Abstract
Rationale
Cue-exposure therapy (CET) consists of exposing patients to the cause of their affliction in a controlled environment and after psychological preparation. Ever since it was conceived, it has been suggested as a treatment for different types of behavioural impairments, from anxiety disorders to substance abuse. In the field of addictive behaviour, many different findings have been shown regarding the effectiveness of this therapy.
Objectives
This study aims to examine the underlying neurobiological mechanisms of the effects of CET in patients with alcohol use disorder using resting-state functional magnetic resonance imaging (rs-fMRI).
Methods
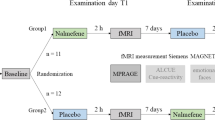
In a randomized, controlled study, we examined patients after inpatient detoxification as well as healthy controls. Patients underwent nine sessions of CET spaced over 3 weeks. Rs-fMRI was conducted before treatment and 3 weeks after treatment onset in patients, healthy controls received only one rs-fMRI measurement. The final participant sample with complete data included 35 patients in the CET group, 17 patients in the treatment-as-usual group, and 43 HCs.
Results
Our results show differences in the Salience Network when comparing the CET group to the treatment-as-usual group (TAU). Functional connectivity between the anterior cingulate Cortex (ACC) and the insula was increased after CET, whereas it was decreased from ACC to the putamen and globus pallidus. Further, increased connectivity with the precuneus was found in the dorsal attention network after cue exposure treatment.
Conclusions
These findings suggest that cue exposure therapy changes the resting-state brain connectivity with additional effects to the standard psychotherapy treatment. Hence, our study results suggest why including CET in standard therapies might improve the preparation of patients in front of daily situations.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Alcohol use disorder (AUD) is a disorder often characterized by impulsive and compulsive behaviour. Alterations of resting-state networks in individuals with AUD have been widely studied. In general, the resting brain of these patients shows network-specific patterns of aberrant functional connectivity that have been related to different emotional and behavioural outcomes (Müller-Oehring et al. 2015). The main resting-state networks affected in AUD are the default mode network (DMN) and the salience network (SN). A within-network connectivity deterioration was mainly reported in both, and also, a larger outside-network connectivity has been suggested as a compensation mechanism (Chanraud et al. 2011; Müller-Oehring et al. 2015; Sullivan et al. 2013).
The main nodes of these networks are brain regions that are known to be affected and deteriorated by alcohol abuse. Areas like the anterior cingulate cortex (ACC) or the insula, parts of the SN, have been reported to show aberrant metabolism and/or functional connectivity (Goldstein and Volkow 2011; Müller-Oehring et al. 2015; Sullivan et al. 2013). Moreover, some brain regions altered in alcohol abuse disorder are the main nodes of the DMN. The posterior cingulate cortex (PCC) and the medial prefrontal cortex (mPFC) have been reported to present enhanced functional connectivity in AUD individuals (Chanraud et al. 2011; Müller-Oehring et al. 2015). Further, frontostriatal functional connectivity was shown to be associated with alcohol-related variables like craving or self-efficacy to abstain from alcohol (Gerchen et al. 2019; Gerchen et al. 2021).
The dorsal attention network (DAN) is involved in goal-directed, voluntary control of visuospatial attention (Corbetta and Shulman 2002; Kincade et al. 2005). In a previous study, this network showed increased connectivity with the somatosensory cortex in AUD patients (Farré-Colomés et al. 2021). Its dynamic interaction with the SN (Vossel et al. 2014) is very interesting in terms of saliency processing and cue-reactivity, key features of AUD.
In the past years, the study of AUD has increasingly focused on cue-reactivity. This phenomenon is described as the psychological or physiological response that results from exposure to alcohol-related stimuli. Considering the current situation of alcohol in our society, this component of alcoholism can be decisive in maintaining the abstinence of patients and improving their quality of life within our society. So often, AUD individuals find cues in the environment that relate to alcohol use, and it facilitates relapse significantly (Conlkin and Tiffany 2002; Schacht et al. 2013). Cue exposure in our society is a very serious problem for patients who seek rehabilitation (Yalachkov et al. 2012). Cue-reactivity is the direct result of cue exposure. It can be described as a learned intrinsic response to some environmental cues triggering specific neuronal responses. After a long association period, this learning process is considered to end up with addictive behaviour (Drummond 2000; Everitt and Robbins 2005; Mellentin et al. 2017). Learning theory states that addiction is a learning process achieved by reinforcement mechanisms. For this reason, it is considered that cue reactivity can be suppressed or weakened by new learning based on exposing relevant drug cues but deleting the habitual behaviour (drug use).
Alcohol-related cues are known to activate the putamen, the ACC and the mPFC, the Orbitofrontal Cortex (OFC), the Striatum, and many other areas in AUD patients (Courtney et al. 2016; Grüsser et al. 2004; Heinz et al. 2009; Vollstädt-Klein et al. 2010), all being important due to their connection with the insula (Grüsser et al. 2004; Menon and Uddin 2010). Daily exposition to alcohol cues triggers responses related to neural processes linked to craving and alcohol drinking in AUD patients (Miranda Jr. et al. 2020; Monti et al. 2000). Still, these outcomes derived from cue exposure seem to be sensitive to medication in both treatment seekers and non-treatment seekers and in adults and adolescents (Miranda Jr. et al. 2020). In this context, cue-exposure treatment (CET), sometimes referred to as cue-extinction training or cue-exposure therapy, is based on repeated exposure to environmental stimuli that were previously associated with drug use in order to decrease the inner response of the patient in front of these cues (Conlkin and Tiffany 2002). Thus, the final aim of this procedure is to eliminate learned responses to drug cues using repeated non-reinforced exposure (Mellentin et al. 2017).
Previous studies report how patients who received the treatment as usual with CET in addition show reduced cue-elicited activation in the striatum, the ACC, the dorsal prefrontal cortex (dPFC), the insula, and the IFG (Courtney et al. 2016; Kiefer et al. 2015; Vollstädt-Klein et al. 2010). These studies suggest that CET may improve the response of neural substrates in conditioned cue reactivity, despite not having demonstrated strong effects on substance use itself (Conlkin and Tiffany 2002). Small improvements have been linked to CET in a review by Mellentin et al. (2017) regarding the final drinking intensity, frequency, total drinking score, and latency to relapse. However, no studies have focused on the effects of CET on resting-state functional connectivity.
In this study, we aimed to characterize the changes in the resting brain that CET can promote after a period of training in abstinent, treatment-seeking patients with AUD. To our knowledge, this approach is being used for the first time, which might improve knowledge of the CET effect in AUD patients. The analysis of resting-state connectivity patterns may help to understand if there is an actual change in brain connectivity and, moreover, be in line with the findings by Vollstädt-Klein et al. (2011) where reduced neural cue-reactivity was reported after CET. Based on previous studies (Müller-Oehring et al. 2015), we hypothesised that resting-state connectivity would change in patients after a 3-week CET program compared to patients with usual treatment only. Within the SN, resting-state functional connectivity of the ACC was hypothesised to be decreased towards striatal regions and increased towards the inferior parietal lobe and the precuneus in patients with CET. In the DAN, we hypothesised a reduction in functional connectivity between the FEF and motor areas of the cortex, described in a previous study (Farré-Colomés et al. 2021). As a change in the DMN, we expected an increase in frontal cortex functional connectivity with the cingulate cortex, as compensation for the AUD effects on this network.
Materials and methods
Participants
Study participants with AUD were recruited in the Department of Addictive Behaviour and Addiction Medicine, Central Institute of Mental Health in Mannheim, Germany (Deutsches Register Klinischer Studien (German Clinical Trials Registry) ID: DRKS00003388). Suitable participants were selected after undergoing a pre-treatment screening and were diagnosed with AUD according to the Diagnostic and Statistical Manual of Mental Disorders (DSM-5, assessed with Structured Clinical Interview (SCID-I) for DSM-IV (Wittchen et al. 1997) due to the unavailability of a SCID for DSM-5 at the time of examination). They did not present other axis I psychiatric disorders or substance abuse (except for nicotine). Patients were abstinent from alcohol on an average of 11 days ± 5.6 (standard deviation), with a range of 5 to 31 abstinence days. The severity of alcohol dependence was assessed using the Alcohol Dependence Scale (ADS) (Skinner and Horn 1984). The age range included participants between 18 and 65 years old at the time of testing. Using a randomization function (2:1), the selected participants were randomly distributed into two groups, the Treatment-As-Usual (TAU) group and the Cue Exposure Treatment (CET) group. Of the initial 84 selected patients, 26 did not undergo the second scanning session (Fig. 1). The study was completed by 58 patients with 39 participants in the CET group and 19 participants in the TAU group. From the CET group, 4 subjects were excluded after checking the quality of the images. In the TAU group, 2 subjects were excluded after the quality check. A total of 45 healthy controls (HC) were recruited from the community by advertisements, of whom 2 were excluded after a quality check. All participants were granted written informed consent to participate in the study. The study protocol was approved by the ethics committee of the Medical Faculty Mannheim at the University of Heidelberg (2011-303N-MA). Questionnaire scores and characteristics of the analysed sample, with respect to the different groups, are presented in Table 1.
Study design
This study was part of a larger project. The original sample size estimation was performed related to the main outcome of tasked-based fMRI, and these results are published elsewhere (Becker et al. 2018; Kirsch et al. 2015). In the current article, we report exploratory resting-state analyses from this project.
Before engaging in the study, participants with AUD had to overcome controlled abstinence for a period of 5 to 21 days under medical supervision until no severe alcohol withdrawal symptoms were present. Treatment-as-Usual (TAU) consisted of a qualified alcohol detoxification treatment at the clinic for 3 weeks. It consisted of health education, psychotherapeutic treatment in single and group sessions, competence training, relaxation exercises, sports programs, occupational therapy, and sociotherapy. Participants that were included in the CET group received 5 to 9 standardized individual CET sessions (mean 6.86, SD 0.96) additionally to TAU. These sessions followed the guidelines of a validated treatment manual (Mann 2006) that has been previously applied in other studies (Kiefer et al. 2015; Vollstädt-Klein et al. 2011). AUD participants were scanned before and after the treatment (Fig. 1). Moreover, the Obsessive Compulsive Drinking Scale (OCDS) (Anton 2000) and the Alcohol Abstinence Self-Efficacy Scale (AASE) (DiClemente et al. 1994) were fulfilled also before and after the 3-week treatment to assess the severity of alcohol craving. For the CET training sessions, it was asked to the participant to imagine critical drinking situations. Then the participant was exposed to the favourite alcoholic drink. Participants were informed in the first session about the drug-associated reactions this exposure could evoke in them, but also that this is expected to improve their reaction over time during the sessions. Besides, they were informed that CET sessions would continue until they stop feeling the craving. Participants then had to set up a hierarchy of situations that could trigger a relapse. In the following sessions, patients had to imagine one of these critical situations before being exposed to their favourite drink. They were instructed to handle the alcohol bottle, pour a drink, and smell the drink without consuming it. Meanwhile, participants had to be informed several times about the craving-related feelings, cognition, and physical reactions. Patients were asked not to stop focusing their attention on the situation they had imagined. The CET training sessions lasted between 30 and 90 min and ended when the participant stopped feeling any craving (Mann 2006). HC participants were only scanned once and underwent the same assessment as the AUD participants at the first session.
fMRI data acquisition
Functional imaging data was obtained by using a 3 T whole-body tomography Siemens Magnetom Trio Scanner (Siemens Medical Systems, Erlangen, Germany). Participants were asked to close their eyes during the 9 min of resting-state acquisition. They were also instructed to keep thoughts flowing, to not think of anything in particular, and to avoid attaching to any thought. High-resolution 3-dimensional T1-weighted anatomical images (MPRAGE) were collected to evaluate individual brain morphology (repetition time (TR) = 2.3 s, eco time (TE) = 3.03 ms, flip angle = 9°, field of view (FOV) = 256 × 256 mm2, slice thickness = 1 mm, voxel dimensions = 1 × 1 × 1 mm3, matrix size = 256 × 256, and 192 sagittal slices). For the measurement of the blood oxygen level-dependent (BOLD) contrast, 267 T2*-weighted echo-planar images (EPI) (TR = 2 s, TE = 30 ms, flip angle = 80°, 28 transversal slices, slice thickness = 4 mm, 1 mm gap, voxel dimensions = 3 × 3 × 3 mm3, FOV = 192 × 192 mm2, and matrix size = 64 × 64) were acquired in descending order.
fMRI preprocessing
Imaging data were processed using the CONN toolbox v20b (Whitfield-Gabrieli and Nieto-Castanon 2012) performed in MATLAB 2020a through SPM12 (Wellcome Department of Imaging Neuroscience, London, UK). The default pipeline from the CONN toolbox was used for the preprocessing of structural and functional images. It is composed of several steps: realignment and unwarping (with subject motion estimation and correction), centering, slice-time correction, outlier detection with ART-based (Artefact Detection Tools) identification for scrubbing with conservative parameters (95th percentiles, z-value threshold of 3 in global-signal and 0.5 mm subject-motion threshold), segmentation (white and grey matter, cerebrospinal fluid), normalization (Montreal Neurological Institute atlas), and smoothing at 8 mm full-width half maximum (FWHM) Gaussian kernel. Following the preprocessing steps, the blister variables provided by the CONN toolbox were regressed out of the signal (white matter, CSF, realignment, and scrubbing). Data were filtered using a 0.008–0.09 Hz band-pass and linear trends were suppressed using linear detrending. Individuals with excessive head movement (> 3 mm/3°) and/or other artefacts like signal drop out or incomplete field of view were discarded. Consequently, 8 subjects were discarded from the study.
Connectivity analyses
Second-level analyses were based on a seed-to-voxel first-level connectivity analysis. The CONN toolbox provides default seeds for each major network of the brain. After revision of the literature, we decided to use the DMN, the SN, and the DAN displayed in Fig. 2 (Chanraud et al. 2011; Corbetta and Shulman 2002; Kincade et al. 2005; Müller-Oehring et al. 2015; Sullivan et al. 2013; Vossel et al. 2014). To compare connectivity at rest, the toolbox uses the images generated in the first-level analyses. A seed-to-voxel approach was used to compare differential connectivity patterns in the resting brains of the different groups after treatment. Seeds were located by the CONN toolbox default parameters: the posterior cingulate cortex (PCC) was used for the DMN, the anterior cingulate cortex (ACC) for the SN, and the frontal eye fields (FEF) for the DAN. An introductory approach was conducted by comparing both AUD groups to the HC group. We analysed possible differences between groups regarding age, gender, and smoking status using a Chi2 test. Both treatment groups differed significantly in the gender of the participants. Further, the control group showed differences from both treatment groups in the smoking status. Therefore, we controlled for these variables (including them as covariates of no interest). All the data and statistics on demographic data can be found in Table 1.
Representation of the examined resting-state networks and their corresponding nodes. A DMN display including the middle prefrontal cortex, the right and left angular gyri, and the posterior cingulate cortex. B DAN depiction including the right and left frontal eye fields and the right and left intraparietal sulcus. C SN representation including the anterior cingulate cortex, the right and left anterior insula, the right and left rostral prefrontal cortex, and the right and left supramarginal gyri. D Location of the posterior cingulate cortex (x = 89, y = 84, z = 100) used as a seed to examine the DMN. E Display of the right and left frontal eye fields (x = 98, y = 147, z = 123) used as a seed to examine the DAN. F Detail of the anterior cingulate cortex (x = 96, y = 160, z = 71) used as a seed to examine the SN
Changes between pre-treatment and post-treatment were examined for each of the study groups (TAU, CET, and HC). Differences between the AUD participants and the HC are already described elsewhere (Gerchen et al. 2019; Gerchen et al. 2021) and were also included in the supplementary material. Hence, in this study, we report differences in post-treatment between HC, TAU, and CET groups. The probability of a family-wise error (FWE) was set to 0.05 to control for multiple statistical testing. For this purpose, we used the AlphaSim method implemented in the NeuroElf toolbox (www.neuroelf.net) using the 25000 Monte Carlo simulations with a voxel-level primary threshold of p < .005, resulting in a cluster-level extent threshold of k > 40 contiguous voxels. Smoothness estimation was conducted by SPM and based on residual images taking the maximum of the 3 estimated parameters in x, y, and z directions. We looked at the interaction between group and time and created ROIs as functional masks derived from significant interactions, i.e., differences in connectivity changes from pre-treatment (T1) to post-treatment (T2) between CET and TAU. Then we applied small volume correction (SVC) using these ROIs as masks when looking at the post-hoc comparison at T2 between CET vs TAU. The same statistical parameters were used to create the masks and to perform the SVC (p < 0.005 and k > 40).
Results
The study sample included participants with moderate alcohol dependence according to the alcohol dependence scale (ADS). Alcohol urge (AUQ) in the treatment groups showed an initial drinking urge not much higher than the HC group. When considering the compulsive drinking scale (OCDS), both treatment groups presented intermediate levels at pre-treatment. The temptation to drink questionnaire (AASE-V) displayed intermediate temptation levels and did not present differences between treatment groups. However, the HC group showed a significantly lower temptation to drink compared to both treatment groups. It is of note that the CET group started with a slightly higher mean score in this questionnaire and displayed lower post-treatment mean scores, despite not being significant. In the craving pre-treatment, the TAU group reported significantly less craving than the CET group both before and after the scan. However, the CET reported that post-treatment had significantly lower craving levels than TAU after the scanning session.
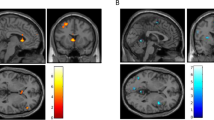
The resting-state fMRI analyses showed interaction effects between treatment and time. The SN and the DAN presented major differences between the two different treatments at T2. CET participants presented a higher increase in resting-state BOLD connectivity (i.e., T2 > T1) in the DAN between the FEF and the precuneus [(x, y, z) = (−14, −46, 48), t = 4.28, k = 284], p < 0.005 uncorrected (whole-brain analysis), compared to TAU participants (Table 2, Fig. 3). In the SN, the CET group showed increased resting-state BOLD connectivity bilaterally from the ACC to the insula [(x, y, z) = (−32, −30, 12), t = 4.24, k = 70; (x, y, z) = (36, −30, 22), t = 3.75, k = 40] when compared to the TAU group (Table 3, Fig. 4). For this same network, the CET group displayed decreased resting-state BOLD connectivity bilaterally between the ACC and the putamen and globus pallidus [(x, y, z) = (14, 8, -6), t = 4.67, k = 64; (x, y, z) = (−16, 2, 0), t = 3.91, k = 40] in comparison with the TAU group (Fig. 5). There were further results from this comparison, including weakened functional connectivity within the ACC, with the supramarginal gyrus, and with the middle temporal gyrus (Table 4). The DMN results presented enhanced rsFC between the PCC and the precuneus, inferior parietal lobule, and angular gyrus (Table 5).
Discussion
After the 3-week duration treatment, we could confirm our hypothesis by finding major differences in resting-state connectivity between CET participants and TAU participants. This study was designed to compare resting-state functional connectivity between CET and TAU in participants with AUD. Major differences in key regions related to cue-reactivity and salience processing were found. The main finding was increased functional connectivity in the SN between the ACC and the Insula in the CET group, while decreased functional connectivity was found between the ACC and the putamen and globus pallidus in the same group. Regarding the DAN, CET participants presented enhanced functional connectivity between the FEF and the precuneus after treatment. Also, the PCC showed increased rsFC with the precuneus in the DMN.
The precuneus has been related to consciousness due to its implication in the DMN and, more importantly, in many neuropsychiatric disorders that include impaired consciousness (Cavanna 2007). Its functions are suggested to be related to self-referential processes, autobiographic memory retrieval, and cognitive control (Cavanna 2007; Cavanna and Trimble 2006; Konova et al. 2013). The meta-analysis done by Konova et al. (2013) identified the precuneus, along with other regions, to be commonly activated by cognitive-based interventions that involve self-referential processing, cognitive control, and attention. Our study shows an increase of rsFC between the PCC and the precuneus after CET in the DMN. It is remarkable that also the FEF (from the DAN) displayed increased resting-state BOLD connectivity with the precuneus after CET. This increase in FC during the resting state with the precuneus is remarkable and could be of interest since it shows a pattern of increased connectivity between 3 important nodes—FEF–Precuneus–PCC
Previous studies have described an increase in fMRI response in the FEF area, one of the core nodes of the DAN, in front of reward-related visual stimuli (Chen et al. 2020; Serences 2008). Our results show an increase of rsFC between the FEF and the Precuneus after CET. This connectivity change is related to enhanced interaction between a visual and cue-reactive area of the brain, the FEF (Chen et al. 2020), with another that can integrate autobiographic memories and is related to decision-making, the precuneus (Cavanna 2007; Cavanna and Trimble 2006). Therefore, CET alters the functional connectivity between autobiographical and salience-detecting brain regions. This suggests that functional connectivity relates the saliency of visual cues with self-centered memories, but it should still be demonstrated whether these memories support or hinder abstinent behaviour. The changes in the DAN may support better regulation of the cue-reactivity exerted by the SN in the bottom-up regulation of attention. In AUD, the SN is overactivated, so the dynamic regulation between the DAN and the SN described by Vossel et al. (2014) is disrupted, bringing stronger salience effects in front of alcohol cues. This process ends in a flawed imbalance between bottom-up and top-down regulation of attention typical of substance use disorders (Zilverstand et al. 2018). Our results suggest CET might have an influence on this balance and further impact on the function of visuospatial attention guidance. Improved DAN connectivity can be related to better attentional and salience control in front of alcohol cues (Corbetta and Shulman 2002; Kincade et al. 2005).
The putamen and the globus pallidus displayed decreased functional connectivity in the SN from the ACC in the CET group. Both areas are part of the dorsal region of the striatum, and previous studies have related enhanced activation of the dorsal striatum in front of alcohol cues with elevated levels of alcohol consumption and dependence (Everitt and Robbins 2016; Vollstädt-Klein et al. 2010). Our results comparing AUD and HC showed higher resting-state BOLD connectivity within these regions in AUD. In agreement with these results, metabolic activity in the ACC and the striatum has also been reported to be higher in patients with AUD (Bralet et al. 2022). The ACC has been related to different decision-making and cognitive control processes (Menon and Uddin 2010) by inhibitory control in cognitive-based attention (Becker et al. 2018). This area was described to be stimulated by drug-related cues in several studies and different drug types and related to drug reward anticipation by encoding the motivational value of stimuli (Grüsser et al. 2004; Heinz et al. 2009; Schacht et al. 2013; Vollstädt-Klein et al. 2010). It is suggested to be the one selecting and evaluating external stimuli to propose the form of action to be implemented by the motor system accordingly (Bush et al. 2002). Since ACC collects environmental information and inputs, then processes the drug-related cues and relates them to the dorsal striatum, we suggest that this increase of activation in front of drug-related cues may cause the activation of the dorsal striatum which is the main responsible for craving and drug-seeking behaviour (Everitt and Robbins 2005; Müller-Oehring et al. 2015). Therefore, the decrease of resting-state connectivity between the ACC and the striatum after CET would protect the patient from excessive craving and drinking desires in front of alcohol-related cues by splitting this synchronic activity of both regions as a compensation mechanism. The ACC still receives the salient stimuli of environmental cues, but the striatum might be less sensitive to them because it is not receiving the same amount of stimulation as before the treatment. A decrease in resting-state connectivity from the ACC to the striatum may reflect an improvement of cue-reactivity control by compensation of aberrant AUD connectivity between these regions (Müller-Oehring et al. 2015; Schacht et al. 2013). Thus, in front of alcohol-related cues, the participant would not “feel” the urge to drink and could probably resist craving more easily.
Despite expecting an increase in functional connectivity with the IPL or the precuneus, we reported an increase in functional connectivity between the ACC and the insula, which rectifies one of the most described impairments in AUD. It has been widely described as a decrease in functional connectivity in the ACC and the insula in AUD (Everitt and Robbins 2016; Koob and Volkow 2016; Sullivan et al. 2013). Our results also reported this connectivity impairment in AUD participants when compared to HC. The insula has been repeatedly described as a region processing interoceptive information, mediating the internal state of the individual and the environmental inputs arriving (Eckert et al. 2009; Koob and Volkow 2016; Taylor et al. 2009). The region has been further implicated in salience awareness (Menon and Uddin 2010), being involved in interoceptive integration and emotional salience (Taylor et al. 2009). Since the insula is described as receiving multimodal sensory inputs, the decrease in functional connectivity between the insula and the ACC would reflect changes in sensory processing. Lower functional connectivity between these regions may reflect a reduction of the effect that alcoholic cues have on the participant’s emotional salience.
CET has been proposed as an inhibitory training that could facilitate the increase of cognitive control in the subjects undergoing it (Becker et al. 2018). It was proposed that new associations should be made by active inhibition and learning to restore undesired behaviours instead of unlearning or eliminating these previously established associations (Gass and Chandler 2013). The effect of CET in AUD has been analyzed in previous studies and demonstrated to have an impact on impaired cue-induced activation of several brain regions, like the ACC among others (Kiefer et al. 2015). Our results are in line with that study, showing decreased connectivity between the ACC and the dorsal striatum as a consequence of CET. Accordingly, the inhibitory training may have interfered in the functional connectivity synchronizing ACC and the striatum’s activation and therefore helped to obstruct the cue-reactivity processing in the brain before it turns into an overwhelming craving.
Becker et al. (2018), who reported also data from our sample, reported an increase in self-reported clinical outcome measures after CET. They found a positive correlation between increased ACC activation and the improvement of self-reported efficacy to abstain in CET participants. In that study, authors associated these changes with an elevated baseline reward sensitivity. However, it further suggested that brain functional connectivity changes can be related to a perceived improvement in self-efficacy.
In conclusion, our results, together with previous studies, improve our knowledge of the neural mechanisms underlying CET by providing fMRI evidence of its effect on resting-state connectivity. Nevertheless, we must keep in mind that our results are derived from a specific sample with patients abstinent for a determined time period. Results should be taken into consideration carefully because the impact of CET reported in this study may not last forever. Moreover, studies with more participants and better group demographics would help provide better statistical strength. Nowadays alcohol cues are present everywhere and abstinent patients must face different levels of exposure every day. Exposure to alcohol cues they were not really prepared for. CET prepares patients to face these situations they will repeatedly live in the near future, enhancing their ability to hold on to spontaneous alcohol consumption appetite through inhibition training. Because of this, CET includes the familiarisation of habitual scenarios where the patient will face different kinds of alcohol cues. A recent study reports habituation of the salience processing after repeated drug cue exposure in abstinent and treatment-engaged patients (Ekhtiari et al. 2021). Further studies are needed to see how long these effects last and to see if there is an impact on relapse rates and other clinical outcomes.
References
Anton RF (2000) Obsessive-compulsive aspects of craving: development of the obsessive compulsive drinking scale. Addiction 95:S211–S217. https://doi.org/10.1080/09652140050111771
Becker A, Gerchen MF, Kirsch M, Hoffmann S, Kiefer F, Kirsch P (2018) Striatal reward sensitivity predicts therapy-related neural changes in alcohol addiction. Eur Arch Psychiatry Clin Neurosci 268(3):231–242. https://doi.org/10.1007/s00406-017-0805-y
Bralet MC, Mitelman SA, Goodman CR, Lincoln S, Hazlett EA, Buchsbaum MS (2022) Fluorodeoxyglucose positron emission tomography scans in patients with alcohol use disorder. Alcohol Clin Exp Res 46(6):994–1010. https://doi.org/10.1111/acer.14845
Bush G, Vogt BA, Holmes J, Dale AM, Greve D, Jenike MA, Rosen BR (2002) Dorsal anterior cingulate cortex: a role in reward-based decision making. PNAS 99(1):523–528. https://doi.org/10.1073/pnas.012470999
Cavanna AE (2007) The precuneus and consciousness. CNS Spectrums 12(7):454–452. https://doi.org/10.1017/s1092852900021295
Cavanna AE, Trimble MR (2006) The precuneus: a review of its functional anatomy and behavioural correlates. Brain 129(Pt 3):564–583. https://doi.org/10.1093/brain/awl004
Chanraud S, Pitel AL, Pfefferbaum A, Sullivan EV (2011) Disruption of functional connectivity of the default-mode network in alcoholism. Cereb Cortex 21(10):2272–2281. https://doi.org/10.1093/cercor/bhq297
Chen X, Zirnsak M, Vega GM, Moore T (2020) Frontal eye field neurons selectively signal the reward value of prior actions. Prog Neurobiol 195:101881. https://doi.org/10.1016/j.pneurobio.2020.101881
Conlkin CA, Tiffany ST (2002) Applying extinction research and theory to cue-exposure addiction treatments. Addiction 97(2):155–167. https://doi.org/10.1046/j.1360-0443.2002.00014.x
Corbetta M, Shulman GL (2002) Control of goal-directed and stimulus-driven attention in the brain. Nat Rev Neurosci 3(3):201–215. https://doi.org/10.1038/nrn755
Courtney KE, Schacht JP, Hutchison K, Roche DJ, Ray LA (2016) Neural substrates of cue reactivity: association with treatment outcomes and relapse. Addict Biol 21(1):3–22. https://doi.org/10.1111/adb.12314
DiClemente CC, Carbonari JP, Montgomery RP, Hughes SO (1994) The alcohol abstinence self-efficacy scale. J Stud Alcohol 55(2):141–148. https://doi.org/10.15288/jsa.1994.55.141
Drummond DC (2000) What does cue-reactivity have to offer clinical research? Addiction 95(Suppl 2):S129–S144. https://doi.org/10.1080/09652140050111708
Eckert MA, Menon V, Walczak A, Ahlstrom J, Denslow S, Horwitz A, Dubno JR (2009) At the heart of the ventral attention system: the right anterior insula. Hum Brain Mapp 30(8):2530–2541. https://doi.org/10.1002/hbm.20688
Ekhtiari H, Kuplicki R, Aupperle RL, Paulus MP (2021) It is never as good the second time around: brain areas involved in salience processing habituate during repeated drug cue exposure in treatment engaged abstinent methamphetamine and opioid users. NeuroImage 238:118180. https://doi.org/10.1016/j.neuroimage.2021.118180
Everitt BJ, Robbins TW (2005) Neural systems of reinforcement for drug addiction: from actions to habits to compulsion. Nat Neurosci 8(11):1481–1489. https://doi.org/10.1038/nn1579
Everitt BJ, Robbins TW (2016) Drug addiction: updating actions to habits to compulsions ten years on. Annu Rev Psychol 67:23–50. https://doi.org/10.1146/annurev-psych-122414-033457
Farré-Colomés À, Gerhardt S, Luderer M, Sobanski E, Kiefer F, Vollstädt-Klein S (2021) Common and distinct neural connectivity in attention-deficit/hyperactivity disorder and alcohol use disorder studied using resting-state functional magnetic resonance imaging. Alcohol Clin Exp Res 45(5):948–960. https://doi.org/10.1111/acer.14593
Gass JT, Chandler LJ (2013) The plasticity of extinction: contribution of the prefrontal cortex in treating addiction through inhibitory learning. Front Psychiatry 4:46. https://doi.org/10.3389/fpsyt.2013.00046
Gerchen MF, Rentsch A, Kirsch M, Kiefer F, Kirsch P (2019) Shifts in the functional topography of frontal cortex-striatum connectivity in alcohol use disorder. Addict Biol 24(6):1245–1253. https://doi.org/10.1111/adb.12692
Gerchen MF, Weiss F, Kirsch M, Rentsch A, Halli P, Kiefer F, Kirsch P (2021) Dynamic frontostriatal functional peak connectivity (in alcohol use disorder). Hum Brain Mapp 42(1):36–46. https://doi.org/10.1002/hbm.25201
Goldstein RZ, Volkow ND (2011) Dysfunction of the prefrontal cortex in addiction: neuroimaging findings and clinical implications. Nat Rev Neurosci 12(11):652–669. https://doi.org/10.1038/nrn3119
Grüsser SM, Wrase J, Klein S, Hermann D, Smolka MN, Ruf M, Weber-Fahr W, Flor H, Mann K, Braus DF, Heinz A (2004) Cue-induced activation of the striatum and medial prefrontal cortex is associated with subsequent relapse in abstinent alcoholics. Psychopharmacology 175(3):296–302. https://doi.org/10.1007/s00213-004-1828-4
Heinz A, Beck A, Grusser SM, Grace AA, Wrase J (2009) Identifying the neural circuitry of alcohol craving and relapse vulnerability. Addict Biol 14(1):108–118. https://doi.org/10.1111/j.1369-1600.2008.00136.x
Heinz, A., Kiefer, F., Smolka, M. N., Endrass, T., Beste, C., Beck, A., Liu, S., Genauck, A., Romund, L., Banaschewski, T., Bermpohl, F., Deserno, L., Dolan, R. J., Durstewitz, D., Ebner-Priemer, U., Flor, H., Hansson, A. C., Heim, C., Hermann, D., . . . Spanagel, R. (2020). Addiction research consortium: losing and regaining control over drug intake (ReCoDe)-from trajectories to mechanisms and interventions. Addict Biol, 25(2), e12866. DOI: https://doi.org/10.1111/adb.12866
Kiefer F, Kirsch M, Bach P, Hoffmann S, Reinhard I, Jorde A, von der Goltz C, Spanagel R, Mann K, Loeber S, Vollstadt-Klein S (2015) Effects of D-cycloserine on extinction of mesolimbic cue reactivity in alcoholism: a randomized placebo-controlled trial. Psychopharmacology 232(13):2353–2362. https://doi.org/10.1007/s00213-015-3882-5
Kincade JM, Abrams RA, Astafiev SV, Shulman GL, Corbetta M (2005) An event-related functional magnetic resonance imaging study of voluntary and stimulus-driven orienting of attention. J Neurosci 25(18):4593–4604. https://doi.org/10.1523/JNEUROSCI.0236-05.2005
Kirsch M, Gruber I, Ruf M, Kiefer F, Kirsch P (2015) Real-time fMRI neurofeedback can reduce striatal cue reactivity to alcohol stimuli. Addiction Biology 21(4):982–992. https://doi.org/10.1111/adb.12278
Konova AB, Moeller SJ, Goldstein RZ (2013) Common and distinct neural targets of treatment: changing brain function in substance addiction. Neurosci Biobehav Rev 37(10 Pt 2):2806–2817. https://doi.org/10.1016/j.neubiorev.2013.10.002
Koob GF, Volkow ND (2016) Neurobiology of addiction: a neurocircuitry analysis. Lancet Psychiatry 3(8):760–773. https://doi.org/10.1016/S2215-0366(16)00104-8
Mann K (2006) Qualifizierte Entzugsbehandlung von Alkoholabhängigen: ein Manual zur Pharmako-und Psychotherapie; mit 6 Tabellen und 20 Arbeitsblättern; alle Arbeitsblätter auf CDROM. Deutscher Ärzteverlag
Mellentin AI, Skot L, Nielsen B, Schippers GM, Nielsen AS, Stenager E, Juhl C (2017) Cue exposure therapy for the treatment of alcohol use disorders: a meta-analytic review. Clin Psychol Rev 57:195–207. https://doi.org/10.1016/j.cpr.2017.07.006
Menon V, Uddin LQ (2010) Saliency, switching, attention and control: a network model of insula function. Brain Struct Funct 214(5-6):655–667. https://doi.org/10.1007/s00429-010-0262-0
Miranda R Jr, O'Malley SS, Treloar Padovano H, Wu R, Falk DE, Ryan ML, Fertig JB, Chun TH, Muvvala SB, Litten RZ (2020) Effects of alcohol cue reactivity on subsequent treatment outcomes among treatment-seeking individuals with alcohol use disorder: a multisite randomized, double-blind, placebo-controlled clinical trial of varenicline. Alcohol Clin Exp Res 44(7):1431–1443. https://doi.org/10.1111/acer.14352
Monti PM, Rohsenow DJ, Hutchison KE (2000) Toward bridging the gap between biological, psychobiological and psychosocial models of alcohol craving. Addiction 95(S2):S229–S236. https://doi.org/10.1080/09652140050111799
Müller-Oehring EM, Jung YC, Pfefferbaum A, Sullivan EV, Schulte T (2015) The resting brain of alcoholics. Cereb Cortex 25(11):4155–4168. https://doi.org/10.1093/cercor/bhu134
Schacht JP, Anton RF, Myrick H (2013) Functional neuroimaging studies of alcohol cue reactivity: a quantitative meta-analysis and systematic review. Addict Biol 18(1):121–133. https://doi.org/10.1111/j.1369-1600.2012.00464.x
Serences JT (2008) Value-based modulations in human visual cortex. Neuron 60(6):1169–1181. https://doi.org/10.1016/j.neuron.2008.10.051
Skinner HA, Horn JL (1984) Alcohol dependence scale (ADS) user’s guide. Addiction Research Foundation, Toronto, Canada
Sullivan EV, Muller-Oehring E, Pitel AL, Chanraud S, Shankaranarayanan A, Alsop DC, Rohlfing T, Pfefferbaum A (2013) A selective insular perfusion deficit contributes to compromised salience network connectivity in recovering alcoholic men. Biol Psychiatry 74(7):547–555. https://doi.org/10.1016/j.biopsych.2013.02.026
Taylor KS, Seminowicz DA, Davis KD (2009) Two systems of resting state connectivity between the insula and cingulate cortex. Hum Brain Mapp 30(9):2731–2745. https://doi.org/10.1002/hbm.20705
Vollstädt-Klein S, Loeber S, Kirsch M, Bach P, Richter A, Bühler M, von der Goltz C, Hermann D, Mann K, Kiefer F (2011) Effects of cue-exposure treatment on neural cure reactivity in alcohol dependence: a randomized trial. Biol Psychiatry 69(11):1060–1066. https://doi.org/10.1016/j.biopsych.2010.12.016
Vollstädt-Klein S, Wichert S, Rabinstein J, Buhler M, Klein O, Ende G, Hermann D, Mann K (2010) Initial, habitual and compulsive alcohol use is characterized by a shift of cue processing from ventral to dorsal striatum. Addiction 105(10):1741–1749. https://doi.org/10.1111/j.1360-0443.2010.03022.x
Vossel S, Geng JJ, Fink GR (2014) Dorsal and ventral attention systems: distinct neural circuits but collaborative roles. Neuroscientist 20(2):150–159. https://doi.org/10.1177/1073858413494269
Whitfield-Gabrieli S, Nieto-Castanon A (2012) Conn: a functional connectivity toolbox for correlated and anticorrelated brain networks. Brain Connect 2(3):125–141. https://doi.org/10.1089/brain.2012.0073
Wittchen HU, Wunderlich U, Gruschwitz S, Zaudig M (1997) SKID I. Strukturiertes Klinisches Interview für DSM-IV. In: Achse I: Psychische Störungen. Interviewheft und Beurteilungsheft. Eine deutschsprachige, erweiterte Bearb. d. amerikanischen Originalversion des SKID I. Hogrefe
Yalachkov Y, Kaiser J, Naumer MJ (2012) Functional neuroimaging studies in addiction: multisensory drug stimuli and neural cue reactivity. Neurosci Biobehav Rev 36(2):825–835. https://doi.org/10.1016/j.neubiorev.2011.12.004
Zilverstand A, Huang AS, Alia-Klein N, Goldstein RZ (2018) Neuroimaging impaired response inhibition and salience attribution in human drug addiction: a systematic review. Neuron 98(5):886–903. https://doi.org/10.1016/j.neuron.2018.03.048
Acknowledgements
We would like to thank Alena Rentsch for her assistance in data collection.
Funding
Open Access funding enabled and organized by Projekt DEAL. Deutsche Forschungsgemeinschaft (DFG, German Research Foundation): Project-ID 421888313, Project-ID 402170461/TRR 265 (Heinz et al. 2020), and SFB 636/D6
Author information
Authors and Affiliations
Contributions
PK and FK were responsible for the concept and design of the original study. AFC, HT, and SVK were responsible for the analysis plan. MFG and MK contributed to the acquisition of fMRI and psychometric data. MK and SH took care of the preparation of clinical data. AFC and HT performed the data analysis with the supervision of SVK. AFC, HT, and SVK contributed to the interpretation of data. AFC drafted the manuscript. All authors provided critical revision of the manuscript for important intellectual content. PK, FK, and SVK procured study funding. All authors critically reviewed the content and approved the final version for publication.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Farré-Colomés, À., Tan, H., Gerhardt, S. et al. Cue-exposure treatment influences resting-state functional connectivity—a randomized controlled fMRI study in alcohol use disorder. Psychopharmacology 241, 513–524 (2024). https://doi.org/10.1007/s00213-024-06531-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-024-06531-x