Abstract
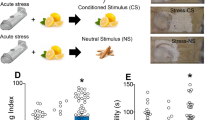
Post-traumatic stress disorder (PTSD) is a psychiatric illness that can increase the risk for developing an alcohol use disorder (AUD). While clinical data has been useful in identifying similarities in the neurobiological bases of these disorders, preclinical models are essential for understanding the mechanism(s) by which stressors increase the risk for escalated alcohol consumption. The purpose of these studies was to examine if exposure of male Long-Evans rats to the synthetically derived predator odor 2,5-dihydro-2,4,5-trimethylthiazoline (TMT; a component of fox feces) would increase sweetened alcohol self-administration, potentially by facilitating transfer of salience towards cues, and alter neuronal response to alcohol as measured by the immediate early gene c-Fos. In experiment 1, rats exposed to repeated (4×) TMT showed reductions in port entries in Pavlovian conditioned approach and increases in sweetened alcohol self-administration. In experiment 2, rats exposed to repeated TMT showed blunted basolateral amygdala c-Fos response to alcohol. In experiment 3, rats exposed to single, but not repeated TMT, showed increases in sweetened alcohol self-administration, and no change in anxiety-like behavior or hyperarousal. In experiment 4, rats continued to show a significant corticosterone response to TMT after repeated exposures. In summary, exposure of male rats to TMT can cause escalations in sweetened alcohol self-administration and reduction in BLA response to alcohol. These studies outline and utilize a novel preclinical model that can be used to further neurobiological understanding of the emergence of escalated alcohol consumption following stress exposure.






Similar content being viewed by others
References
APA (2013) Diagnostic and statistical manual of mental disorders, 5th edn. American Psychiatric Association, Washington DC
Apfelbach R, Soini HA, Vasilieva NY, Novotny MV (2015) Behavioral responses of predator-naive dwarf hamsters (Phodopus campbelli) to odor cues of the European ferret fed with different prey species. Physiol Behav 146:57–66. https://doi.org/10.1016/j.physbeh.2015.04.014
Asok A, Ayers LW, Awoyemi B, Schulkin J, Rosen JB (2013) Immediate early gene and neuropeptide expression following exposure to the predator odor 2,5-dihydro-2,4,5-trimethylthiazoline (TMT). Behav Brain Res 248:85–93. https://doi.org/10.1016/j.bbr.2013.03.047
Bangasser DA, Valentino RJ (2014) Sex differences in stress-related psychiatric disorders: neurobiological perspectives. Front Neuroendocrinol 35:303–319. https://doi.org/10.1016/j.yfrne.2014.03.008
Barone FC, Deegan JF, Price WJ, Fowler PJ, Fondacaro JD, Ormsbee HS 3rd (1990) Cold-restraint stress increases rat fecal pellet output and colonic transit. Am J Phys 258:G329–G337. https://doi.org/10.1152/ajpgi.1990.258.3.G329
Besheer J, Fisher KR, Grondin JJ, Cannady R, Hodge CW (2012) The effects of repeated corticosterone exposure on the interoceptive effects of alcohol in rats. Psychopharmacology 220:809–822. https://doi.org/10.1007/s00213-011-2533-8
Besheer J, Fisher KR, Lindsay TG, Cannady R (2013) Transient increase in alcohol self-administration following a period of chronic exposure to corticosterone. Neuropharmacology 72:139–147. https://doi.org/10.1016/j.neuropharm.2013.04.036
Besheer J, Fisher KR, Jaramillo AA, Frisbee S, Cannady R (2014) Stress hormone exposure reduces mGluR5 expression in the nucleus accumbens: functional implications for interoceptive sensitivity to alcohol. Neuropsychopharmacology 39:2376–2386. https://doi.org/10.1038/npp.2014.85
Blaine SK, Sinha R (2017) Alcohol, stress, and glucocorticoids: from risk to dependence and relapse in alcohol use disorders. Neuropharmacology 122:136–147. https://doi.org/10.1016/j.neuropharm.2017.01.037
Blanchard DC, Markham C, Yang M, Hubbard D, Madarang E, Blanchard RJ (2003) Failure to produce conditioning with low-dose trimethylthiazoline or cat feces as unconditioned stimuli. Behav Neurosci 117:360–368. https://doi.org/10.1037/0735-7044.117.2.360
Brodnik ZD, Black EM, Clark MJ, Kornsey KN, Snyder NW, Espana RA (2017) Susceptibility to traumatic stress sensitizes the dopaminergic response to cocaine and increases motivation for cocaine. Neuropharmacology 125:295–307. https://doi.org/10.1016/j.neuropharm.2017.07.032
Dielenberg RA, McGregor IS (2001) Defensive behavior in rats towards predatory odors: a review. Neurosci Biobehav Rev 25:597–609. https://doi.org/10.1016/s0149-7634(01)00044-6
Dopfel D, Perez PD, Verbitsky A, Bravo-Rivera H, Ma Y, Quirk GJ, Zhang N (2019) Individual variability in behavior and functional networks predicts vulnerability using an animal model of PTSD. Nat Commun 10:2372. https://doi.org/10.1038/s41467-019-09926-z
Edwards S, Baynes BB, Carmichael CY, Zamora-Martinez ER, Barrus M, Koob GF, Gilpin NW (2013) Traumatic stress reactivity promotes excessive alcohol drinking and alters the balance of prefrontal cortex-amygdala activity. Transl Psychiatry 3:e296. https://doi.org/10.1038/tp.2013.70
Endres T, Fendt M (2009) Aversion- vs fear-inducing properties of 2,4,5-trimethyl-3-thiazoline, a component of fox odor, in comparison with those of butyric acid. J Exp Biol 212:2324–2327. https://doi.org/10.1242/jeb.028498
Fendt M, Endres T (2008) 2,3,5-Trimethyl-3-thiazoline (TMT), a component of fox odor-just repugnant or really fear-inducing? Neurosci Biobehav Rev 32:1259–1266. https://doi.org/10.1016/j.neubiorev.2008.05.010
Fenster RJ, Lebois LAM, Ressler KJ, Suh J (2018) Brain circuit dysfunction in post-traumatic stress disorder: from mouse to man. Nat Rev Neurosci 19:535–551. https://doi.org/10.1038/s41583-018-0039-7
Finn DA, Helms ML, Nipper MA, Cohen A, Jensen JP, Devaud LL (2018) Sex differences in the synergistic effect of prior binge drinking and traumatic stress on subsequent ethanol intake and neurochemical responses in adult C57BL/6J mice. Alcohol 71:33–45. https://doi.org/10.1016/j.alcohol.2018.02.004
Fitzpatrick CJ, Morrow JD (2016) Pavlovian conditioned approach training in rats. J Vis Exp:e53580. https://doi.org/10.3791/53580
Fitzpatrick CJ et al (2013) Variation in the form of Pavlovian conditioned approach behavior among outbred male Sprague-Dawley rats from different vendors and colonies: sign-tracking vs. goal-tracking. PLoS One 8:e75042. https://doi.org/10.1371/journal.pone.0075042
Fitzpatrick CJ, Geary T, Creeden JF, Morrow JD (2019) Sign-tracking behavior is difficult to extinguish and resistant to multiple cognitive enhancers. Neurobiol Learn Mem 163:107045. https://doi.org/10.1016/j.nlm.2019.107045
Gilpin NW, Weiner JL (2017) Neurobiology of comorbid post-traumatic stress disorder and alcohol-use disorder. Genes Brain Behav 16:15–43. https://doi.org/10.1111/gbb.12349
Hwa LS, Neira S, Pina MM, Pati D, Calloway R, Kash TL (2019) Predator odor increases avoidance and glutamatergic synaptic transmission in the prelimbic cortex via corticotropin-releasing factor receptor 1 signaling. Neuropsychopharmacology 44:766–775. https://doi.org/10.1038/s41386-018-0279-2
Jacobsen LK, Southwick SM, Kosten TR (2001) Substance use disorders in patients with posttraumatic stress disorder: a review of the literature. Am J Psychiatry 158:1184–1190. https://doi.org/10.1176/appi.ajp.158.8.1184
Jaramillo AA, Randall PA, Frisbee S, Fisher KR, Besheer J (2015) Activation of mGluR2/3 following stress hormone exposure restores sensitivity to alcohol in rats. Alcohol 49:525–532. https://doi.org/10.1016/j.alcohol.2015.03.008
Jaramillo AA, Randall PA, Frisbee S, Besheer J (2016) Modulation of sensitivity to alcohol by cortical and thalamic brain regions. Eur J Neurosci 44:2569–2580. https://doi.org/10.1111/ejn.13374
Jaramillo AA, Randall PA, Stewart S, Fortino B, Van Voorhies K, Besheer J (2018) Functional role for cortical-striatal circuitry in modulating alcohol self-administration. Neuropharmacology 130:42–53. https://doi.org/10.1016/j.neuropharm.2017.11.035
Kessler RC, Crum RM, Warner LA, Nelson CB, Schulenberg J, Anthony JC (1997) Lifetime co-occurrence of DSM-III-R alcohol abuse and dependence with other psychiatric disorders in the National Comorbidity Survey. Arch Gen Psychiatry 54:313–321. https://doi.org/10.1001/archpsyc.1997.01830160031005
King CE, Becker HC (2019) Oxytocin attenuates stress-induced reinstatement of alcohol seeking behavior in male and female mice. Psychopharmacology 236:2613–2622. https://doi.org/10.1007/s00213-019-05233-z
Koob GF, Volkow ND (2016) Neurobiology of addiction: a neurocircuitry analysis. Lancet Psychiatry 3:760–773. https://doi.org/10.1016/S2215-0366(16)00104-8
Leriche M, Mendez M, Zimmer L, Berod A (2008) Acute ethanol induces Fos in GABAergic and non-GABAergic forebrain neurons: a double-labeling study in the medial prefrontal cortex and extended amygdala. Neuroscience 153:259–267. https://doi.org/10.1016/j.neuroscience.2008.01.069
Liberzon I, Sripada CS (2008) The functional neuroanatomy of PTSD: a critical review. Prog Brain Res 167:151–169. https://doi.org/10.1016/S0079-6123(07)67011-3
Makhijani VH, Van Voorhies K, Besheer J (2018) The mineralocorticoid receptor antagonist spironolactone reduces alcohol self-administration in female and male rats. Pharmacol Biochem Behav 175:10–18. https://doi.org/10.1016/j.pbb.2018.07.011
Manjoch H, Vainer E, Matar M, Ifergane G, Zohar J, Kaplan Z, Cohen H (2016) Predator-scent stress, ethanol consumption and the opioid system in an animal model of PTSD. Behav Brain Res 306:91–105. https://doi.org/10.1016/j.bbr.2016.03.009
Mattina MJ, Pignatello JJ, Swihart RK (1991) Identification of volatile components of bobcat (Lynx rufus) urine. J Chem Ecol 17:451–462. https://doi.org/10.1007/BF00994344
McGregor IS, Schrama L, Ambermoon P, Dielenberg RA (2002) Not all ‘predator odours’ are equal: cat odour but not 2,4,5 trimethylthiazoline (TMT; fox odour) elicits specific defensive behaviours in rats. Behav Brain Res 129:1–16. https://doi.org/10.1016/s0166-4328(01)00324-2
Meyer EM, Long V, Fanselow MS, Spigelman I (2013) Stress increases voluntary alcohol intake, but does not alter established drinking habits in a rat model of posttraumatic stress disorder. Alcohol Clin Exp Res 37:566–574. https://doi.org/10.1111/acer.12012
Monnikes H, Schmidt BG, Tache Y (1993) Psychological stress-induced accelerated colonic transit in rats involves hypothalamic corticotropin-releasing factor. Gastroenterology 104:716–723. https://doi.org/10.1016/0016-5085(93)91006-4
Morrison SE, Bamkole MA, Nicola SM (2015) Sign tracking, but not goal tracking, is resistant to outcome devaluation. Front Neurosci 9:468. https://doi.org/10.3389/fnins.2015.00468
Morrow JD, Maren S, Robinson TE (2011) Individual variation in the propensity to attribute incentive salience to an appetitive cue predicts the propensity to attribute motivational salience to an aversive cue. Behav Brain Res 220:238–243. https://doi.org/10.1016/j.bbr.2011.02.013
Patel R, Girard TA, Pukay-Martin N, Monson C (2016) Preferential recruitment of the basolateral amygdala during memory encoding of negative scenes in posttraumatic stress disorder. Neurobiol Learn Mem 130:170–176. https://doi.org/10.1016/j.nlm.2016.02.003
Pati D, Pina MM, Kash TL (2019) Ethanol-induced conditioned place preference and aversion differentially alter plasticity in the bed nucleus of stria terminalis. Neuropsychopharmacology 44:1843–1854. https://doi.org/10.1038/s41386-019-0349-0
Perez-Gomez A et al (2015) Innate predator odor aversion driven by parallel olfactory subsystems that converge in the ventromedial hypothalamus. Curr Biol 25:1340–1346. https://doi.org/10.1016/j.cub.2015.03.026
Piggott VM et al (2019) Single-prolonged stress impairs prefrontal cortex control of amygdala and striatum in rats. Front Behav Neurosci 13:18. https://doi.org/10.3389/fnbeh.2019.00018
Randall PA, Stewart RT, Besheer J (2017) Sex differences in alcohol self-administration and relapse-like behavior in Long-Evans rats. Pharmacol Biochem Behav 156:1–9. https://doi.org/10.1016/j.pbb.2017.03.005
Randall PA, McElligott ZA, Besheer J (2019a) Role of mPFC and nucleus accumbens circuitry in modulation of a nicotine plus alcohol compound drug state. Addict Biol:e12782. https://doi.org/10.1111/adb.12782
Randall PA, Vetreno RP, Makhijani VH, Crews FT, Besheer J (2019b) The Toll-like receptor 3 agonist Poly(I:C) induces rapid and lasting changes in gene expression related to glutamatergic function and increases ethanol self-administration in rats. Alcohol Clin Exp Res 43:48–60. https://doi.org/10.1111/acer.13919
Rau V, Fanselow MS (2009) Exposure to a stressor produces a long lasting enhancement of fear learning in rats. Stress 12:125–133. https://doi.org/10.1080/10253890802137320
Ryabinin AE, Criado JR, Henriksen SJ, Bloom FE, Wilson MC (1997) Differential sensitivity of c-Fos expression in hippocampus and other brain regions to moderate and low doses of alcohol. Mol Psychiatry 2:32–43. https://doi.org/10.1038/sj.mp.4000206
Substance Abuse and Mental Health Services Administration (SAMHSA). (2019). Key substance use and mental health indicators in the United States: Results from the 2018 National Survey on Drug Use and Health (HHS Publication No. PEP19-5068, NSDUH Series H-54). Rockville, MD: Center for Behavioral Health Statistics and Quality, Substance Abuse and Mental Health Services Administration. Retrieved from https://www.samhsa.gov/data
Schmidt KT et al (2019) Stress-induced alterations of norepinephrine release in the bed nucleus of the stria terminalis of mice. ACS Chem Neurosci 10:1908–1914. https://doi.org/10.1021/acschemneuro.8b00265
Schreiber AL, McGinn MA, Edwards S, Gilpin NW (2019) Predator odor stress blunts alcohol conditioned aversion. Neuropharmacology 144:82–90. https://doi.org/10.1016/j.neuropharm.2018.10.019
Schwendt M et al (2018) A novel rat model of comorbid PTSD and addiction reveals intersections between stress susceptibility and enhanced cocaine seeking with a role for mGlu5 receptors. Transl Psychiatry 8:209. https://doi.org/10.1038/s41398-018-0265-9
Seo D, Sinha R (2014) The neurobiology of alcohol craving and relapse. Handb Clin Neurol 125:355–368. https://doi.org/10.1016/B978-0-444-62619-6.00021-5
Sharp BM (2017) Basolateral amygdala and stress-induced hyperexcitability affect motivated behaviors and addiction. Transl Psychiatry 7:e1194. https://doi.org/10.1038/tp.2017.161
Shorter D, Hsieh J, Kosten TR (2015) Pharmacologic management of comorbid post-traumatic stress disorder and addictions. Am J Addict 24:705–712. https://doi.org/10.1111/ajad.12306
Sinha R, Fox HC, Hong KI, Hansen J, Tuit K, Kreek MJ (2011) Effects of adrenal sensitivity, stress- and cue-induced craving, and anxiety on subsequent alcohol relapse and treatment outcomes. Arch Gen Psychiatry 68:942–952. https://doi.org/10.1001/archgenpsychiatry.2011.49
Sripada CS, Angstadt M, McNamara P, King AC, Phan KL (2011) Effects of alcohol on brain responses to social signals of threat in humans. Neuroimage 55:371–380. https://doi.org/10.1016/j.neuroimage.2010.11.062
Stafford D, LeSage MG, Glowa JR (1998) Progressive-ratio schedules of drug delivery in the analysis of drug self-administration: a review. Psychopharmacology 139:169–184. https://doi.org/10.1007/s002130050702
Tomie A, Grimes KL, Pohorecky LA (2008) Behavioral characteristics and neurobiological substrates shared by Pavlovian sign-tracking and drug abuse. Brain Res Rev 58:121–135. https://doi.org/10.1016/j.brainresrev.2007.12.003
Valyear MD, Villaruel FR, Chaudhri N (2017) Alcohol-seeking and relapse: a focus on incentive salience and contextual conditioning. Behav Process 141:26–32. https://doi.org/10.1016/j.beproc.2017.04.019
VanElzakker MB, Dahlgren MK, Davis FC, Dubois S, Shin LM (2014) From Pavlov to PTSD: the extinction of conditioned fear in rodents, humans, and anxiety disorders. Neurobiol Learn Mem 113:3–18. https://doi.org/10.1016/j.nlm.2013.11.014
Vilpoux C, Warnault V, Pierrefiche O, Daoust M, Naassila M (2009) Ethanol-sensitive brain regions in rat and mouse: a cartographic review, using immediate early gene expression. Alcohol Clin Exp Res 33:945–969. https://doi.org/10.1111/j.1530-0277.2009.00916.x
Whitaker AM, Gilpin NW (2015) Blunted hypothalamo-pituitary adrenal axis response to predator odor predicts high stress reactivity. Physiol Behav 147:16–22. https://doi.org/10.1016/j.physbeh.2015.03.033
Yager LM, Robinson TE (2013) A classically conditioned cocaine cue acquires greater control over motivated behavior in rats prone to attribute incentive salience to a food cue. Psychopharmacology 226:217–228. https://doi.org/10.1007/s00213-012-2890-y
Yu L, Wang L, Zhao X, Song M, Wang X (2016) Role of single prolonged stress in acquisition of alcohol conditioned place preference in rats. Life Sci 151:259–263. https://doi.org/10.1016/j.lfs.2016.03.004
Zoladz PR et al (2018) Predator-based psychosocial stress model of PTSD differentially influences voluntary ethanol consumption depending on methodology. Alcohol 70:33–41. https://doi.org/10.1016/j.alcohol.2018.01.004
Funding
This work was supported by the National Institute of Health AA026537 (JB), AA011605(JB) and by the Bowles Center for Alcohol Studies. VHM was supported by AA027436 and NS007431, and JPF was supported by GM089569.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All procedures were carried out in accordance with the NIH Guide for Care and Use of Laboratory Animals, and institutional guidelines. All protocols were approved by the UNC Institutional Animal Care and Use Committee (IACUC). UNC-Chapel Hill is accredited by the Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC).
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Makhijani, V.H., Franklin,, J.P., Van Voorhies, K. et al. The synthetically produced predator odor 2,5-dihydro-2,4,5-trimethylthiazoline increases alcohol self-administration and alters basolateral amygdala response to alcohol in rats. Psychopharmacology 238, 67–82 (2021). https://doi.org/10.1007/s00213-020-05659-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-020-05659-w