. The former changes in the first decimal when the basis is changed, whereas the latter changes only in the second or third decimal place. As the difference between them defines the delocalisation energy, its dependence on the basis is obvious.
5 Conclusions
The delocalisation, resonance energy obtained using the BLW approach, is determined by the incompleteness of the chosen one-electron basis set, and consequently will be one-electron basis set dependent. The highlighted extreme cases of ANO basis set for H2 one-centre and H6 or the aug-cc-pVQZ for benzene, though not used very often in VB calculations, show the expected problem of reduction in the resonance energy, due to the fact that the applied orbital restrictions become meaningless in the case of a near-complete basis set. Therefore, different models used to describe this reference will give different results. The VB-delocal (vertical) resonance energies are almost independent of the basis set. However, employing a realistic basis set and “as long as the atomic characteristics are well retained in the basis functions”, the BLW will generate stable results [13]. We infer that the resonance energy definition using the BLW approach [11, 13] does not seem comparable with the Pauling and Wheland concept [9] and should be actually referred to as delocalisation energy.
6 Comment on “How to properly compute the resonance energy within the ab initio valence bond theory?”—a following publication in this issue
Mo, Hiberty and Schleyer [28] (MHS hereafter) have presented various approaches to calculate the resonance energy and came to many conclusions and findings including a computational way of calculating the resonance energy with the block-localised wave function method [11, 12] (BLW), finding the compression energy (\( \Updelta E_{c} \)) or lack of any significant basis set dependency in the BLW resonance energy definition. We would like to take this opportunity to comment on some of these findings and present our point of view.
It is important to realise first that resonance energy is not an observable property, and thus, there is no way to define this property unequivocally using quantum mechanics, which means that slightly different definitions and interpretations of these definitions may lead to completely different results and conclusions, but nature does not provide a mechanism to decide who is right or wrong. In our method, we try to adhere as strictly as possible to the Pauling and Wheland [9] definition using modern valence bond techniques.
Secondly, we stress the difference between the Pauling–Wheland’s resonance energy (PRE) and the vertical resonance energy (VRE), while MHS call Pauling–Wheland a VRE method. We do not understand this and refer for this and the original definition to the original paper [9]. We considered the VRE before as well [7, 29]. The Pauling–Wheland’s concept of the resonance energy, in the case of the benzene, is “the extra energy of the molecule resulting from resonance among the five independent structures”, which in other words mean an energy difference between the multi-structure (e.g. two Kekulé and three Dewar structures in the case of benzene) resonating VB wave function and the energy calculated for the most stable structure (Kekulé structure), both coming from the very same calculations. In the case of VRE, both energies come from separate calculations. The authors repeatedly say [11, 13, 28] that the Pauling–Wheland’s resonance energy is the vertical resonance energy, hence misinterpreting our VBSCF PRE results. Moreover, any reference to the relation between the VRE and the adiabatic (theoretical) resonance energy (ARE/TRE) is purely artificial and not the subject of our publication. For that, we refer to our previous article [7].
We are willing to concede that the BLW approach is not the Pauling–Sherman [10] recipe, but it is definitely not Pauling–Wheland either. We were led astray by the willingness Mo et al. showed to include external improvements.
Going further in that direction, according to MHS, the delocalisation energy and the resonance energy are the same. We realise that this misunderstanding is widespread, but we think the discussion should be about the resonance. As stated many times, we use the Pauling–Wheland’s definition [9]. This is an n-electron (correlation) effect, whereas the delocalisation is a one-electron effect. They may point in the same direction, but they are not the same. MHS use a (de)localisation approach in the BLW method, which determines their definition of the resonance energy. It is possible to use real resonance with BLW functions, as some of us have demonstrated [29], and the resulting Pauling–Wheland’s resonance energy is almost like our VBSCF PRE. They call their resonance energy misleadingly the Pauling–Wheland resonance energy, though without a reference, which is even more confusing.
MHS state in their paper that atoms are defined by basis sets [28]. However, this is not real; plane wave basis sets are equally capable of describing atoms and molecules, and this does not negate the concept of atoms, as it is the VB orbitals that should exhibit the atomic character. We realise that employing big-sized basis sets make the identification more cumbersome, but identifying basis sets with atoms is not based on theory. The idea of possible infinite basis sets, one centre or otherwise, is at the basis of Ab Initio quantum chemistry.
Furthermore, it is clear that MHS missed the difference between a one-electron basis set and a n-electron basis set: the former is one-electron functions, such as atomic orbitals or molecular orbitals, whereas the latter is n-electron functions, such as determinants, configuration state functions or structures. Deficiencies in one set can never be remedied by expansion of the other set. The fact that MHS say that correlation is unimportant shows that they miss the point. Resonance in the Pauling–Wheland definition is correlation. The fact that they consider correlation a minor correction to basically a Hartree–Fock function indicates that their theory is a one-electron (Hartree–Fock or DFT) theory. The explicitly correlated basis functions [30] to which MHS refer are two-electron functions, and we have not used them yet in our VB methodology.
The results reported in [31] are the Ab Initio valence bond results. Perhaps the “sophisticated analysis of experimental and computational data’’ produces something that is not the resonance energy. Our results match those of Cooper et al. [32] as they should do because both spin-coupled VB [8] and VBSCF [18, 33] are in essence the same. They are related to resonance, since they are calculated from a wave function with resonance. The experimental results definitely do not contain explicit resonance and the computational results probably not either, since they surely do not include Cooper et al. and only quote Mo himself. So, our calculations do not agree with Mo’s calculations and that was, we thought, rather obvious.
Using the VB-local method, the real benzene molecule is poorly described, but it has the advantage of allowing some interpretation, though it is not as obvious as it might seem due to the arbitrary assignment of basis functions to atomic orbitals. If we employ the delocal model (the spin-coupled model), the wave function is much better, as it includes now the ionic structures, but the PRE is even smaller. To get the largest (but we are not aiming for that) resonance energy, we can do a full π-CI (one can still use VB, but an orthogonal calculation gives the same answer) and include a Kekulè structure with local atomic orbitals. Then, the resonance energy is \( E_{kekule\,atomic} \) (−230.480868 Hartree)–\( E_{full\,\pi - CI} \) (−230.709822 Hartree), which is 143.7 kcal/mole, all in the same basis (6-31G). So we can get any number we like between 20 and 143 kcal/mol, but as we said, we are not aiming for that.
Compression energy of the hypothetical 1,3,5-cyclohexatriene was also calculated by some of us [7] and represented in the thermocycle for benzene. The authors’ remark that “VRE of benzene must be at least 30 kcal/mole larger than its ARE” is not valid. Since “optimal” or “rigid” 1,3,5-cyclohexatriene is a hypothetical system, the calculated compression energy depends on the way one captures this system in computro. In our CPL paper, we used VB, and we find consistent results for PRE, VRE and TRE. The compression energy of 1,3,5-cyclohexatriene calculated with BLW method cannot be compared with the one calculated with the VB method, as the two approaches are totally different.
Unfortunately, MHS have missed the main point of our paper [31]; in order to calculate a resonance energy, the energy of a, even in computro, non-existing reference molecule has to be calculated. This is only possible by posing limits on the expansion space of the real molecule. This can be done in several ways, each with their own advantages and disadvantages. One way is to limit the one-electron space (limiting the expansion freedom of the orbitals in the calculation) leading to the BLW approach [11, 12] and results in a delocalisation energy comparable to the Hückel method, and the other way is to limit the n-electron space (limiting the number of determinants (structures) in which the wave function is expanded), resulting in a resonance energy according to the Pauling–Wheland definition. The disadvantage of restricting the freedom of the orbitals (one-electron space) is that enlarging the basis set leads necessarily to a delocalisation energy of zero, whereas in the same limit of a complete one-electron basis set, the resonance energy calculated using the VB method (where the number of structures is limited) converges to a finite, non-zero limit. The only reason we did our H6 case is that we tried to approach a limit, like we used to do in Ab Initio quantum chemistry. The basis set dependence of the BLW method is thus clearly different from the VB behaviour. MHS cannot be in disagreement with this statement, because they also admit that they prefer to restrict the BLW calculations to medium-sized basis sets.
It is important to note that both methods have their place in computational chemistry and that neither can be qualified as being either right or wrong. We actually used the BLW concept and showed [29] that, if properly optimised, the doubly occupied BLW functions can take the place of the singlet-coupled singly occupied orbitals in a really resonating set of structures. Again, the use of large basis sets does not pose a problem, as it is the VB orbitals that should be analysed. The restriction to medium-sized basis sets does not allow the standard quantum chemical approach to employ as big basis as possible to test the stability of the answers when increasing the basis. In the past, this has revealed real artefacts [34, 35], and we should not be afraid to go there.
Finally, the MHS’ remarks about “Norbeck and Gallup’s calculated resonance energies to be underestimated” and the reference to the “true energy of the reference Kekulé structure” imply incorrectly that the resonance energy is a measurable property. Our remark [31] about the “very different nature” of BLW and VB was just trying to be friendly towards BLW.
References
Kistiakowsky GB, Ruhoff JR, Smith HA, Vaughan WE (1936) J Am Chem Soc 58:146–153
Dewar MJS, De Llano C (1969) J Am Chem Soc 91:789–795
Slayden SW, Liebmann JF (2001) Chem Rev 101:1541–1566
Shaik S, Shurki A, Danovich D, Hiberty PC (2001) Chem Rev 101:1501–1539
Tantardini GF, Raimondi M, Simonetta M (1977) J Am Chem Soc 99:2913–2918
Dijkstra F, van Lenthe JH, Havenith RWA, Jenneskens LW (2003) Int J Quantum Chem 91:566–574
van Lenthe JH, Havenith RWA, Dijkstra F, Jenneskens LW (2002) Chem Phys Lett 361:203–208
Cooper DL, Gerratt J, Raimondi M (1986) Nature 323:699–701
Pauling L, Wheland GW (1933) J Chem Phys 1:362–374
Pauling L, Sherman J (1933) J Chem Phys 1:606–617
Mo Y, Schleyer PVR (2006) Chem Eur J 12:2009–2020
Mo Y, Peyerimhoff SD (1998) J Chem Phys 109:1687–1697
Mo Y (2009) J Phys Chem A 113:5163–6169
Mo Y (2003) J Phys Chem A 119:1300–1306
Mo Y, Song L, Lin Y (2007) J Phys Chem A 111:8291–8301
Kerber RC (2006) J Chem Educ 83:223–227
Truhlar DG (2007) J Chem Educ 84:781–782
van Lenthe JH, Balint-Kurti GG (1983) J Chem Phys 78:5699–5713
Hiberty PC, Byrman CP, van Lenthe JH (1994) J Chem Phys 101:5969–5976
Hiberty PC (1997) THEOCHEM 35:398–399
Mo Y, Wu W, Zhang Q (1994) J Phys Chem 98:10048–10053
Verbeek J, Langenberg JH, Byrman CP, Dijkstra F, Engelberts JJ, Zielinski ML and van Lenthe JH (1988–2010) TURTLE, an ab initio VB/VBSCF program Utrecht, The Netherlands
Guest MF, Bush IJ, van Dam HJJ, Sherwood P, Thomas JMH, van Lenthe JH, Havenith RWA, Kendrick J (2005) Mol Phys 103:719–747
Hehre WJ, Ditchfield R, Pople JA (1972) J Chem Phys 56:2256–2261
Krishnan R, Binkley JS, Seeger R, Pople JA (1980) J Chem Phys 72:650–654
Dunning TH Jr (1989) J Chem Phys 90:1007–1023
Havenith RWA (2007) Specially designed ANO -type basis. Private communication
Mo Y, Hiberty PC, Schleyer PvR (2010) Theo Chem Acc following paper in this issue
Zielinski ML, van Lenthe JH (2008) J Phys Chem A 112:13197–13202
Klopper W, Manby FR, Ten-No S, Valeev EF (2006) Int Rev Phys Chem 25:427–468
Zielinski ML, Havenith RWA, Jenneskens LW, van Lenthe JH (2010) Theo Chem Acc This publication
Cooper DL, Gerratt J, Raimondi M (1990) Topics Current Chem 153:41–55
van Lenthe JH, Balint-Kurti GG (1980) Chem Phys Lett 76:138–142
Ransil BJ (1960) J Chem Phys 34:2109–2118
Kestner NR (1968) J Chem Phys 48:252–257
Acknowledgments
R. W. A. H. acknowledges financial support from the Netherlands Organisation for Scientific Research (NWO), grant 700.53.401. M. Z. acknowledges financial support from the Netherlands Organisation for Scientific Research (NWO-CW), ECHO-project grant 700.55.030. We gratefully acknowledge NWO/NCF for supercomputer time on TERAS/ASTER/HUYGENS, SARA (the Netherlands, project numbers SG-032, SH-028).
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Zielinski, M., Havenith, R.W.A., Jenneskens, L.W. et al. A comparison of approaches to estimate the resonance energy. Theor Chem Acc 127, 19–25 (2010). https://doi.org/10.1007/s00214-010-0793-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00214-010-0793-8