Abstract
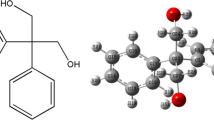
Following up on an earlier theoretical report by Knapp-Mohammady (Phys Lett A 372:1881–1884, 2008) on the ground state of the neutral Ru complex NAMI-A (trans-imidazoledimethylsulfoxide-tetrachlororuthenate), we first report here a quantum-chemical study of the effect of both oxidation and reduction of the parent molecule to form the anionic and cationic species. The new structures are compared with the equilibrium nuclear structure reported earlier for the neutral complex. We anticipate that one such Ru cluster, with potential as an anti-cancer drug, will interact via an appropriate receptor, rather than directly with DNA. A receptor for NAMI-A binding in here proposed to be the steroid hormone, estrogen, C18H24O2. The biomolecular structure of the dicomplex is predicted from restricted Hartree–Fock theory and density functional theory (DFT) calculations. The vibrational frequencies of NAMI-A and the dicomplex with estrogen are also reported. Some maps of the ground-state electron-density for the three neutral biomolecular species are finally presented. The use of vibrational spectroscopy, vibrational absorption (VA) and vibrational circular dichorism (VCD) are advocated to be measured, simulated and be used to understand the nature of the interaction of the Ru complex NAMI-A in complex with estrogen. Our aim in presenting these spectral simulations is to motivate the measurement of the VA and VCD spectra of estrogen, the Ru complex NAMI-A and finally of the estrogen–Ru NAMI-A complex. It should also be instructive to measure the VA and VCD spectra of estrogen and the estrogen receptor, both alone, together and finally together in the presence of the Ru NAMI-A complex to substantiate our claim that the Ru complex NAMI-A ties up estrogen, and hence prevents estrogen binding to the estrogen receptor.












Similar content being viewed by others
References
Knapp-Mohammady M, March NH, Suhai S (2008) Phys Lett A 372:1881
He Q, Liang CH, Lippard SJ (2000) Proc Natl Acad Sci USA 97:5768
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman V, Montgomery JA, Vreven V, Kudin KN, Burant JC, Millam JM, Iyengar SS, Tomasi J, Barone V, Mennucci B, Cossi M, Scalmani V, Rega V, Petersson GA, Nakatsuji H, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Klene M, Li X, Knox JE, Hratchian HP, Cross JB, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Ayala PY, Morokuma K, Voth GA, Salvador P, Dannenberg JJ, Zakrzewski VG, Dapprich S, Daniels AD, Strain MC, Farkas O, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Ortiz JV, Cui Q, Baboul AG, Clifford S, Cioslowski J, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Gonzalez C, Pople JA (2003) Gaussian 03, Revision A.1. Gaussian, Inc., Pittsburgh
Minaeva VA, Minaev BF, Hovorun DM (2008) Ukr Biokhim Zh. 80(4):82
March NH, Plaskett JS (1956) Proc Roy Soc (Lond) A235 419
March NH (1992) Electron density theory of atoms and molecules. Academic, New York
Parr R, Yang W (1998) Density-functional theory of atoms and molecules. Oxford University Press, New York
Ruedenberg K (1977) J Chem Phys 66:375
March NH (1981) J Chem Phys 74:2376
Hellmann H (1937) Einführung in die Quantenchemie, Deuticke, Leipzig
Feynman RP (1939) Phys Rev 56:340
Chen GC, Zeng Q, Tse GMK (2008) Med Res Rev 28:954
Nabiev IR, Sokolov KV, Voloshin ON (1990) J Raman Spectrosc 21(6):333
Barnett SM, Butler IS, Top S, Jacuen G (1998) Vib Spectrosc 8(2):263
Acknowledgments
N. H. March wishes to acknowledge that his contribution to the present article was brought to fruition during two consecutive visits to the Division of Molecular Biophysics at DKFZ. It is a pleasure to thank Professor S. Suhai for generous hospitality and for arranging the two scholarships to support the visits. Thanks also are due to Dr. B. Bythell for valuable discussions and for his help in displaying the HOMO and LUMO orbitals presented in this study. Finally, we would like to thank K.J. Jalkanen for his invitation to present this work in the Suhai Festschrift of the Springer journal Theoretical Chemistry Accounts and for scientific discussions and suggestions on how to improve this work. We would also like to acknowledge useful and insightful suggestions by the various reviewers of this article.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to Professor Sándor Suhai on the occasion of his 65th birthday and published as part of the Suhai Festschrift Issue.
Appendix
Appendix
1.1 Effect of anionic charging on neutral complex NAMI-A and on the dicomplex with estrogen
The main purpose of this Appendix is to record the change in structure of the originally neutral complex NAMI-A discussed in the main text in some detail, when two additional electrons are added. The first point of considerable interest is that the nuclear structure shown in Fig. 1 is robust against this addition of two electrons in a spin-compensated orbital. However, Fig. 13 compares the anionic equilibrium structure with that of the neutral complex, with bond lengths recorded there. The main change in structure which is worth emphasizing here is the extension of the Ru–S distance to a value of 2.89 Å in the anionic species. This increase of some 0.3 Å is of course, chemically significant. The final point on (NAMI-A)2− is that the binding energy of two additional electrons is substantial. The HOMO of this anion is shown in Fig. 14. As mentioned briefly already in the main text, in marked contrast to the above behavior of NAMI-A on anionic charging, removing two electrons leads to structural instability and we shall not therefore elaborate further on the cationic complexes.
Turning to the dicomplex of NAMI-A with estrogen, we have made a more limited study of the effect of ionic charging. We content ourselves therefore in stressing that the dicomplex anion is also quite stable. Figure 15 shows the shape at the HOMO for such a dicomplex, while Fig. 16 depicts the LUMO. Further details are available from M. Knapp-Mohammady on request.
Rights and permissions
About this article
Cite this article
Knapp-Mohammady, M., March, N.H. Quantum-chemical study of the potential anti-cancer drug Ru–NAMI-A in complex with estrogen and the simulated VA and VCD spectra of estrogen, the Ru–NAMI-A drug, and possible/proposed estrogen–Ru–NAMI-A complexes. Theor Chem Acc 125, 293–303 (2010). https://doi.org/10.1007/s00214-009-0686-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00214-009-0686-x