Abstract
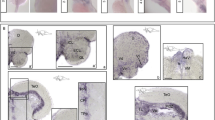
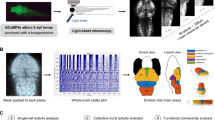
Genome-wide analysis has identified the transcription factor, RRN3 (or TIF-1A), on human chromosome 16p13.11 as a candidate gene associated with mental disorders. Both genetic and biochemical experiments have demonstrated that RRN3 plays a major role in the transcriptional regulation of ribosomal DNA and cell growth. Previous research has suggested that loss of RRN3 from mature neurons reproduces the chronic nature of neurodegenerative processes. Here, we report the first generation and characterization of rrn3 mutant zebrafish in larval and adult stages using the CRISPR/Cas9 genome editing technique. Homozygous knockout zebrafish exhibited morphological changes, such as pericardial oedema and deformed heads, and died at the larval stage of embryonic development. Behaviourally, the locomotion and shoaling behaviour of adult rrn3+/− zebrafish was not significantly different compared with rrn3+/+ zebrafish. Notably, rrn3+/− zebrafish demonstrated abnormal locomotor activity in response to ethanol. We found decreased norepinephrine expression in the brains of rrn3+/− zebrafish when treated with ethanol. In summary, our results indicated that rrn3 was closely associated with early embryonic development in zebrafish. Furthermore, behavioural and neurochemical research revealed the importance of genetic differences in drug sensitivity. The results suggest that caution should be taken when treating RRN3 heterozygous patients.





Similar content being viewed by others
Abbreviations
- CNVs:
-
Copy number variations
- NE:
-
Norepinephrine
- WISH:
-
Whole-mount in situ hybridization
- dpf:
-
Days post-fertilization
- mpf:
-
Months post-fertilization
- gRNA:
-
Guide RNA
- RT-qPCR:
-
Real-time quantitative polymerase chain reaction
- HPLC:
-
High-precision liquid chromatography
- CPP:
-
Conditioned place preference
References
Ali S, Champagne DL, Alia A, Richardson MK (2011) Large-scale analysis of acute ethanol exposure in zebrafish development: a critical time window and resilience. PLoS One 6:e20037
Banaschewski T, Dopfner M (2014) DMS-5 - attention-deficit/hyperactivity disorder. Z Kinder Jugendpsychiatr Psychother 42:271–275 (quiz 276-277)
Brownstein CA, Kleiman RJ, Engle EC, Towne MC, D’Angelo EJ, Yu TW, Beggs AH, Picker J, Fogler JM, Carroll D, Schmitt RC, Wolff RR, Shen Y, Lip V, Bilguvar K, Kim A, Tembulkar S, O’Donnell K, Gonzalez-Heydrich J (2016) Overlapping 16p13.11 deletion and gain of copies variations associated with childhood onset psychosis include genes with mechanistic implications for autism associated pathways: Two case reports. Am J Med Genet A 170A:1165–1173
Cavanaugh AH, Evans A, Rothblum LI (2008) Mammalian Rrn3 is required for the formation of a transcription competent preinitiation complex containing RNA polymerase I. Gene Expr 14:131–147
Chatterjee D, Gerlai R (2009) High precision liquid chromatography analysis of dopaminergic and serotoninergic responses to acute alcohol exposure in zebrafish. Behav Brain Res 200:208–213
de Abreu MS, Genario R, Giacomini A, Demin KA, Lakstygal AM, Amstislavskaya TG, Fontana BD, Parker MO, Kalueff AV (2020) Zebrafish as a Model of Neurodevelopmental Disorders. Neuroscience 445:3–11
Dlugos CA, Brown SJ, Rabin RA (2011) Gender differences in ethanol-induced behavioral sensitivity in zebrafish. Alcohol 45:11–18
Dwivedi S, Medishetti R, Rani R, Sevilimedu A, Kulkarni P, Yogeeswari P (2019) Larval zebrafish model for studying the effects of valproic acid on neurodevelopment: An approach towards modeling autism. J Pharmacol Toxicol Methods 95:56–65
Evsyukov V, Domanskyi A, Bierhoff H, Gispert S, Mustafa R, Schlaudraff F, Liss B, Parlato R (2017) Genetic mutations linked to Parkinson’s disease differentially control nucleolar activity in pre-symptomatic mouse models. Dis Model Mech 10:633–643
Gerlai R, Chatterjee D, Pereira T, Sawashima T, Krishnannair R (2009) Acute and chronic alcohol dose: population differences in behavior and neurochemistry of zebrafish. Genes Brain Behav 8:586–599
Gerlai R, Lahav M, Guo S, Rosenthal A (2000) Drinks like a fish: zebra fish (Danio rerio) as a behavior genetic model to study alcohol effects. Pharmacol Biochem Behav 67:773–782
Grewal SS, Evans JR, Edgar BA (2007) Drosophila TIF-IA is required for ribosome synthesis and cell growth and is regulated by the TOR pathway. J Cell Biol 179:1105–1113
Gumuslu KE, Savli H, Sunnetci D, Cine N, Kara B, Eren Keskin S, Akkoyunlu RU (2015) A CGH array study in nonsyndromic (primary) autism patients: deletions on 16p13.11, 16p11.2, 1q21.1, 2q21.1q21.2, and 8p23.1. Turk J Med Sci 45:313–319
Hwang WY, Fu Y, Reyon D, Maeder ML, Tsai SQ, Sander JD, Peterson RT, Yeh JR, Joung JK (2013) Efficient genome editing in zebrafish using a CRISPR-Cas system. Nat Biotechnol 31:227–229
Jao LE, Wente SR, Chen W (2013) Efficient multiplex biallelic zebrafish genome editing using a CRISPR nuclease system. Proc Natl Acad Sci U S A 110:13904–13909
Kalueff AV, Stewart AM, Gerlai R (2014) Zebrafish as an emerging model for studying complex brain disorders. Trends Pharmacol Sci 35:63–75
Kessler RC, Angermeyer M, Anthony JC, De Graaf R, Demyttenaere K, Gasquet I, De Girolamo G, Gluzman S, Gureje O, Haro JM, Kawakami N, Karam A, Levinson D, Medina Mora ME, Oakley Browne MA, Posada-Villa J, Stein DJ, Adley Tsang CH, Aguilar-Gaxiola S, Alonso J, Lee S, Heeringa S, Pennell BE, Berglund P, Gruber MJ, Petukhova M, Chatterji S, Ustun TB (2007) Lifetime prevalence and age-of-onset distributions of mental disorders in the World Health Organization’s World Mental Health Survey Initiative. World Psychiatry 6:168–176
Kim OH, Cho HJ, Han E, Hong TI, Ariyasiri K, Choi JH, Hwang KS, Jeong YM, Yang SY, Yu K, Park DS, Oh HW, Davis EE, Schwartz CE, Lee JS, Kim HG, Kim CH (2017) Zebrafish knockout of down syndrome gene, DYRK1A, shows social impairments relevant to autism. Mol Autism 8:50
Kimmel CB, Ballard WW, Kimmel SR, Ullmann B, Schilling TF (1995) Stages of embryonic development of the zebrafish. Dev Dyn 203:253–310
Kreiner G, Bierhoff H, Armentano M, Rodriguez-Parkitna J, Sowodniok K, Naranjo JR, Bonfanti L, Liss B, Schütz G, Grummt I, Parlato R (2013) A neuroprotective phase precedes striatal degeneration upon nucleolar stress. Cell Death Differ 20:1455–1464
Lange M, Norton W, Coolen M, Chaminade M, Merker S, Proft F, Schmitt A, Vernier P, Lesch KP, Bally-Cuif L (2012) The ADHD-linked gene Lphn3.1 controls locomotor activity and impulsivity in zebrafish. Mol Psychiatry 17:855
Lovinger DM (1997) Serotonin’s role in alcohol’s effects on the brain. Alcohol Health Res World 21:114–120
Mali P, Yang L, Esvelt KM, Aach J, Guell M, DiCarlo JE, Norville JE, Church GM (2013) RNA-guided human genome engineering via Cas9. Science 339:823–826
Mandt BH, Larson C, Fay T, Bludeau P, Allen RM, Deitrich RA, Radcliffe RA (2018) Quantitative trait loci for sensitivity to acute ethanol and ethanol consummatory behaviors in rats. Alcohol 66:55–67
Mayer C, Zhao J, Yuan X, Grummt I (2004) mTOR-dependent activation of the transcription factor TIF-IA links rRNA synthesis to nutrient availability. Genes Dev 18:423–434
Miller G, Panov KI, Friedrich JK, Trinkle-Mulcahy L, Lamond AI, Zomerdijk JC (2001) hRRN3 is essential in the SL1-mediated recruitment of RNA Polymerase I to rRNA gene promoters. EMBO J 20:1373–1382
Miller N, Greene K, Dydinski A, Gerlai R (2013) Effects of nicotine and alcohol on zebrafish (Danio rerio) shoaling. Behav Brain Res 240:192–196
Miller NY, Gerlai R (2011) Shoaling in zebrafish: what we don’t know. Rev Neurosci 22:17–25
Morozova TV, Mackay TF, Anholt RR (2014) Genetics and genomics of alcohol sensitivity. Mol Gen Genomics : MGG 289:253–269
Orger MB, de Polavieja GG (2017) Zebrafish behavior: opportunities and Challenges. Annu Rev Neurosci 40:125–147
Panach K, Garg A, Ahmad Z (2017) Heterozygous null LDLR mutation in a familial hypercholesterolemia patient with an atypical presentation because of alcohol abuse. Circ Cardiovasc Genet 10:e001767
Parlato R, Kreiner G, Erdmann G, Rieker C, Stotz S, Savenkova E, Berger S, Grummt I, Schutz G (2008) Activation of an endogenous suicide response after perturbation of rRNA synthesis leads to neurodegeneration in mice. J Neurosci 28:12759–12764
Pautassi RM, Suarez AB, Hoffmann LB, Rueda AV, Rae M, Marianno P, Camarini R (2017) Effects of environmental enrichment upon ethanol-induced conditioned place preference and pre-frontal BDNF levels in adolescent and adult mice. Sci Rep 7:8574
Peng J, Wagle M, Mueller T, Mathur P, Lockwood BL, Bretaud S, Guo S (2009) Ethanol-modulated camouflage response screen in zebrafish uncovers a novel role for cAMP and extracellular signal-regulated kinase signaling in behavioral sensitivity to ethanol. J Neurosci 29:8408–8418
Rieker C, Engblom D, Kreiner G, Domanskyi A, Schober A, Stotz S, Neumann M, Yuan X, Grummt I, Schutz G, Parlato R (2011) Nucleolar disruption in dopaminergic neurons leads to oxidative damage and parkinsonism through repression of mammalian target of rapamycin signaling. J Neurosci 31:453–460
Schuckit MA (2000) Genetics of the risk for alcoholism. Am J Addict 9:103–112
Stepanchick A, Zhi H, Cavanaugh AH, Rothblum K, Schneider DA, Rothblum LI (2013) DNA binding by the ribosomal DNA transcription factor rrn3 is essential for ribosomal DNA transcription. J Biol Chem 288:9135–9144
Sumbre G, de Polavieja GG (2014) The world according to zebrafish: how neural circuits generate behavior. Front Neural Circuits 8:91
Tran S, Gerlai R (2013) Time-course of behavioural changes induced by ethanol in zebrafish (Danio rerio). Behav Brain Res 252:204–213
Tropeano M, Ahn JW, Dobson RJ, Breen G, Rucker J, Dixit A, Pal DK, McGuffin P, Farmer A, White PS, Andrieux J, Vassos E, Ogilvie CM, Curran S, Collier DA (2013) Male-biased autosomal effect of 16p13.11 copy number variation in neurodevelopmental disorders. PLoS One 8:e61365
Tupala E, Tiihonen J (2004) Dopamine and alcoholism: neurobiological basis of ethanol abuse. Prog Neuropsychopharmacol Biol Psychiatry 28:1221–1247
Vengeliene V, Bilbao A, Molander A, Spanagel R (2008) Neuropharmacology of alcohol addiction. Br J Pharmacol 154:299–315
Walker ER, McGee RE, Druss BG (2015) Mortality in mental disorders and global disease burden implications: a systematic review and meta-analysis. JAMA Psychiat 72:334–341
Yuan X, Zhao J, Zentgraf H, Hoffmann-Rohrer U, Grummt I (2002) Multiple interactions between RNA polymerase I, TIF-IA and TAF(I) subunits regulate preinitiation complex assembly at the ribosomal gene promoter. EMBO Rep 3:1082–1087
Yuan X, Zhou Y, Casanova E, Chai M, Kiss E, Grone HJ, Schutz G, Grummt I (2005) Genetic inactivation of the transcription factor TIF-IA leads to nucleolar disruption, cell cycle arrest, and p53-mediated apoptosis. Mol Cell 19:77–87
Funding
This work was supported by grants from the National Natural Science Foundation of China (NSFC, No. 81771632 and No. 81271509) to Qiang Li and (NSFC, No. 81601329) to Xiuyun Liu as well as Natural Science Foundation of Shanghai (21ZR1410100) to Qiang Li.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, F., Lin, J., Li, T. et al. Rrn3 gene knockout affects ethanol-induced locomotion in adult heterozygous zebrafish. Psychopharmacology 239, 621–630 (2022). https://doi.org/10.1007/s00213-021-06056-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-021-06056-7