Abstract
Rationale
Drug combinations are commonly used in pain management, which can produce potent analgesic effects with reduced dosage and adverse effects.
Objective
This study was designed to evaluate the anti-nociceptive effects and adverse effects of new combinations of flupirtine (a Kv7 potassium channel opener) and antihistamines (promethazine, fexofenadine) on acute and chronic pain in mice, and the possible mechanisms behind the synergistic analgesic effects were preliminarily investigated.
Methods
In acetic acid writhing test, carrageenan-induced inflammatory pain model, and paclitaxel-induced neuropathic pain model, the interaction indexes (γ) between flupirtine and antihistamines were determined by isobolographic analysis. Furthermore, the Kv7 channel blocker XE991 was used to determine whether the effects of single agents and drug combinations on paclitaxel- and carrageenan-induced mechanical allodynia were mediated by Kv7 channels. Finally, hepatotoxicity markers, liver histopathology, and the rotarod test were used to investigate the adverse effects of drugs in combination doses.
Results
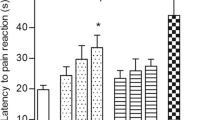
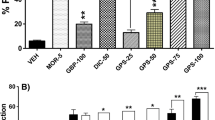
The interaction indexes of flupirtine-promethazine and flupirtine-fexofenadine in all the above three pain models were lower than 1. The analgesic effects of flupirtine (13 mg/kg), promethazine (5 mg/kg), fexofenadine (20 mg/kg), and their combinations were antagonized significantly by XE991 (3 mg/kg). And the adverse effects of flupirtine and antihistamines in combination doses were not significantly different from the vehicle group.
Conclusions
Flupirtine and antihistamines produced synergistic analgesic effects in all the above pain models. The analgesic effects of antihistamines were partially mediated by Kv7/M channels, and the activation of Kv7/M channels may be partly responsible for the synergistic analgesic effects between flupirtine and antihistamines.









Similar content being viewed by others
References
Abd-Elsayed A, Jackson M, Gu SL, Fiala K, Gu J (2019) Neuropathic pain and Kv7 voltage-gated potassium channels: the potential role of Kv7 activators in the treatment of neuropathic pain. Mol Pain 15:1744806919864256. https://doi.org/10.1177/1744806919864256
Akuodor GC, Mbah CC, Anyalewechi NA, Idris-Usman MIT, Osunkwo UA (2011) Pharmacological profile of aqueous extract of Bridelia ferruginea stem bark in the relief of pain and fever. J Med Plants Res 5:5366–5369
Al-Mazidi S, Alotaibi M, Nedjadi T, Chaudhary A, Alzoghaibi M, Djouhri L (2018) Blocking of cytokines signalling attenuates evoked and spontaneous neuropathic pain behaviours in the paclitaxel rat model of chemotherapy-induced neuropathy. Eur J Pain 22:810–821. https://doi.org/10.1002/ejp.1169
Amidi N, Izadidastenaei Z, Araghchian M, Ahmadimoghaddam D (2020) A behavioral study of promethazine interaction with analgesic effect of diclofenac: pain combination therapy. J Pharmacopuncture 23:18. https://doi.org/10.3831/KPI.2020.23.003
Busserolles J, Tsantoulas C, Eschalier A, García JAL (2016) Potassium channels in neuropathic pain: advances, challenges, and emerging ideas. Pain 157:S7–S14. https://doi.org/10.1097/j.pain.0000000000000368
Cardia GFE, Silva-Filho SE, Silva EL, Uchida NS, Cavalcante HAO, Cassarotti LL, Cuman RKN (2018) Effect of lavender (Lavandula angustifolia) essential oil on acute inflammatory response. Evid-Based Compl Alt 2018. https://doi.org/10.1155/2018/1413940
Chen X, Li W, Hiett SC, Obukhov AG (2016) Novel roles for Kv7 channels in shaping histamine-induced contractions and bradykinin-dependent relaxations in pig coronary arteries. PLoS One 11:e0148569. https://doi.org/10.1371/journal.pone.0148569
Crunkhorn P, Meacock SCR (1971) Mediators of the inflammation induced in the rat paw by carrageenin. Brit J Pharmacol 42:92–402. https://doi.org/10.1111/j.1476-5381.1971.tb07124.x
Das P, Delost MD, Qureshi MH, Smith DT, Njardarson JT (2018) A survey of the structures of US FDA approved combination drugs. J Med Chem 62:4265–4311. https://doi.org/10.1021/acs.jmedchem.8b01610
Dhawale T, Kumar S, Dharmadhikari S (2019) Evaluation of analgesic and anti-inflammatory activity of ibuprofen and duloxetine in animal models. Natl J Physiol Pharm Pharmacol 9:842. https://doi.org/10.5455/njppp.2019.9.0414207062019
Fernando MP, Alberto HL, Guadalupe VDM, Agustina CM, Fernando NG, Eva AH, Eva GTM (2021) Neo-clerodane diterpenic influence in the antinociceptive and anti-inflammatory properties of Salvia circinnata Cav. J Ethnopharmacol 268:113550. https://doi.org/10.1016/j.jep.2020.113550
Foreman JC (1987) Substance P and calcitonin gene-related peptide: effects on mast cells and in human skin. Int Arch Allergy Imm 82:366–371. https://doi.org/10.3389/fendo.2017.00015
Friedman BW, Adewunmi V, Campbell C, Solorzano C, Esses D, Bijur PE, Gallagher EJ (2013) A randomized trial of intravenous ketorolac versus intravenous metoclopramide plus diphenhydramine for tension-type and all nonmigraine, noncluster recurrent headaches. Ann Emerg Med 62:311–318. https://doi.org/10.1016/j.annemergmed.2013.03.017
Galeotti N, Ghelardini C, Bartolini A (2002) Antihistamine antinociception is mediated by Gi-protein activation. Neuroscience 109:811–818. https://doi.org/10.1016/S0306-4522(01)00537-1
Goodchild CS, Kolosov A, Tucker AP, Cooke I (2008) Combination therapy with flupirtine and opioid: studies in rat pain models. Pain Med 9:928–938. https://doi.org/10.1111/j.1526-4637.2008.00514.x
Harish S, Bhuvana K, Bengalorkar GM, Kumar TN (2012) Flupirtine: clinical pharmacology. J Anaesthesiol, Clin Pharmacol 28:172. https://doi.org/10.4103/0970-9185.94833
Hayashi H, Iwata M, Tsuchimori N, Matsumoto T (2014) Activation of peripheral KCNQ channels attenuates inflammatory pain. Mol Pain 10:1744–8069. https://doi.org/10.1186/1744-8069-10-15
Heydrnejad MS, Samani RJ, Aghaeivanda S (2015) Toxic effects of silver nanoparticles on liver and some hematological parameters in male and female mice (Mus musculus). Biol Trace Elem Res 165:153–158. https://doi.org/10.1007/s12011-015-0247-1
Kandasamy R, Morgan MM (2021) “Reinventing the wheel” to advance the development of pain therapeutics. Behav Pharmacol 32:142. https://doi.org/10.1097/FBP.0000000000000596
Kaplan S, Ehlken B, Hamann X (2019) The impact of risk minimization measures on compliance and prescribing practices of flupirtine in Germany. Curr Med Res Opin 35:33–40. https://doi.org/10.1080/03007995.2018.1499507
Kárpáti A, Yoshikawa T, Nakamura T, Iida T, Matsuzawa T, Kitano H, Yanai K (2018) Histamine elicits glutamate release from cultured astrocytes. J Pharmacol Sci 137:122–128. https://doi.org/10.1016/j.jphs.2018.05.002
Khalilzadeh E, Azarpey F, Hazrati R, Saiah GV (2018) Evaluation of different classes of histamine H1 and H2 receptor antagonist effects on neuropathic nociceptive behavior following tibial nerve transection in rats. Eur J Pharmacol 834:221–229. https://doi.org/10.1016/j.ejphar.2018.07.011
Kirkpatrick DR, McEntire DM, Smith TA, Dueck NP, Kerfeld MJ, Hambsch ZJ, Agrawal DK (2016) Transmission pathways and mediators as the basis for clinical pharmacology of pain. Expert Rev Clin Phar 9:1363–1387. https://doi.org/10.1080/17512433.2016.1204231
Kolosov A, Goodchild CS, Williams ED, Cooke I (2012) Flupirtine enhances the anti-hyperalgesic effects of morphine in a rat model of prostate bone metastasis. Pain Med 13:1444–1456. https://doi.org/10.1111/j.1526-4637.2012.01502.x
Lawson K (2019) Pharmacology and clinical applications of flupirtine: current and future options. World J Pharmacol 8:1–13. https://doi.org/10.5497/wjp.v8.i1.1
Lemmerhirt CJ, Rombach M, Bodtke A (2015) Oxidation potentials of N-modified derivatives of the analgesic flupirtine linked to potassium Kv7 channel opening activity but not hepatocyte toxicity. ChemMedChem 10:368–379. https://doi.org/10.1002/cmdc.201402442
Ma Y, Liu W, Liang L, Ye J, Huang C, Zhuang T, Zhang G (2022) Synergistic antinociceptive effects of indomethacin–pregabalin and meloxicam–pregabalin in paclitaxel-induced neuropathic pain. Biomedicines 10:1413. https://doi.org/10.3390/biomedicines10061413
Mansouri MT, Naghizadeh B, Ghorbanzadeh B (2015) Ellagic acid enhances the antinociceptive action of venlafaxine in mouse acetic acid-induced pain: an isobolographic analysis. Pharmacol Rep 67:473–477. https://doi.org/10.1016/j.pharep.2014.11.004
Mitra SK, Venkataranganna MV, Sundaram R (1998) Protective effect of HD-03, a herbal formulation, against various hepatotoxic agents in rats. J Ethnopharmacol 63:181–186. https://doi.org/10.1016/S0378-8741(98)00088-9
Mogil JS (2012) Sex differences in pain and pain inhibition: multiple explanations of a controversial phenomenon. Nat Rev Neurosci 13:859–866. https://doi.org/10.1038/nrn3360
Munro G, Dalby-Brown W (2007) Kv7 (KCNQ) channel modulators and neuropathic pain. J Med Chem 50:2576–2582. https://doi.org/10.1021/jm060989l
O’Neill A, Lirk P (2022) Multimodal analgesia. Anesthesiol Clin 40:455–468
Panula P, Chazot PL, Cowart M, Gutzmer R, Leurs R, Liu WL, Haas HL (2015) International union of basic and clinical pharmacology XCVIII. Histamine receptors. Pharmacol Rev 67:601–655. https://doi.org/10.1124/pr.114.010249
Parvathy SS, Masocha W (2013) Matrix metalloproteinase inhibitor COL-3 prevents the development of paclitaxel-induced hyperalgesia in mice. Med Prin Pract 22:35–41. https://doi.org/10.1159/000341710
Patil MA, Matter BA, Raol YH, Bourne DW, Kelley RA, Kompella UB (2018) Brain distribution and metabolism of flupirtine, a nonopioid analgesic drug with antiseizure effects, in neonatal rats. Pharmaceutics 10:281. https://doi.org/10.3390/pharmaceutics10040281
Pourfakhr P, Ziaei SH, Etezadi F, Khajavi MR, Sharifnia MR (2019) Diphenhydramine definitely suppresses fentanyl-induced cough during general anesthesia induction: a double-blind, randomized, and placebo-controlled study. Acta Med Iran:316–319. https://doi.org/10.18502/acta.v57i5.1868
Priya L, Govindarao D, Vinay MS (2019) Efficacy of flupirtine maleate as pre-emptive analgesic. Indian J Clin Anaesth 6:391–394
Qabazard B, Masocha W, Khajah M, Phillips OA (2020) H2S donor GYY4137 ameliorates paclitaxel-induced neuropathic pain in mice. Biomed Pharmacother 127:110210. https://doi.org/10.1016/j.biopha.2020.110210
Sato A, Arichi S, Kojima F, Hayashi T, Ohba T, Cheung DL, Ishibashi H (2020) Histamine depolarizes rat intracardiac ganglion neurons through the activation of TRPC non-selective cation channels. Eur J Pharmacol 886:173536. https://doi.org/10.1016/j.ejphar.2020.173536
Senior JR (2014) Evolution of the Food and Drug Administration approach to liver safety assessment for new drugs: current status and challenges. Drug safety 37:9–17. https://doi.org/10.1007/s40264-014-0182-7
Slivicki RA, Xu Z, Kulkarni PM, Pertwee RG, Mackie K, Thakur GA, Hohmann AG (2018) Positive allosteric modulation of cannabinoid receptor type 1 suppresses pathological pain without producing tolerance or dependence. Biol Psychiatry 84:722–733. https://doi.org/10.1016/j.biopsych.2017.06.032
Smith SB, Crager SE, Mogil JS (2004) Paclitaxel-induced neuropathic hypersensitivity in mice: responses in 10 inbred mouse strains. Life Sci 74:2593–2604. https://doi.org/10.1016/j.lfs.2004.01.002
Staff NP, Fehrenbacher JC, Caillaud M, Damaj MI, Segal RA, Rieger S (2020) Pathogenesis of paclitaxel-induced peripheral neuropathy: a current review of in vitro and in vivo findings using rodent and human model systems. Exp neurol 324:113121. https://doi.org/10.1016/j.expneurol.2019.113121
Stepanovic-Petrovic RM, Micov AM, Tomic MA, Kovacevic JM, Boškovic´ BD (2014) Antihyperalgesic/antinociceptive effects of ceftriaxone and its synergistic interactions with different analgesics in inflammatory pain in rodents. Anesthesiology 120:737–750. https://doi.org/10.1097/ALN.0000435833.33515.ba
Teng BC, Song Y, Zhang F, Ma TY, Qi JL, Zhang HL, Wang K (2016) Activation of neuronal Kv7/KCNQ/M-channels by the opener QO58-lysine and its anti-nociceptive effects on inflammatory pain in rodents. Acta Pharmacol Sin 37:1054–1062. https://doi.org/10.1038/aps.2016.33
Teodoro FC, Júnior MFT, Zampronio AR, Martini AC, Rae GA, Chichorro JG (2013) Peripheral substance P and neurokinin-1 receptors have a role in inflammatory and neuropathic orofacial pain models. Neuropeptides 47:199–206. https://doi.org/10.1016/j.npep.2012.10.005
Tomic MA, Vuckovic SM, Stepanovic-Petrovic RM, Ugrešic ND, Prostran MŠ, Boškovic B (2010) Synergistic interactions between paracetamol and oxcarbazepine in somatic and visceral pain models in rodents. Anesthesia & Analgesia 110:1198–1205. https://doi.org/10.1213/ANE.0b013e3181cbd8da
Tsai DS, Huang MH, Tsai JC, Chang YS, Chiu YJ, Lin YC, Peng WH (2015) Analgesic and anti-inflammatory activities of Rosa taiwanensis nakai in mice. J Med Food 18:592–600. https://doi.org/10.1089/jmf.2014.3197
Vena GA, Cassano N, Buquicchio R, Ventura MT (2008) Antiinflammatory effects of H1-antihistamines: clinical and immunological relevance. Curr Pharm Design 14:2902–2911. https://doi.org/10.2174/138161208786369777
Wang H, Xu H, Wu LJ, Kim SS, Chen T, Koga K, Zhuo M (2011) Identification of an adenylyl cyclase inhibitor for treating neuropathic and inflammatory pain. Sci Transl Med 3:65ra3. https://doi.org/10.1126/scitranslmed.300126
Wu Z, Yang Q (2022) KCNQ/Kv7 channels as therapeutic target to treat neuropathic pain. In: Rajendram R, Patel VB, Preedy VR, Martin CR (eds) The neurobiology, physiology, and psychology of pain. Elsevier, Amsterdam, pp 3–12. https://doi.org/10.1016/B978-0-12-820589-1.00001-4
Yadav NK, Shukla T, Upmanyu N, Pandey SP, Khan MA, Jain DK (2019) Concise review: therapeutic potential of flupirtine maleate. J Drug Deliv Ther 9:67–471. https://doi.org/10.22270/jddt.v9i1-s.2350
Zhang F, Mi Y, Qi JL, Li JW, Si M, Guan BC, Zhang HL (2013) Modulation of Kv7 potassium channels by a novel opener pyrazolo [1, 5-a] pyrimidin-7 (4H)-one compound QO-58. Brit J Pharmacol 168:1030–1042. https://doi.org/10.1111/j.1476-5381.2012.02232.x
Zhang F, Liu S, Jin L, Tang L, Zhao X, Yang T, Li H (2020) Antinociceptive efficacy of retigabine and flupirtine for gout arthritis pain. Pharmacology 105:471–476. https://doi.org/10.1159/000505934
Zhuang T, Xiong J, Hao S, Du W, Liu Z, Liu B, Chen Y (2021) Bifunctional μ opioid and σ1 receptor ligands as novel analgesics with reduced side effects. Eur J Med Chem 223:113658. https://doi.org/10.1016/j.ejmech.2021.113658
Acknowledgements
We are grateful for the financial support from the Scientific Research Foundation of Jiangsu Ocean University (KQ22025) and the Project Funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions (2022JSPAPD006).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, Y., Xiao, X., Huang, C. et al. Flupirtine and antihistamines exert synergistic anti-nociceptive effects in mice. Psychopharmacology 240, 881–897 (2023). https://doi.org/10.1007/s00213-023-06329-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-023-06329-3